Randomized Comparison of HighDos Oral Vitamins versus Placebo

Randomized Comparison of High-Dos Oral Vitamins versus Placebo in the Tr to. Assess Chelation Therapy (TACT) Gervasio A. Lamas, MD, FACC Professor of Clinical Medicine Columbia University Division of Cardiology Mount Sinai Medical Center Miami Beach, FL For the TACT Investigators

Study Organization • Funding agencies: NCCAM and NHLBI • Clinical Coordinating Center: Mount Sinai Medical Center, Miami Beach FL • Data Coordinating Center and EQOL Coordinating Center: Duke Clinical Research Institute • Clinical Events Committee: Brigham and Women’s Hospital • Central Pharmacy: Universal Arts, Miami FL • Vitamins: Douglas Labs, Pittsburgh PA

Background TACT tested whether EDTA chelation reduced a composite cardiovascular endpoint in post MI patients. Chelation practitioners also use high doses of antioxidant vitamins and minerals in conjunction with intravenous chelation. Oral vitamins and minerals therefore constituted a potential confounder.

Design Rationale • To clarify the contributions of high dose vitamins vs. IV chelation, TACT was designed as a 2 x 2 factorial trial, with patients randomized to 4 groups: 1. 2. 3. 4. Active oral vitamins + active IV chelation infusions Placebo oral vitamins + active IV chelation infusions Active oral vitamins + placebo IV chelation infusions Placebo oral vitamins + placebo IV chelation infusions All patients also received a lower-dose daily vitamin regimen during the infusion phase to prevent chelation-related deficiencies (vitamin B 6, zinc, copper, manganese and chromium)

TACT: High Dose Oral Treatment 3 caplets twice a day for the duration of the study. Vitamin A Vitamin C Vitamin D 3 Vitamin E Vitamin K Thiamin Niacin Vitamin. B 6 Folate Vitamin B 12 Biotin Panthothenic Acid Calcium Iodine Magnesium Zinc Selenium Copper Manganese Chromium Molybdenum Potassium Choline Inositol PABA Vanadium Citrus Flavonoids Double-blind active or placebo high dose vitamins were shipped from a central pharmacy to sites. Lamas GA, Goertz C, Boineau R, et. al. Design of the Trial to Assess Chelation Therapy (TACT). Am Heart J. 2012 Jan; 163(1): 7 -12.
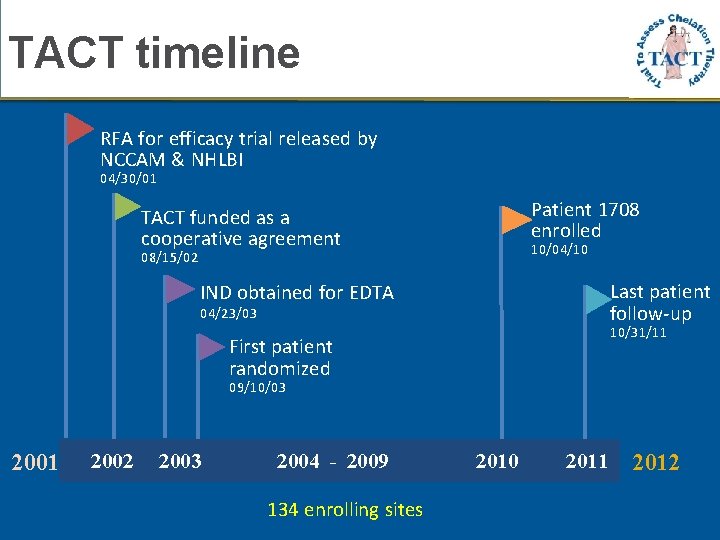
TACT timeline RFA for efficacy trial released by NCCAM & NHLBI 04/30/01 Patient 1708 enrolled TACT funded as a cooperative agreement 10/04/10 08/15/02 Last patient follow-up IND obtained for EDTA 04/23/03 10/31/11 First patient randomized 09/10/03 2001 2002 2003 2004 - 2009 134 enrolling sites 2010 2011 2012

Eligibility Age 50 or older MI > 6 months prior Creatinine <2. 0 mg/d. L No coronary or carotid revascularization within 6 months No active heart failure or heart failure hospitalization within 6 months Able to tolerate 500 cc infusions weekly No cigarette smoking within 3 months Signed informed consent

Primary Endpoint Primary composite endpoint: time to first occurrence of either death, MI, stroke, coronary revascularization, or hospitalization for angina

Sample Size Original plan was to randomize 2372 patients and follow up a minimum of 1 year - 85% power for detecting a 25% difference. In 2009, due to slow enrollment, blinded investigators asked for a reduction of total sample size to 1700, with a compensatory increase in followup to maintain same unconditional power. DSMB approved the request.

Data. Analysis Treatment comparisons as randomized (intent to treat) Two sided statistical testing Log-rank test using time to first event Prespecified analysis of factorial groups Interim monitoring using Lan-De. Mets alphaspending function with O’Brien-Fleming-type monitoring boundaries Because of multiple DSMB reviews due to length of study, the final level of significance was 0. 036
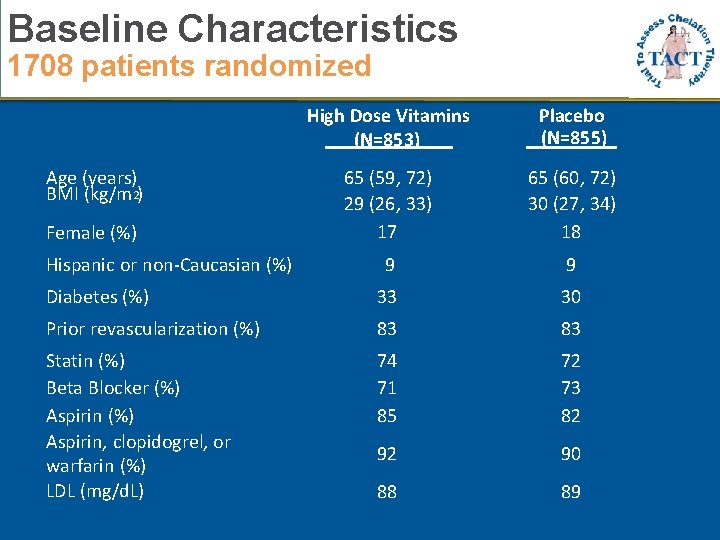
Baseline Characteristics 1708 patients randomized High Dose Vitamins (N=853) Placebo (N=855) 65 (59, 72) 29 (26, 33) 17 65 (60, 72) 30 (27, 34) 18 9 9 Diabetes (%) 33 30 Prior revascularization (%) 83 83 Statin (%) Beta Blocker (%) Aspirin, clopidogrel, or warfarin (%) LDL (mg/d. L) 74 71 85 72 73 82 92 90 88 89 Age (years) BMI (kg/m 2) Female (%) Hispanic or non-Caucasian (%)
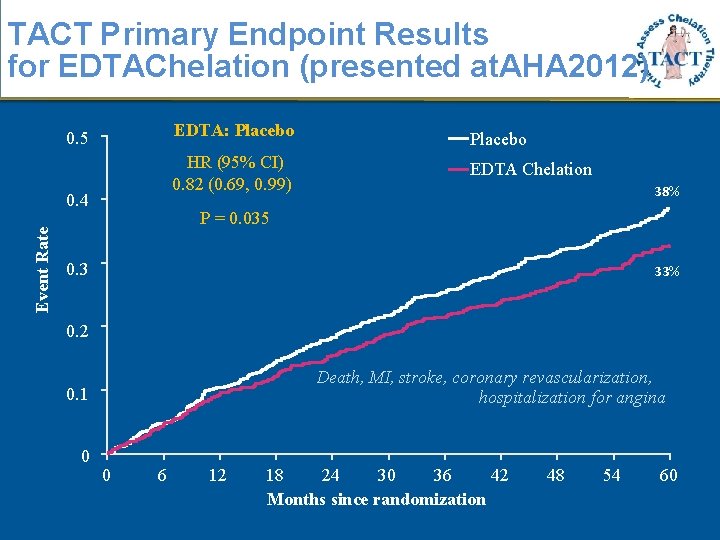
TACT Primary Endpoint Results for EDTAChelation (presented at. AHA 2012) 0. 5 Event Rate 0. 4 EDTA: Placebo HR (95% CI) 0. 82 (0. 69, 0. 99) EDTA Chelation 38% P = 0. 035 0. 3 33% 0. 2 Death, MI, stroke, coronary revascularization, hospitalization for angina 0. 1 0 0 6 12 18 24 30 36 42 Months since randomization 48 54 60
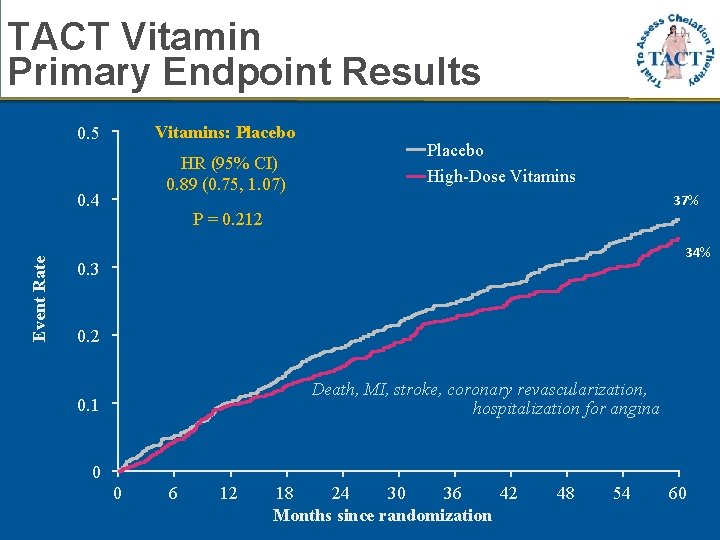
TACT Vitamin Primary Endpoint Results Vitamins: Placebo 0. 5 HR (95% CI) 0. 89 (0. 75, 1. 07) Event Rate 0. 4 Placebo High-Dose Vitamins 37% P = 0. 212 34% 0. 3 0. 2 Death, MI, stroke, coronary revascularization, hospitalization for angina 0. 1 0 0 6 12 18 24 30 36 42 Months since randomization 48 54 60
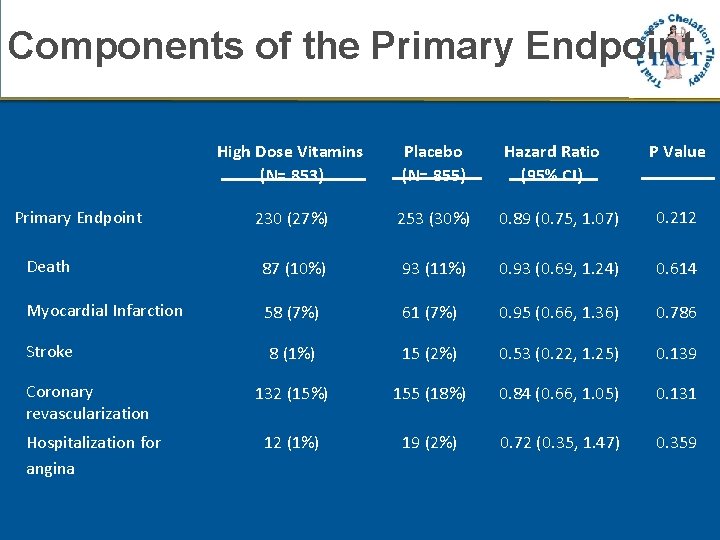
Components of the Primary Endpoint High Dose Vitamins (N= 853) Placebo (N= 855) Hazard Ratio (95% CI) 230 (27%) 253 (30%) 0. 89 (0. 75, 1. 07) 0. 212 Death 87 (10%) 93 (11%) 0. 93 (0. 69, 1. 24) 0. 614 Myocardial Infarction 58 (7%) 61 (7%) 0. 95 (0. 66, 1. 36) 0. 786 Stroke 8 (1%) 15 (2%) 0. 53 (0. 22, 1. 25) 0. 139 132 (15%) 155 (18%) 0. 84 (0. 66, 1. 05) 0. 131 12 (1%) 19 (2%) 0. 72 (0. 35, 1. 47) 0. 359 Primary Endpoint Coronary revascularization Hospitalization for angina P Value
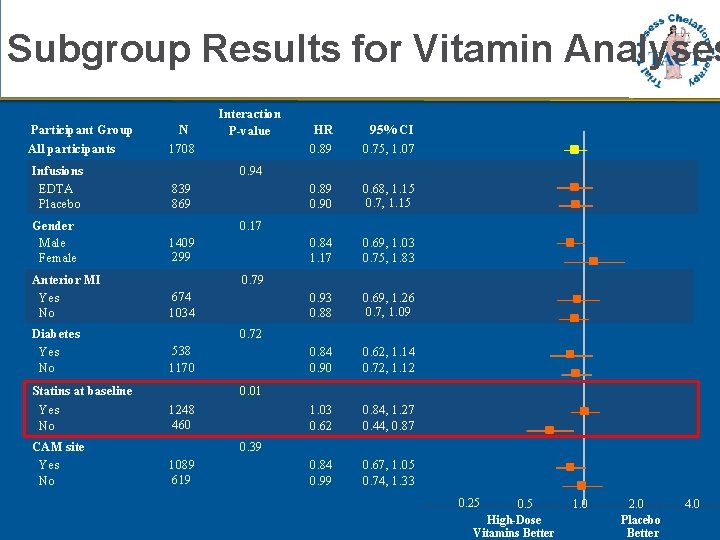
Subgroup Results for Vitamin Analyses Participant Group All participants Infusions EDTA Placebo Gender Male Female Anterior MI Yes No Diabetes Yes No Statins at baseline Yes No CAM site Yes No N 1708 Interaction P-value HR 0. 89 95% CI 0. 75, 1. 07 0. 89 0. 90 0. 68, 1. 15 0. 7, 1. 15 0. 84 1. 17 0. 69, 1. 03 0. 75, 1. 83 0. 93 0. 88 0. 69, 1. 26 0. 7, 1. 09 0. 84 0. 90 0. 62, 1. 14 0. 72, 1. 12 1. 03 0. 62 0. 84, 1. 27 0. 44, 0. 87 0. 84 0. 99 0. 67, 1. 05 0. 74, 1. 33 0. 94 839 869 0. 17 1409 299 0. 79 674 1034 0. 72 538 1170 0. 01 1248 460 0. 39 1089 619 0. 25 0. 5 High-Dose Vitamins Better 1. 0 2. 0 Placebo Better 4. 0
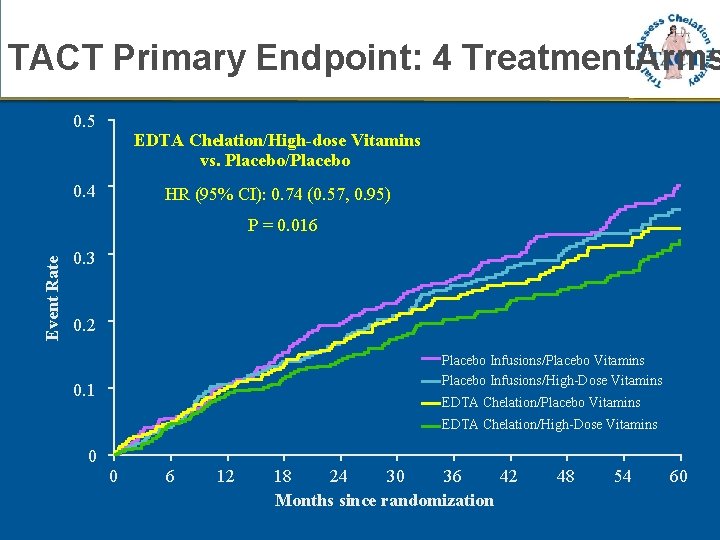
TACT Primary Endpoint: 4 Treatment. Arms 0. 5 EDTA Chelation/High-dose Vitamins vs. Placebo/Placebo 0. 4 HR (95% CI): 0. 74 (0. 57, 0. 95) Event Rate P = 0. 016 0. 3 0. 2 Placebo Infusions/Placebo Vitamins Placebo Infusions/High-Dose Vitamins 0. 1 EDTA Chelation/Placebo Vitamins EDTA Chelation/High-Dose Vitamins 0 0 6 12 18 24 30 36 42 Months since randomization 48 54 60

Adherence • 847 patients (50%) discontinued the high-dose vitamins/placebo; 289 (17%) of which also withdrew consent – The most common reason for discontinuation was patient refusal (75%) – Median duration of treatment: 33. 4 months. – Median duration of treatment among those who prematurely discontinued vitamins: 13. 8 months. – No difference in vitamin discontinuation rate in active vs placebo.

Limitations • Limited power to assess individual components of the composite endpoint. • There was a high vitamin noncompliance rate, limiting interpretation of the vitamin results, although this may introduce a conservative bias. • High consent withdrawal rate limited overall follow-up time. • Hypothesis of proposed benefit of high dose vitamin therapy is unclear, which limits interpretation.

Summary High dose oral vitamins reduced the composite outcome by 11%, which was not statistically significant. When combined with EDTA chelation the benefit was additive and magnitude was statistically significant.

Caveats • The results of this study do not support the use of highdose vitamin and mineral therapy as an adjunct to optimal evidence-based medical therapy in patients with prior myocardial infarction. • These findings should stimulate further research, but are not, by themselves, sufficient to recommend the routine use of chelation therapy and high-dose vitamins in post. MI patients.

Thank you
- Slides: 21