Raltegravir AbacavirLamivudine SHIELD Trial Raltegravir AbacavirLamivudine SHIELD Study

Raltegravir + Abacavir-Lamivudine SHIELD Trial

Raltegravir + Abacavir-Lamivudine SHIELD: Study Design: SHIELD • Background: Open-label, prospective, pilot trial evaluating efficacy of raltegravir in combination with abacavir-lamivudine in treatment-naïve persons with HIV. • Inclusion Criteria (n = 37) - Age ≥ 18 years - Antiretroviral therapy naïve - HIV RNA >1000 copies/m. L - HLA-B*5701 negative - No resistance to any study drug • Treatment Arm - Raltegravir 400 mg BID + Abacavir-Lamivudine QD Source: Young B, et al. HIV Clin Trials. 2010; 11: 260 -9. Raltegravir 400 mg BID + Abacavir-Lamivudine QD (n = 37)
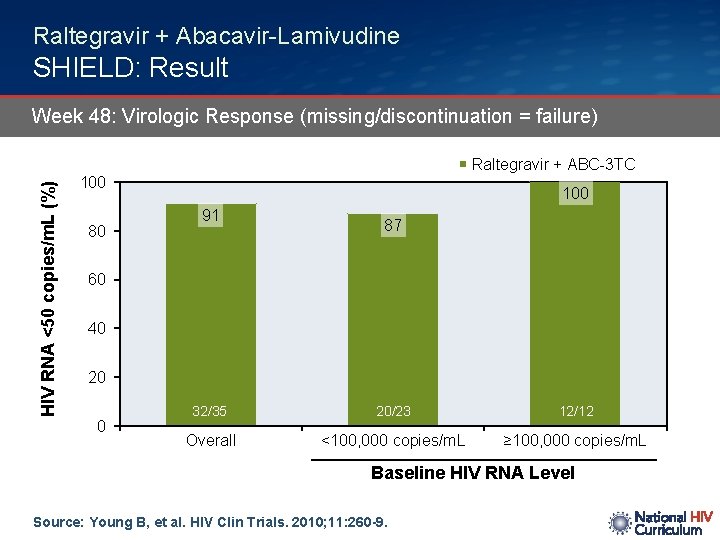
Raltegravir + Abacavir-Lamivudine SHIELD: Result Week 48: Virologic Response (missing/discontinuation = failure) HIV RNA <50 copies/m. L (%) Raltegravir + ABC-3 TC 100 80 100 91 87 60 40 20 0 32/35 20/23 12/12 Overall <100, 000 copies/m. L ≥ 100, 000 copies/m. L Baseline HIV RNA Level Source: Young B, et al. HIV Clin Trials. 2010; 11: 260 -9.
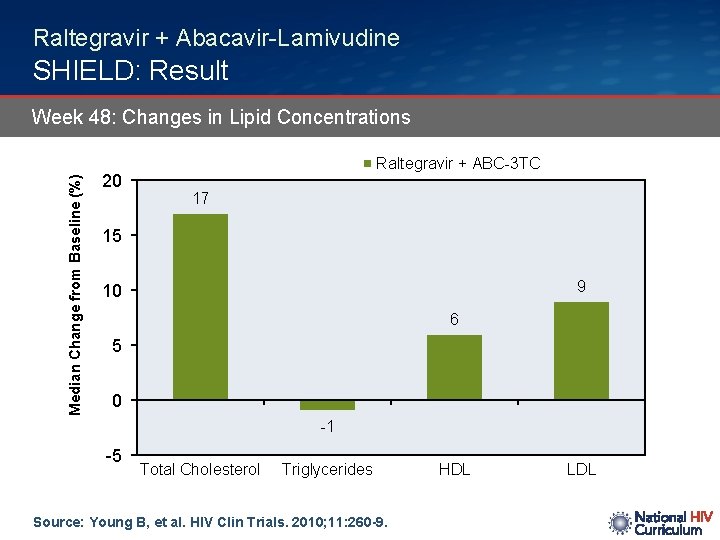
Raltegravir + Abacavir-Lamivudine SHIELD: Result Median Change from Baseline (%) Week 48: Changes in Lipid Concentrations 20 Raltegravir + ABC-3 TC 17 15 9 10 6 5 0 -1 -5 Total Cholesterol Triglycerides Source: Young B, et al. HIV Clin Trials. 2010; 11: 260 -9. HDL LDL

Raltegravir + Abacavir-Lamivudine SHIELD: Conclusions: “In this pilot study, abacavir/lamivudine plus raltegravir was effective and generally well-tolerated over 48 weeks with modest changes in fasting lipids. ” Source: Young B, et al. HIV Clin Trials. 2010; 11: 260 -9.

Acknowledgment The National HIV Curriculum is an AIDS Education and Training Center (AETC) Program supported by the Health Resources and Services Administration (HRSA) of the U. S. Department of Health and Human Services (HHS) as part of an award totaling $800, 000 with 0% financed with non-governmental sources. This project is led by the University of Washington’s Infectious Diseases Education and Assessment (IDEA) Program. The content in this presentation are those of the author(s) and do not necessarily represent the official views of, nor an endorsement, by HRSA, HHS, or the U. S. Government.
- Slides: 6