Randomized DoubleBlind Assessment of the ONSET and OFFSet

Randomized Double-Blind Assessment of the ONSET and OFFSet of the Antiplatelet Effects of Ticagrelor Versus Clopidogrel in Patients with Stable Coronary Artery Disease: The ONSET/OFFSET Study Gurbel PA, Bliden KP, Butler K, Tantry US, Gesheff T, Wei C, Teng R, Antonino MJ, Patil SB, Karunakaran A, Kereiakes DJ, Parris C, Purdy D, Wilson V, Ledley GS, Storey RF
Disclosures Research Grants Honoraria/Consultant - Astra Zeneca - Sanofi/Aventis - Portola - Daiichi Sankyo - Pozen - Lilly - Bayer - Schering Plough

Introduction • Clopidogrel: - slow, variable, irreversible pharmacodynamic effect - non-responsiveness ~15% to 30% - non-responsiveness associated with ischemic events - irreversibility problematic: - patients treated prior to angiography then needing CABG - other unanticipated surgery 1 • Ticagrelor (formerly AZD 6140): - first reversibly binding oral, direct-acting P 2 Y 12 antagonist 2 - faster onset and greater IPA compared to low dose clopidogrel (300 mg load) 3, 4 - clinical efficacy studied in ACS (PLATO trial)5 • Onset and Offset of ticagrelor vs. high dose clopidogrel (600 mg load) never comprehensively characterized 1. Gurbel PA et al. Expert Opin Drug Metab Toxicol. 2009; 5: 989 -1004 2. Tantry US et al. Expert Opin Investig Drug. 2007; 16: 225 -229 3. Husted S et al. Eur Heart J. 2006; 27: 1038 -1047. 4. Storey RF et al. J Am Coll Cardiol. 2007; 50: 1852 -1856. 5. Wallentin L et al. N Engl J Med. 2009; 361: 1045 -1057.

Aims of ONSET/OFFSET Study • To determine the onset and offset of the antiplatelet effect of ticagrelor using the PLATO trial dose compared to high loading dose clopidogrel and placebo in stable CAD patients on background aspirin therapy • To determine cardiopulmonary effects of ticagrelor by objective testing • To measure the pharmacokinetics of ticagrelor therapy

Methods Primary end point: Onset: IPA (20 M ADP, final extent) at 2 hours after the first dose of study drug Offset: Slope of IPA (20 M ADP, final extent) between 4 and 72 hours after the last dose of study drug

Methods Inclusion Criteria: - 18 years of age with documented stable CAD on aspirin therapy (75 to 100 mg/d) Exclusion Criteria: - ACS within 12 mo. - Indication for antithrombotic therapy (eg, warfarin, clopidogrel, or aspirin dose other than 75 to 100 mg/d during the study period) - CHF - EF <35% - FEV 1 - FVC < normal - Bleeding diathesis or severe pulmonary disease - Pregnancy - Current smoking - Moderate or strong cytochrome P 450 3 A inhibitors, substrates, or inducers - Platelet count <100 000/mm 3; Hb<10 g/d. L; Hb. A 1 c >10% - Drug addiction or alcohol abuse - NSAID - Creatinine clearance <30 m. L/min.
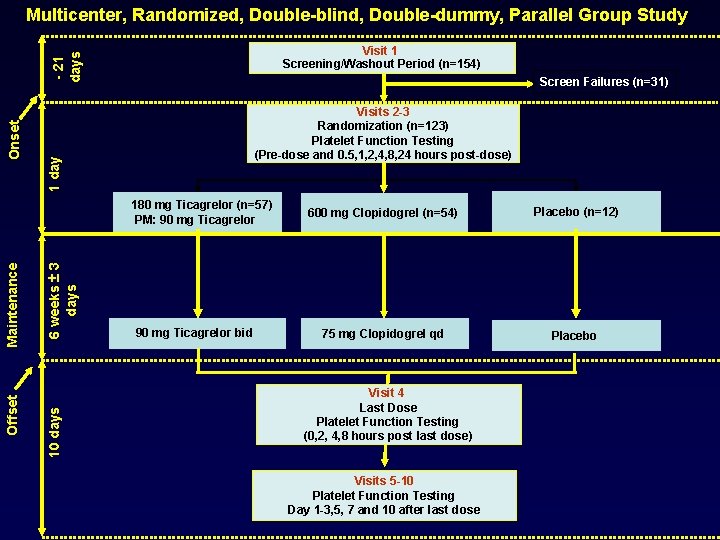
Multicenter, Randomized, Double-blind, Double-dummy, Parallel Group Study Screen Failures (n=31) Visits 2 -3 Randomization (n=123) Platelet Function Testing (Pre-dose and 0. 5, 1, 2, 4, 8, 24 hours post-dose) 1 day Onset - 21 days Visit 1 Screening/Washout Period (n=154) 6 weeks ± 3 days 10 days Offset Maintenance 180 mg Ticagrelor (n=57) PM: 90 mg Ticagrelor bid 600 mg Clopidogrel (n=54) Placebo (n=12) 75 mg Clopidogrel qd Placebo Visit 4 Last Dose Platelet Function Testing (0, 2, 4, 8 hours post last dose) Visits 5 -10 Platelet Function Testing Day 1 -3, 5, 7 and 10 after last dose

Methods Platelet Function Measurements Light Transmittance Aggregometry (Chronolog) * - 5 and 20 M ADP-, and 2 ug/ml collagen, 2 m. M arachidonic acid-induced - Maximum and final extent (6 minutes) * Verify. Now P 2 Y 12 Assay - Platelet reaction units (PRU) and % inhibition (Accumetrics) Vasodilator-stimulated Phosphoprotein (VASP) Phosphorylation * - Platelet reactivity index (PRI, Biocytex) ADP-induced GPIIb/IIIa and P-selectin Expression by Flow Cytometry

Statistical Methods • ITT analysis • Primary comparison between ticagrelor and clopidogrel by Wilcoxon rank sum test • Slope of onset and offset by random coefficients model fitted to: - IPA at 0. 5, 1, and 2 h post-loading (onset) - IPA at 4, 8, 24, 48, and 72 hours after last dose (offset) • Model included fixed effects for: treatment group, hour (relative to last dose or first dose), treatment group by hour interaction, center-by-treatment interaction, and random coefficients for the patient and patient by hour interaction • Statistical analyses carried out by QDS (King of Prussia, PA) using SAS™ (version 8. 2).
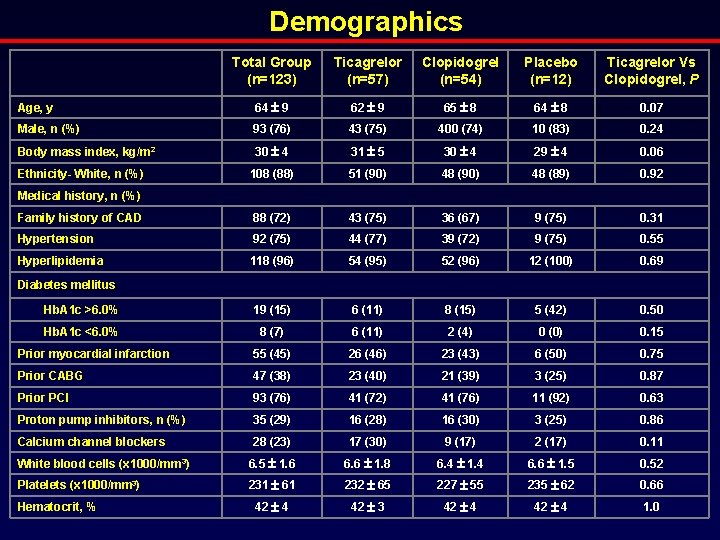
Demographics Total Group (n=123) Ticagrelor (n=57) Clopidogrel (n=54) Placebo (n=12) Ticagrelor Vs Clopidogrel, P Age, y 64 9 62 9 65 8 64 8 0. 07 Male, n (%) 93 (76) 43 (75) 400 (74) 10 (83) 0. 24 Body mass index, kg/m 2 30 4 31 5 30 4 29 4 0. 06 108 (88) 51 (90) 48 (89) 0. 92 Family history of CAD 88 (72) 43 (75) 36 (67) 9 (75) 0. 31 Hypertension 92 (75) 44 (77) 39 (72) 9 (75) 0. 55 Hyperlipidemia 118 (96) 54 (95) 52 (96) 12 (100) 0. 69 Hb. A 1 c >6. 0% 19 (15) 6 (11) 8 (15) 5 (42) 0. 50 Hb. A 1 c <6. 0% 8 (7) 6 (11) 2 (4) 0 (0) 0. 15 Prior myocardial infarction 55 (45) 26 (46) 23 (43) 6 (50) 0. 75 Prior CABG 47 (38) 23 (40) 21 (39) 3 (25) 0. 87 Prior PCI 93 (76) 41 (72) 41 (76) 11 (92) 0. 63 Proton pump inhibitors, n (%) 35 (29) 16 (28) 16 (30) 3 (25) 0. 86 Calcium channel blockers 28 (23) 17 (30) 9 (17) 2 (17) 0. 11 White blood cells (x 1000/mm 3) 6. 5 1. 6 6. 6 1. 8 6. 4 1. 4 6. 6 1. 5 0. 52 Platelets (x 1000/mm 3) 231 61 232 65 227 55 235 62 0. 66 42 4 42 3 42 4 1. 0 Ethnicity- White, n (%) Medical history, n (%) Diabetes mellitus Hematocrit, %
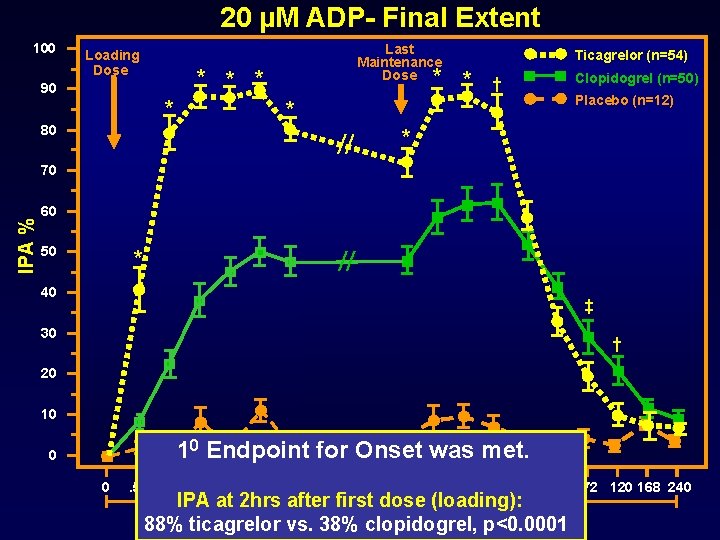
20 µM ADP- Final Extent 100 Loading Dose * 90 * Last Maintenance Dose * * * Ticagrelor (n=54) * Clopidogrel (n=50) † Placebo (n=12) * 80 * IPA % 70 60 50 * 40 ‡ 30 † 20 10 Onset was met. 10 Endpoint for 0 0 . 5 1 2 4 8 24 6 weeks 0 2 4 8 24 48 IPA at 2 hrs after first dose (loading): Onset Maintenance Offset 88% ticagrelor vs. 38% Time clopidogrel, p<0. 0001 (hours) 72 120 168 240
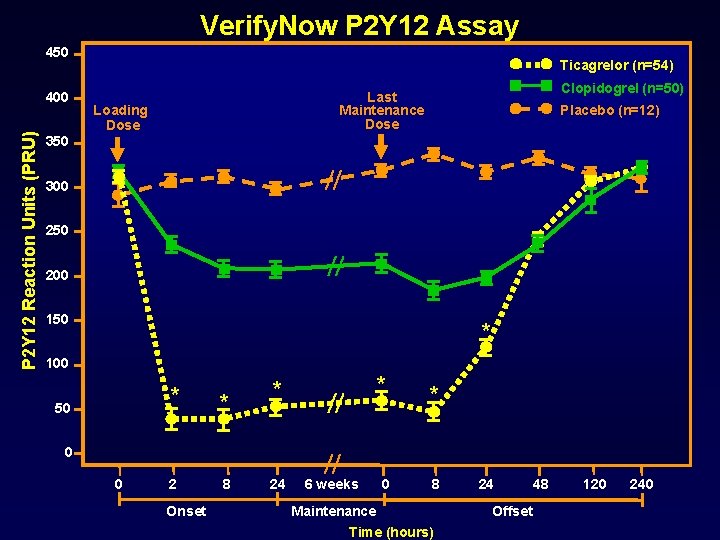
Verify. Now P 2 Y 12 Assay 450 P 2 Y 12 Reaction Units (PRU) 400 Ticagrelor (n=54) Clopidogrel (n=50) Last Maintenance Dose Loading Dose Placebo (n=12) 350 300 250 200 150 * 100 * 50 * * 0 0 2 Onset 8 24 6 weeks * 0 * 8 Maintenance Time (hours) 24 Offset 48 120 240
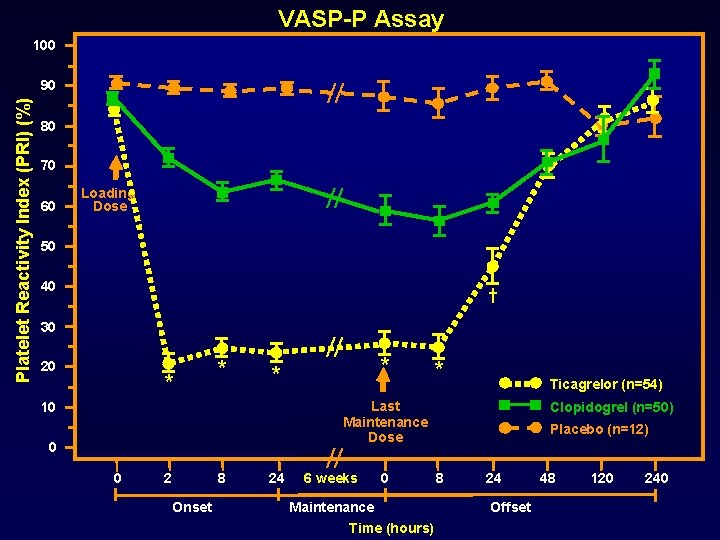
VASP-P Assay 100 Platelet Reactivity Index (PRI) (%) 90 80 70 60 Loading Dose 50 40 † 30 20 * * 10 0 0 2 8 Onset * * 24 * Ticagrelor (n=54) Last Maintenance Dose 6 weeks 0 Maintenance Time (hours) Clopidogrel (n=50) Placebo (n=12) 8 24 Offset 48 120 240
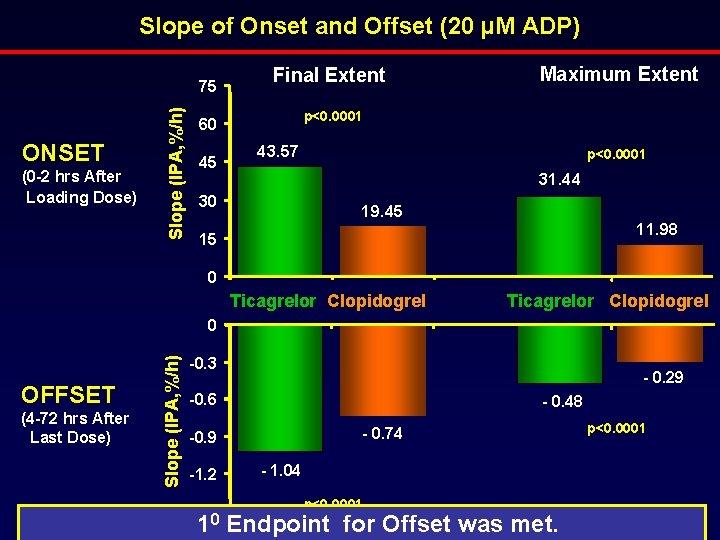
Slope of Onset and Offset (20 µM ADP) ONSET (0 -2 hrs After Loading Dose) Slope (IPA, %/h) 75 Final Extent p<0. 0001 60 45 Maximum Extent 43. 57 p<0. 0001 31. 44 30 19. 45 11. 98 15 0 Ticagrelor Clopidogrel OFFSET (4 -72 hrs After Last Dose) Slope (IPA, %/h) 0 -0. 3 - 0. 29 -0. 6 - 0. 48 - 0. 74 -0. 9 -1. 2 -1. 50 1 - 1. 04 p<0. 0001 Endpoint for Offset was met. p<0. 0001
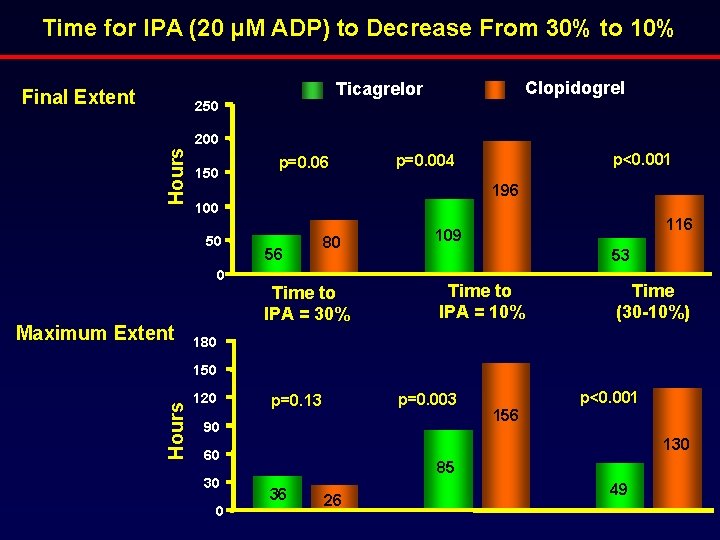
Time for IPA (20 µM ADP) to Decrease From 30% to 10% Final Extent Clopidogrel Ticagrelor 250 Hours 200 150 p<0. 001 p=0. 004 196 100 50 0 Maximum Extent p=0. 06 56 80 Time to IPA = 30% 116 109 53 Time to IPA = 10% Time (30 -10%) 180 Hours 150 120 p=0. 003 p=0. 13 90 130 60 30 0 156 p<0. 001 85 36 26 49

Conclusions • First study to comprehensively characterize onset and offset of the antiplatelet effect of ticagrelor compared with clopidogrel in stable CAD patients. • 3 Major Findings: - Ticagrelor onset is very rapid and markedly greater than high loading dose clopidogrel - Greater inhibitory effect of ticagrelor is sustained during maintenance - Ticagrelor offset as determined by IPA slope was significantly faster than clopidogrel • These effects may explain the lower occurrence of the primary endpoint with ticagrelor therapy as compared to clopidogrel therapy in PLATO whereas numerically less CABG-related bleeding occurred in the ticagrelor group despite greater platelet inhibition.

Investigators and Centers Paul A Gurbel, Kevin P Bliden, Udaya S Tantry, Tania Gesheff, Mark J Antonino Sinai Center for Thrombosis Research, Baltimore, Maryland Kathleen Butler, Cheryl Wei, Renly Teng - Astra. Zeneca LP, Wilmington, Delaware Rob F Storey, Shanker B Patil, Arun Karunakaran - University of Sheffield, United Kingdom Dean J Kereiakes - The Christ Hospital Heart and Vascular Center and The Lindner Research Center, Cincinnati, Ohio Cordel Parris - Dolby Research LLC, Baton Rouge, Louisiana Drew Purdy - Black Hills Clinical Research Center, Rapid City, South Dakota Vance Wilson - Cardiology Consultants, Daytona Beach, Florida Gary Ledley - Drexel University College of Medicine, Philadelphia, Pennsylvania

- Slides: 18