Randomised comparison of bioabsorbable everolimuseluting and biolimuseluting coronary

Randomised comparison of bioabsorbable everolimus-eluting and biolimus-eluting coronary stents in routine clinical care patients with ischemic heart disease Michael Maeng, Evald H. Christiansen, Bent Raungaard, Christian J. Terkelsen, Steen D. Kristensen, Steen Carstensen, Jens Aarøe, Svend Eggert Jensen, Anton B. Villadsen, Jan Ravkilde, Leif Thuesen, Jens F. Lassen, Troels Thim, Ashkan Eftekhari, Steen Carstensen, Karsten T. Veien, Knud N. Hansen, Anders Junker, Hans E. Bøtker, Lisette O. Jensen, On behalf of the SORT OUT VIII investigators at Aarhus University Hospital, Aalborg University Hospital, and Odense University Hospital, Denmark

Purpose • To compare the 1 -year clinical outcomes in routine clinical care patients (“all-comers”) randomised to bioabsorbable-polymer platinum-chromium everolimus-eluting (Synergy, EES) or bioabsorbablepolymer stainless-steel biolimus-eluting (Bio. Matrix Neo. Flex, BES) coronary stent implantation

Methods • 2800 patients randomised to thin-strut bioabsorbable-polymer everolimus-eluting or bioabsorbable-polymer biolimus-eluting coronary stent implantation • Inclusion criteria: – Indication for drug-eluting stent implantation • Exclusion criteria: – Lack of consent, <18 years old, not speaking the national language, participation in other randomized trials, expected survival <1 year

Endpoints • Primary: – Device-related target lesion failure (TLF) hierarchically as cardiac death, myocardial infarction (MI) related to the target lesion, or target lesion revascularisation (TLR) within 12 months • Secondary: – TLR, target vessel revascularization (TVR), stent thrombosis, cardiac death, all-cause mortality, device delivery failure, any MI, patient-related composite endpoint (any MI/death/revascularisation)

Power calculation • Assumptions: – – 5% risk of type 1 error 90% power Noninferiority margin 3% TLF 5% at 1 -year in the BES group • Group size at least 1302 patients
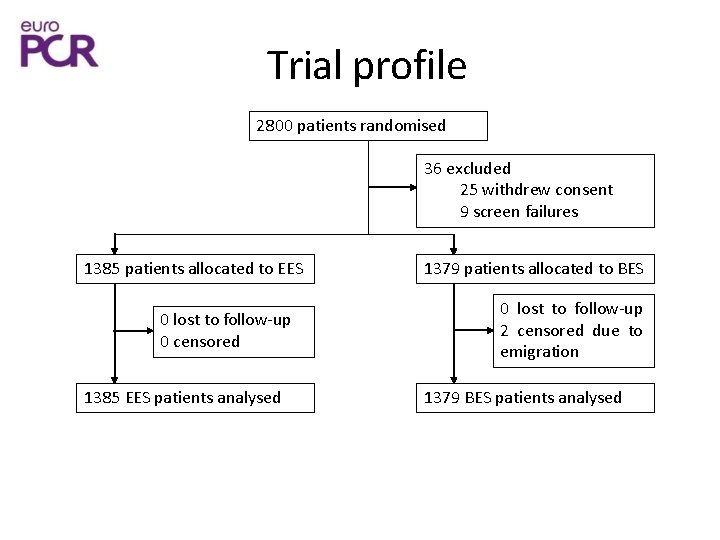
Trial profile 2800 patients randomised 36 excluded 25 withdrew consent 9 screen failures 1385 patients allocated to EES 0 lost to follow-up 0 censored 1385 EES patients analysed 1379 patients allocated to BES 0 lost to follow-up 2 censored due to emigration 1379 BES patients analysed
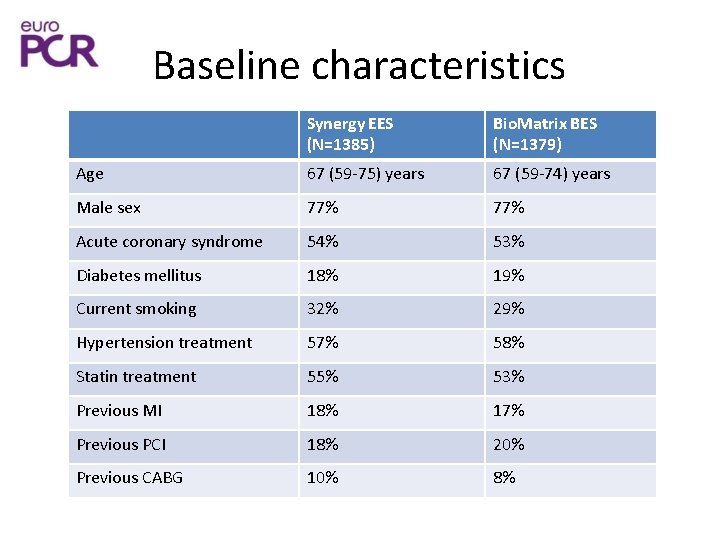
Baseline characteristics Synergy EES (N=1385) Bio. Matrix BES (N=1379) Age 67 (59 -75) years 67 (59 -74) years Male sex 77% Acute coronary syndrome 54% 53% Diabetes mellitus 18% 19% Current smoking 32% 29% Hypertension treatment 57% 58% Statin treatment 55% 53% Previous MI 18% 17% Previous PCI 18% 20% Previous CABG 10% 8%
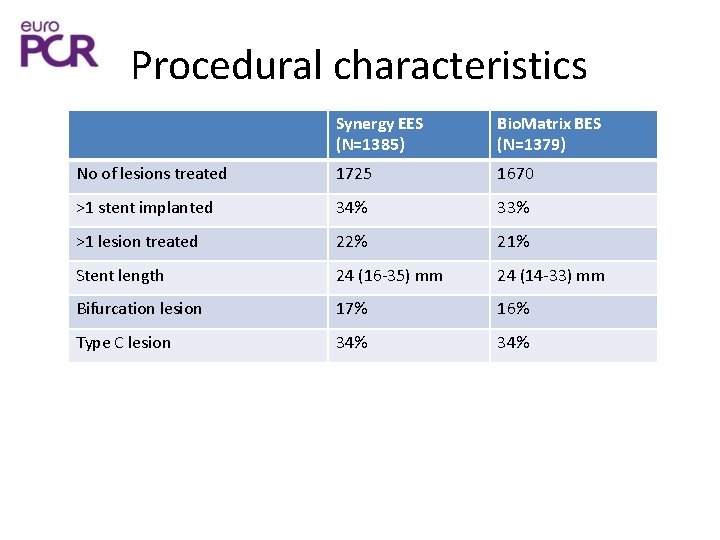
Procedural characteristics Synergy EES (N=1385) Bio. Matrix BES (N=1379) No of lesions treated 1725 1670 >1 stent implanted 34% 33% >1 lesion treated 22% 21% Stent length 24 (16 -35) mm 24 (14 -33) mm Bifurcation lesion 17% 16% Type C lesion 34%
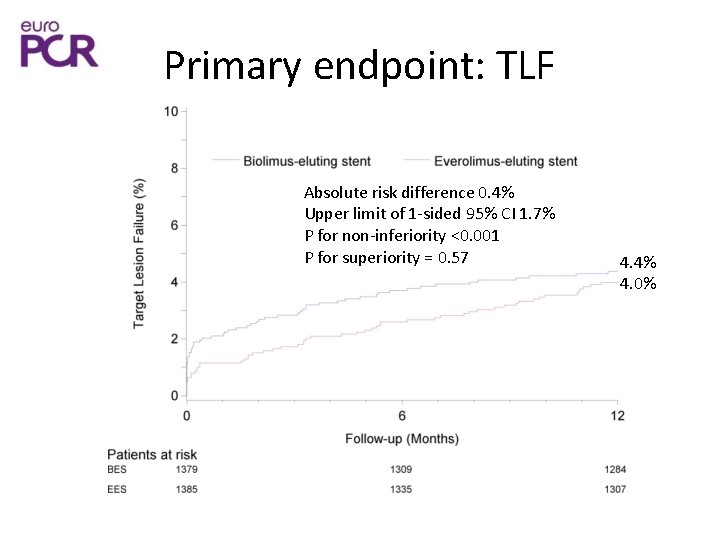
Primary endpoint: TLF Absolute risk difference 0. 4% Upper limit of 1 -sided 95% CI 1. 7% P for non-inferiority <0. 001 P for superiority = 0. 57 4. 4% 4. 0%
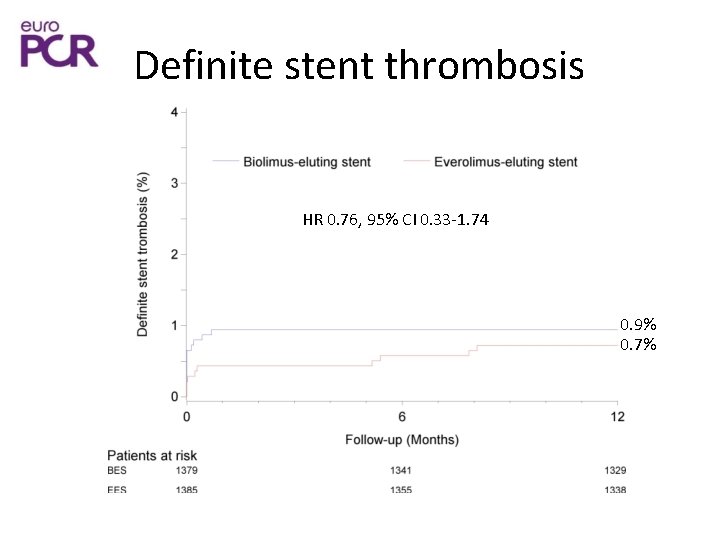
Definite stent thrombosis HR 0. 76, 95% CI 0. 33 -1. 74 0. 9% 0. 7%
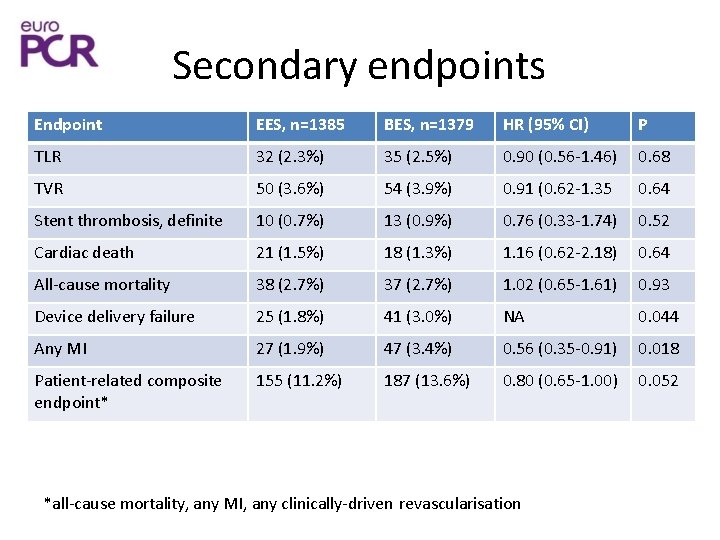
Secondary endpoints Endpoint EES, n=1385 BES, n=1379 HR (95% CI) P TLR 32 (2. 3%) 35 (2. 5%) 0. 90 (0. 56 -1. 46) 0. 68 TVR 50 (3. 6%) 54 (3. 9%) 0. 91 (0. 62 -1. 35 0. 64 Stent thrombosis, definite 10 (0. 7%) 13 (0. 9%) 0. 76 (0. 33 -1. 74) 0. 52 Cardiac death 21 (1. 5%) 18 (1. 3%) 1. 16 (0. 62 -2. 18) 0. 64 All-cause mortality 38 (2. 7%) 37 (2. 7%) 1. 02 (0. 65 -1. 61) 0. 93 Device delivery failure 25 (1. 8%) 41 (3. 0%) NA 0. 044 Any MI 27 (1. 9%) 47 (3. 4%) 0. 56 (0. 35 -0. 91) 0. 018 Patient-related composite endpoint* 155 (11. 2%) 187 (13. 6%) 0. 80 (0. 65 -1. 00) 0. 052 *all-cause mortality, any MI, any clinically-driven revascularisation

Conclusions • At 1 -year follow-up, the thin-strut everolimuseluting Synergy stent was found to be noninferior to the biolimus-eluting Bio. Matrix Neo. Flex stent in treating routine clinical care patients • Successful implantation of the allocated stent was more frequent in the thin-strut Synergy group, which may be associated with the lower rate of MI and the patient-related composite endpoint
- Slides: 12