Randomized Embedded Multifactorial Adaptive Platform trial for Community

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia Background & Design

REMAP-CAP Design • Designed specifically for a pandemic • Need answers rapidly in a new disease • International

REMAP-CAP • Randomised • Rather than random clinician choice • Embedded • Recruitment by clinical staff (and / or research coordinators) • Deliver intervention using usual clinical processes • Multifactorial • Simultaneously assess different aspects of treatment • • • Therapeutic anticoagulation (ward and ICU) Antivirals Immunomodulation Immunoglobulin therapy Vitamin C & Statins (coming soon) • Adaptive
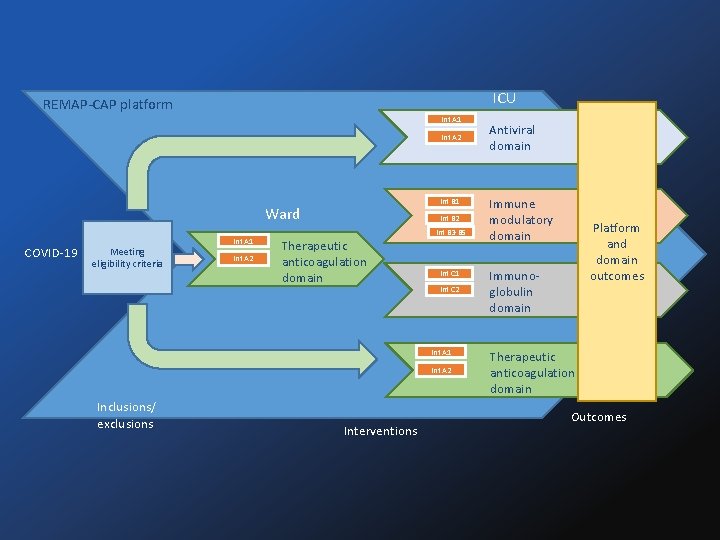
ICU REMAP-CAP platform Int A 1 Int A 2 Int B 1 Ward COVID-19 Meeting eligibility criteria Int A 1 Int A 2 Int B 2 Therapeutic anticoagulation domain Int B 3 -B 5 Int C 1 Int C 2 Int A 1 Int A 2 Inclusions/ exclusions Interventions Antiviral domain Immune modulatory domain Platform and domain outcomes Immunoglobulin domain Therapeutic anticoagulation domain Outcomes

REMAP-CAP • Randomised • Embedded • Multifactorial • Adaptive • Frequent Bayesian adaptive analyses • Response adaptive randomisation • Add / subtract domains and intervention • Platform Trial • All patients with COVID-19 included

Response Adaptive Randomisation • RAR varies randomisation proportions as trial data or interim analyses accrue • Higher probability an intervention is superior increases the proportion assigned for next week • Higher probability an intervention is inferior decreases the proportion assigned for next week • More likely to receive the most beneficial treatment in the trial vs outside trial • If an intervention is superior, trial saves lives

Analysis and RAR during pandemic • Statistical model evaluates all pandemic domains • Evaluates patients who are confirmed COVID-19 and not confirmed COVID 19 separately with borrowing between stratum, as appropriate • Reduction in statistical trigger from 0. 99 to 0. 95 probability of superiority • RAR driven off confirmed COVID-19 for future suspected and confirmed patients • Assesses safety in suspected patients

Primary end-point Days alive and not receiving organ support in ICU at Day 21 • Censored at hospital discharge (no need to follow-up after hospital discharge until usual D 90) • It is VITAL that D 21 endpoint is entered as soon as possible after the end of study day 21 • D 90 follow up still required • D 180 follow up not required for patients

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia Platform eligibility

Pandemic platform eligibility Platform inclusion criteria: 1. Adult patient admitted to hospital with acute illness due to suspected or proven pandemic infection Platform exclusion criteria: 1. Death is deemed to be imminent or inevitable during the next 24 hours AND one or more of the patient, substitute decision maker or attending physician are not committed to full active treatment 2. Patient expected to be discharged from hospital today or tomorrow 3. >14 days have elapsed while admitted to hospital with symptoms of acute illness due to suspected/proven pandemic infection 4. Previous participation in this REMAP within the last 90 days

Inclusion definitions Does the patient have clinically suspected or proven pandemic infection • Clinically suspected = treating clinician believes that pandemic infection is a likely diagnosis • That testing is being done is not sufficient, must think pandemic infection is likely • Suspected or proven hospital-acquired pandemic infection are eligible

Changes to eligibility The following platform eligibility criteria do not apply for patients with clinically suspected or proven pandemic infection: • Is the patient a resident of a nursing home or long-term care facility • Prior to this illness was the patient known to be an inpatient in any healthcare facility within the last 30 days • Does the patient have signs and/or symptoms that are consistent with lower respiratory tract infection • Does the patient have radiological evidence of new onset infiltrate of infective origin • Time window from hospital admission to ICU admission

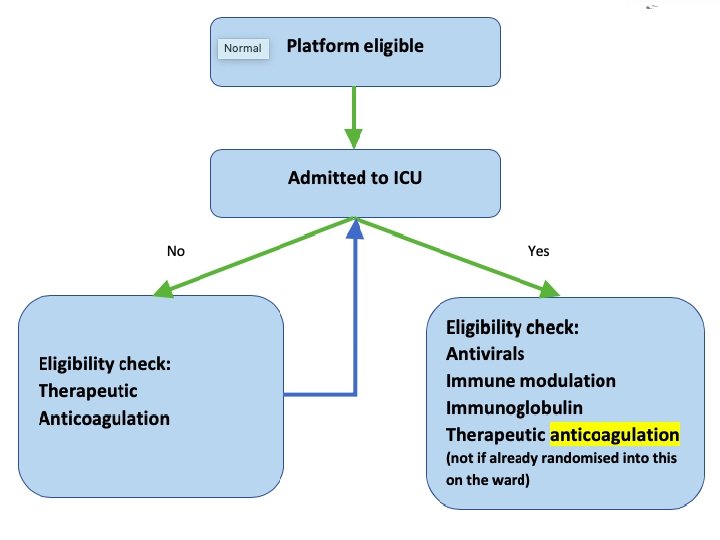
Changes to eligibility Patients may now be eligible in both Moderate and Severe disease states: • Severe State = receiving organ support in ICU • Moderate State = • • In ICU but not receiving organ support Not in ICU Patients may receive allocations while in Moderate state and later receive an allocation to other domains if then progress to Severe state. Patients in Severe state cannot receive

Inclusion definitions ICU • ICU is defined as an area that is capable of providing ICU-level care: • NIV with a sealed mask, invasive mechanical ventilation, or vasopressors via continuous infusion • Existing wards which provide sealed mask NIV which do not have expanded capabilities do not qualify • May be a physical ICU or an area not usually designated as an ICU which has been re-purposed

Definitions ICU Admission • For patients admitted to a location that is not usually designated as an ICU, admission is the time of first administration of qualifying organ support • Mechanical ventilation • Non-invasive ventilation (including HFNO) • Vasopressors or inotropes via continuous infusion • If the patient is not in a physical ICU, do not commence the eligibility process until patient has a qualifying organ failure
Definitions

ICU level domains to come • Vitamin C • Statins

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia Domain eligibility/intervent ions

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia Therapeutic Anticoagulation Domain

Interventions Available Interventions: • Local standard thromboprophylaxis • Using pharmacological agents • Therapeutic anticoagulation with unfractionated heparin (UFH) or subcutaneous low molecular weight heparin (LMWH) Available to patients in the following States: • Moderate (not in ICU or not receiving organ support in ICU)

Eligibility Inclusion Criteria: • Platform eligible • COVID-19 is suspected by the treating clinician or has been confirmed by microbiological testing • Microbiological testing for SARS-Co. V -2 infection of upper or lower respiratory tract secretions (or both) has occurred or is intended to occur

Eligibility Exclusion Criteria: • >48 hours since ICU admission • Note: >48 h since first organ support, if already randomised in Moderate State. • Clinical or laboratory bleeding risk sufficient to contraindicate therapeutic anticoagulation • Intention to continue or commence dual anti-platelet therapy • Therapeutic anticoagulation is already present, or decision to commence therapeutic anticoagulation

Eligibility Exclusion Criteria: • Enrolment in a trial evaluating anticoagulation for COVID-19 infection, where the protocol requires continuation of the treatment assignment • Known or suspected adverse reaction to UFH or LMWH, including HIT • Treating clinician believes that participation is not in the best

Local standard thromboprophylaxis • Standard venous thromboprophylaxis as per local guidelines • Dose of agent should not be sufficient to result in therapeutic anticoagulation • Administered for 14 days or hospital discharge, whichever occurs first • Administration after day study 14 is at the discretion of the treating clinician

Local standard thromboprophylaxis • Therapeutic anticoagulation can be commenced if patient develops an accepted clinical indication for anticoagulation • DVT, proven PE, acute coronary syndrome, systemic embolic event, intermittent haemodialysis, or SLED • Systemic therapeutic anticoagulation for c. RRT is not permitted, unless there is additional indication for anticoagulation. • Regional low-dose heparin permitted to prevent filter clotting, however dose of

Local standard thromboprophylaxis • If unfractionated heparin is used, doses should be below those expected to achieve therapeutic anticoagulation. • If LMWH is used, standard low dose is preferred, but intermediate thromboprophylaxis dose (e. g. no more than double standard low thromboprophylaxis dose) is acceptable if it has become standard practice at the site.

Therapeutic Anticoagulation • Administer UFH or LMWH to achieve systemic anticoagulation. • As per local hospital policy and guidelines for the treatment of VTE • Either agent may be used, and the same patient may be switched between UFH and LMWH • Cease (whichever occurs first): • On study day 15, • 24 hours after liberation from invasive mechanical ventilation, or • ICU or hospital discharge

Discontinuation of intervention • Study intervention should be discontinued if there is clinical bleeding or other complication • Anticoagulation or local standard venous thromboprophylaxis may be recommenced if deemed appropriate by the treating clinician. • Laboratory-proven HIT must result in cessation of UFH or LMWH without recommencement • Study interventions can be discontinued

Concomitant Care • Additional agents intended to modify the patient’s coagulation as a treatment for COVID-19 infection should not be administered • Commencement of any agent that inhibits platelet function is not permitted unless there is an accepted clinical indication • Patients who receive agent(s) which inhibit platelet function as a usual medication may have this medication continued

End-point Secondary endpoints • Serial detection of SARS-Co. V-2 in upper or lower respiratory tract specimens • using only specimens collected for routine clinically indicated testing • Confirmed VTE or PE • RBC transfused between randomization and end of study day 15

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia COVID-19 Antiviral Domain

Interventions Available Interventions: • No antiviral intended to be active against SARS-Co. V-2 infection (no placebo) • Lopinavir/ritonavir (‘Kaletra’) Available to patients in the following States: • Moderate (not receiving organ support in ICU) • Severe (receiving organ support in ICU)

Eligibility Inclusion Criteria: • Platform eligible

Eligibility Exclusion Criteria: • >24 hours since ICU admission • >36 hours of treatment with any non -trial systemic antiviral intended to be active against COVID-19 during this hospital admission • Participating in a trial evaluating an antiviral intended to be active against COVID-19, AND • Protocol requires ongoing administration of

Eligibility Exclusion Criteria: • The treating clinician believes that participation in this domain is not in the best interests of the patient.

Eligibility Intervention exclusion Criteria: • Known hypersensitivity • Receiving an agent specified as an intervention as a usual medication prior to this hospitalisation • Known HIV infection • Severe liver failure • Receiving amiodarone as usual medication prior to hospitalisation, or administration within last 72 hours

Interventions Lopinavir/ritonavir • Lopinavir/ritonavir 400/100 mg • Administered enterally every 12 hours • Preferred route of administration as two 200/50 mg tablets swallowed whole • If patient cannot swallow: 5 m. L of 80/20 mg per m. L suspension via gastric tube • If suspension is not available: four crushed tablets (double dose) via gastric tube • No dose adjustment for renal dysfunction. Adjust dose for liver failure

Interventions Lopinavir/ritonavir • Administered for minimum 5 days, including after discharge from ICU • If patient is discharged from ICU between study day 6 and study day 14, cease at ICU discharge • If patient remains in ICU, discontinue at end of study day 14 • If a patient is readmitted to ICU prior to the end of study day 14, recommence and stop at end of study day 14

Interventions Concomitant care • Additional agents intended to be active against SARS-Co. V-2 infection should not be administered • Concomitant treatment with drugs known to interact with lopinavir/ritonavir should be avoided • Note amiodarone in particular • List of agents provided as appendix to DSA • If no alternative agent is available, treating clinician will need to chose whether to withhold the interacting

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia Immune Modulation Domain

Interventions Available Interventions: • No immune modulation therapy intended to be active against COVID-19 (no placebo) • Interferon-beta-1 a (IFN-β 1 a) • Anakinra (interleukin-1 receptor antagonist) • Tocilizumab (IL-6 inhibition) • Sarilumab (IL-6 inhibition) Available to patients in the following States: • Moderate (not receiving organ support in ICU)

Eligibility Inclusion Criteria: • Platform eligible

Eligibility Exclusion Criteria: • >24 hours since ICU admission • Patient has already received any form of IFN, anakinra, tocilizumab or sarilumab or is on long-term therapy with any of these agents prior to hospital admission • Known condition/treatment resulting in ongoing immune suppression including neutropaenia prior to this hospitalisation

Eligibility Exclusion Criteria: • Patient has been randomised in a trial evaluating an immune modulation agent for proven or suspected COVID-19 infection • where the protocol of that trial requires ongoing administration of study drug • Treating clinician believes that participation is not in the best interests of the patient

Eligibility Intervention exclusion Criteria: • Known hypersensitivity • Receiving an agent specified as an intervention as a usual medication prior to this hospitalization • Intention to prescribe systemic corticosteroids for any reason other than participation in the Corticosteroid DSA (IFN-β 1 a)

Eligibility Intervention exclusion Criteria: • Known sensitivity to proteins produced by E. coli (anakinra) • Pregnancy / breastfeeding (All) • Baseline alanine aminotransferase or aspartate aminotransferase >5 times upper limit of normal (tocilizumab and sarilumab) • Baseline platelet < 50 x 109 / L (tocilizumab and sarilumab)

Interventions Interferon-beta-1 a (IFN-β 1 a) • IFN-β 1 a 11 mcg will be diluted in 0. 9% saline • Administered as bolus via central or peripheral line • Once daily for 6 days or until ICU discharge, whichever occurs first

Interventions Anakinra (IL-1 Ra) • Loading dose of 300 mg anakinra administered as bolus via central or peripheral line • Subsequent maintenance doses of 100 mg every 6 hours • Creatinine clearance <30 m. L/min or RRT, administered every 12 hours • Patients receiving mechanical ventilation (IMV): • Discontinued once the patient has been off IMV for > 24 hours

Interventions Tociluzimab • Single dose IV infusion at 8 mg/kg, max total dose of 800 mg • Option of a second dose if treating clinician believes there has not been sufficient clinical improvement • 12 to 24 hours after the initial dose

Interventions Sarilumab • Single dose IV infusion at 400 mg

Interventions Concomitant care • No additional immune modulating agents for COVID-19 • No systemic corticosteroids, other than those allocated as part of the Corticosteroid Domain of REMAPCAP if patient allocated to IFN-β 1 a

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia Immunoglobulin Therapy Domain

Interventions Available Interventions: • No immunoglobulin against COVID-19 (no placebo) • Convalescent plasma (standard or intensive sampling) Available to patients in the following States: • Moderate (not receiving organ support in ICU)

Eligibility Inclusion Criteria: • Platform eligible • COVID-19 infection is confirmed by microbiological testing

Eligibility Exclusion Criteria: • More than 48 hours has elapsed since ICU admission • Patient has already received treatment with any non-trial prescribed antibody therapy (monoclonal antibody, hyperimmune immunoglobulin, or convalescent plasma) intended to be active against COVID-19 during this hospital admission • The treating clinician believes that

Eligibility Intervention exclusion Criteria: • Known hypersensitivity/allergy to an agent specified as an intervention in this domain will exclude a patient from receiving that agent • Known previous history of transfusion -related acute lung injury will exclude a patient from receiving convalescent plasma • Known objection to receiving plasma products will exclude a patient from

Interventions Convalescent Plasma • Order from local transfusion laboratory as soon as the allocation is revealed. • Two units per patient: issue separately. There must be a minimum of 12 hours between transfusions and both units should be given within 48 hours of randomisation. • Do not proceed with second unit if any

Interventions Convalescent Plasma • Blood and respiratory samples required on Day 1 before plasma given (for all patients) • Some sites are also taking part in Intensive Sampling • SOPs (Standard and Intensive Sampling) available here • Postage kits for standard sampling provided by NHSBT Clinical Trials

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia Domains coming soon

Vitamin C Domain Available Interventions: • No vitamin C (no placebo) • Vitamin C Available to patients in the following States: • Moderate (not receiving organ support in ICU) • Severe (receiving organ support in ICU)

Statin Domain Available Interventions: • No statins (no plcebo) • Simvastatin Available to patients in the following States: • Moderate (not receiving organ support in ICU) • Severe (receiving organ support in ICU)

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia Randomisation and data collection

Login screen Generic login for site clinical users remapcapds 9+++ https: //remapcap. spinnakersoftw

e. CRF Eligibility Assess eligibility of a new patient
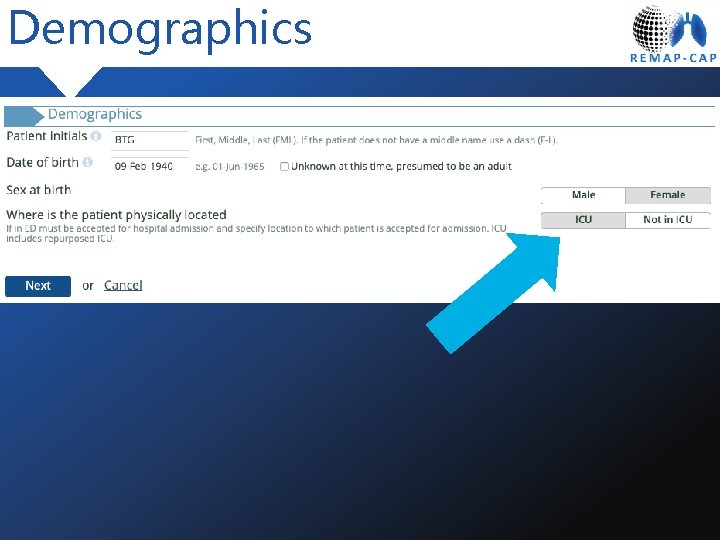
Demographics

Disease and Platform Eligibility

Disease and Platform Eligibility

Disease and Platform Eligibility

Domain and Intervention Eligibility

Domain and Intervention Eligibility
Consent
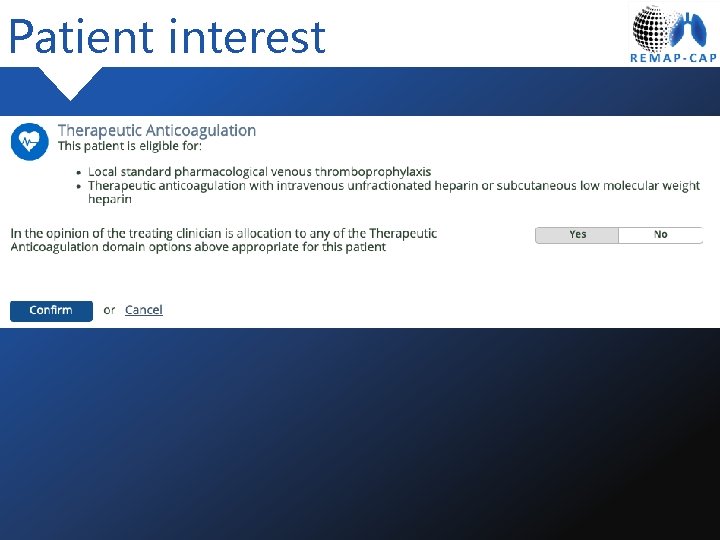
Patient interest

Randomisation
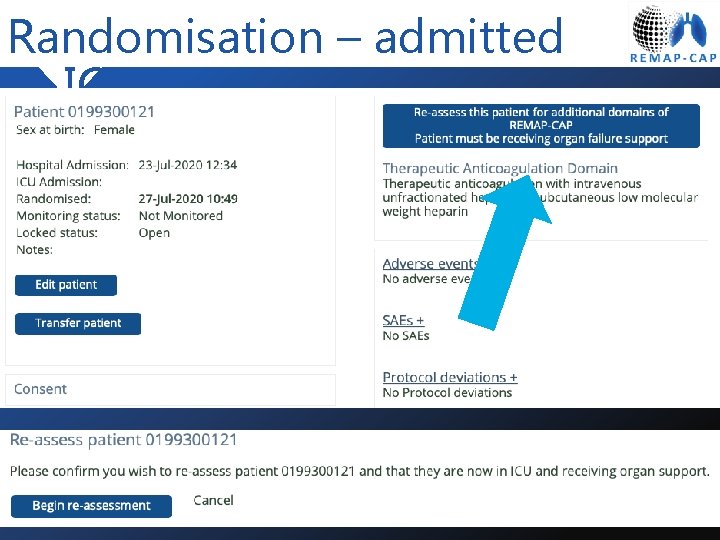
Randomisation – admitted to ICU
Demographics

Disease and Platform Eligibility

Disease and Platform Eligibility

Disease and Platform Eligibility

Disease and Platform Eligibility

Domain and Intervention Eligibility

Domain and Intervention Eligibility

Domain and Intervention Eligibility

Consent

Patient interest
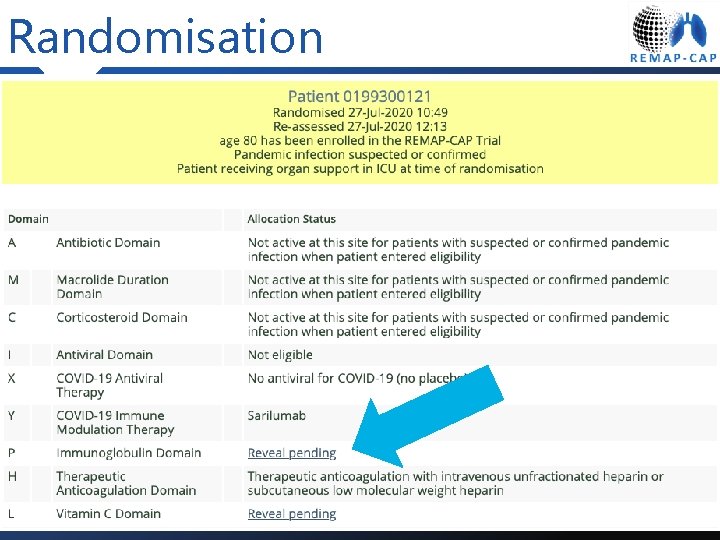
Randomisation

Randomisation
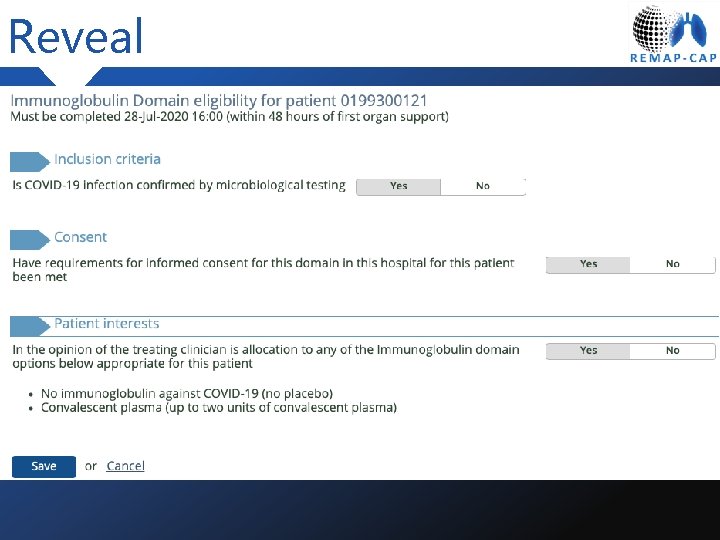
Reveal

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia Consent During the pandemic

Consent Principles • If patient on ward, prospective consent needed • If patient on ICU deferred consent in place – patients can be randomized prior to consent being obtained • Pro. LR to be collected in the first instance • Most patients will not have capacity but will be infectious at the time of enrolment • Per. LRs will be self isolating • Retrospective consent to be gained when noninfectious

Consent Process • Gain Pro. LR • Complete REMAP-CAP Eligibility e. CRF • Document (in the medical record) that the patient did not have capacity to consent When clinically appropriate • Discuss the study with the relative and provide the participant information and consent form • Gain retrospective consent if appropriate • File the signed informed consent form • Fill in data about informed consent in the e. CRF

Data collection • Individual accounts through https: //remapcap. spinnakersoftware. com • Data collected on: • • • Baseline ICU stay Microbiology Discharge Serious adverse events Long-term outcomes

CRF changes • Baseline • Whether the patient is a healthcare worker who has had direct contact with patients with suspected/confirmed COVID-19 within the last 21 days. • Whether the patient is in a physical ICU or area not usually designated as an ICU at eligibility • Additional physiological measurements • Micro CRF • Results of microbiological testing for SARSCo. V-2 (during this acute respiratory illness, up until

CRF changes • Daily data • Location of patient on each study day (physical ICU, or area not usually designated as an ICU) • Corticosteroid administration for all patients with suspected or proven infection • Medication administration • Updated to collect administration of antiviral and immunomodulatory agents that may be active against SARS-Co. V-2

CRF changes • Discharge • Date and time of first and last organ support in ICU • Co-enrolment in another study (study name, participant ID, and allocation if known) • Day 21 • Date and time of ICU discharge and any ICU readmissions prior to day 21 • Hospital discharge date and status (prior to day 21)

SAEs • Report SAE to Sponsor if the event is (suspected to be) related to study medication: • • Use an SAE form in the e. CRF Document in patient file We don’t expect many SAEs to be reported E. g. if a patient dies as a deterioration of COVID-19 not considered SAE

COVID-19 Collaboration • Co-enrolment with RECOVERY, if eligible for REMAP-CAP • If not randomized to RECOVERY • randomize to all available domains • If randomized to RECOVERY • All patients not eligible for antiviral domain • If randomized to tocilizumab, no eligible for immune modulation domain • If randomized to CP, not eligible for immunoglobulin domain

Randomized, Embedded, Multifactorial Adaptive Platform trial for Community. Acquired Pneumonia Start-up

Essential documents • Essential start-up documents checklist • Fully signed contract • Confirmation of capacity and capability • Attendance at training • Delegation log • Database form (domain/intervention chocies) • Needs to be complete in order to be activated

Closing message – to keep in mind • Audit possibilities • Monitoring (central) • Amendments

More information or questions • ukremap-cap@icnarc. org (UK) • prepare_icu@umcutrecht. nl (Sponsor)
Questions? Randomized, Embedded, Multifactorial, Adaptive Platform For Community-Acquired Pneumonia
- Slides: 101