Radiotherapy in Pediatric Hodgkin Lymphoma TASHA MCDONALD MD





















- Slides: 38

Radiotherapy in Pediatric Hodgkin Lymphoma TASHA MCDONALD, MD DEPARTMENT OF RADIATION MEDICINE JUNE 18, 2008

OVERVIEW Case presentation Risk-groups Early/favorable risk Unfavorable risk Toxicities Future directions

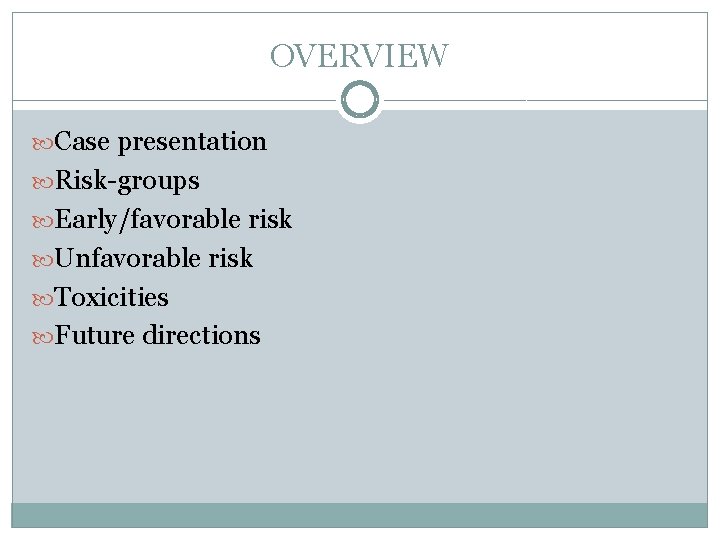
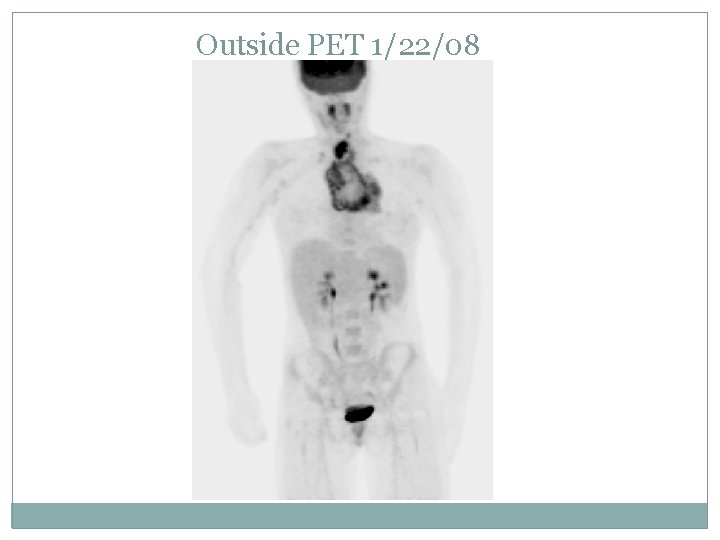
Case presentation L. S. : 18 yo girl presented in 1/08 with 2 months of fatigue, fever, chills, sweats and 10 lb weight loss Developed difficulty swallowing and enlarged neck nodes and SOB when lying down On exam: palpable cervical LAD Underwent US of the neck 1/3/08: irregular 2. 3 x 2. 3 x 3. 4 cm nodule in right neck. LN biopsy on 1/4/08 at Kaiser: nodular sclerosing Hodgkin disease

Outside PET 1/22/08

Outside CT Chest 1/22/08

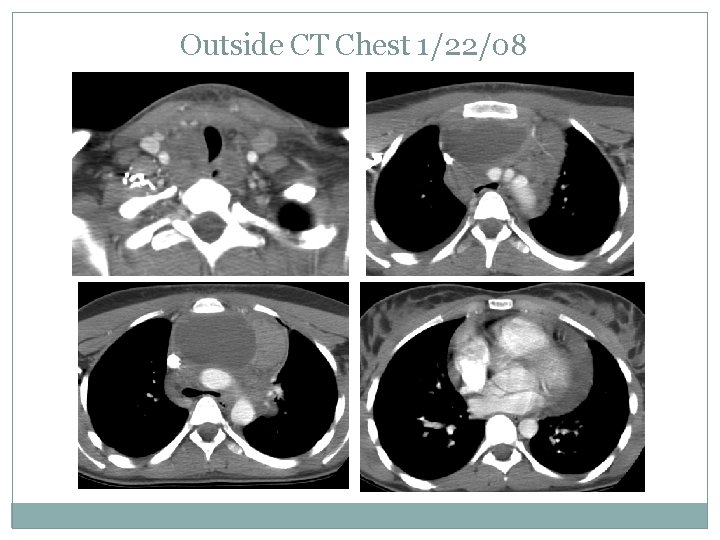
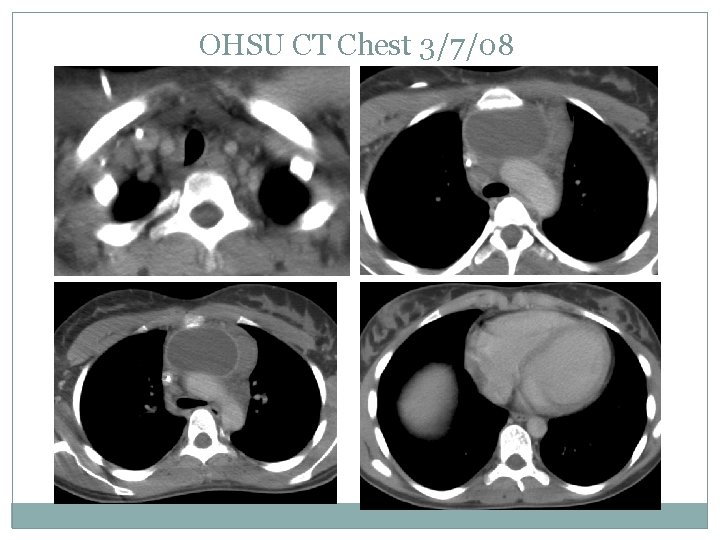
Case presentation Stage IIB NSHD No subdiaphragmatic disease Bone marrow bx was negative Started on COG AHOD 0031 protocol and received 2 cycles of ABVE-PC Re-imaged on 3/7/08 and determined to be a slow early responder protocol

OHSU PET 3/7/08

OHSU CT Chest 3/7/08

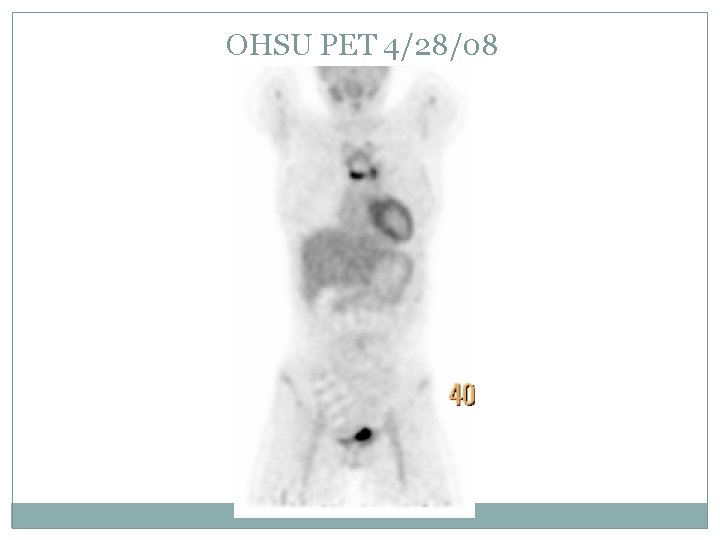
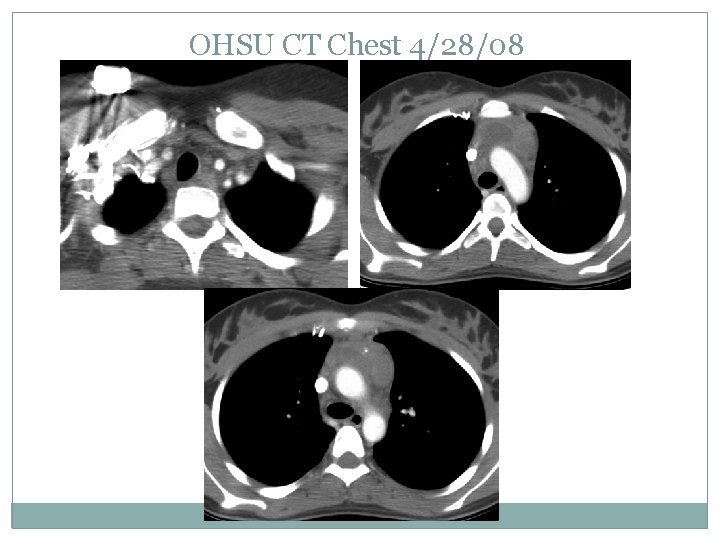
Case presentation Randomized to the augmented therapy arm to receive DECA x 2 followed by 2 more cycles of ABVE -PC CT and PET on 4/28/08 (before the ABVE-PC) showed a 66% reduction tumor size Finished chemotherapy and scheduled to start RT on 5/23/08.

OHSU PET 4/28/08

OHSU CT Chest 4/28/08

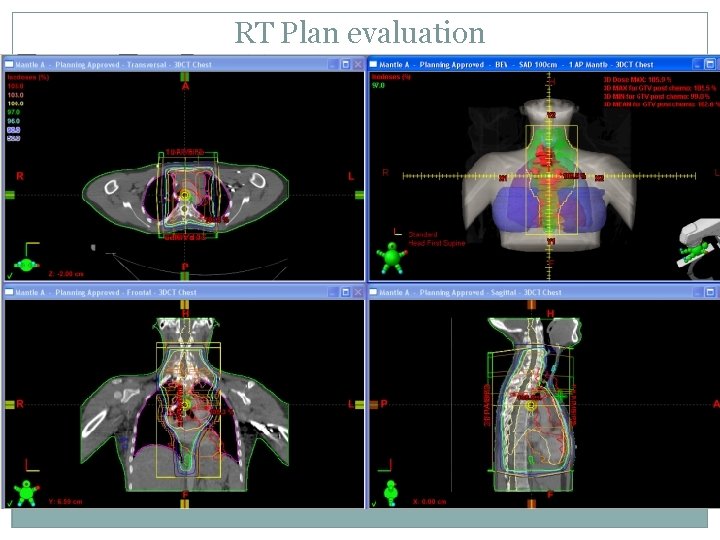
RT Guidelines for AHOD 0031 Protocol IFRT for all pts except those who achieve rapid early response after 2 cycles of chemo AND CR after 4 cycles of chemo. IFRT with 21 Gy in 14 fxs given with AP: PA fields RT to start w/in 4 weeks of last chemo cycle GTV = LN>1. 5 cm; CTV = anatomical compartment of LN; PTV = 1. 0 cm margin to CTV RT fields adapted to response of chemo are not permitted except if treating the mediastinum

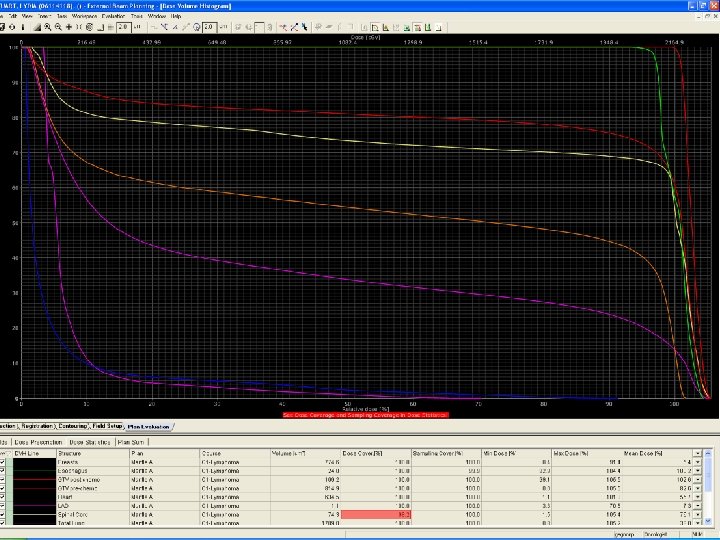
RT Plan evaluation

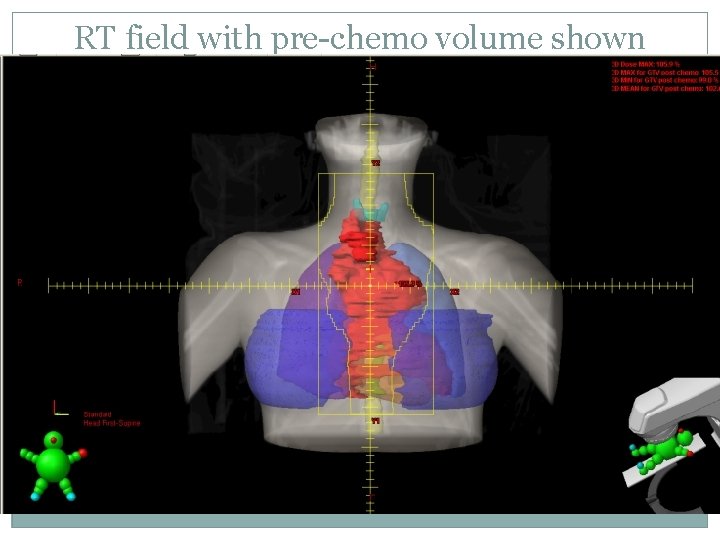
RT field with pre-chemo volume shown

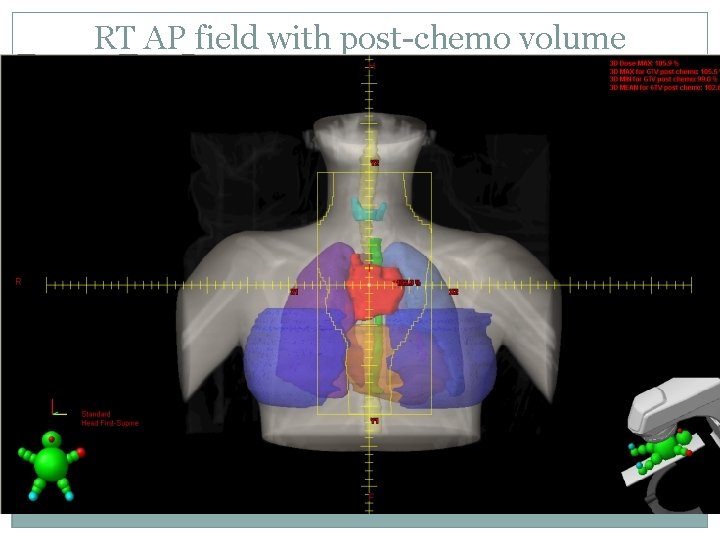
RT AP field with post-chemo volume


History Treated with full-dose (35 -45 Gy) extended-field RT w/ excellent disease control but significant late toxicity Chemotherapy (MOPP or ABVD) was shown to salvage relapsed disease after RT and improve DFS when used as part of initial therapy 1, 2 Low-dose RT (15 -25 Gy) following chemotherapy was shown to produce excellent EFS and OS 3 -5 Chemotherapy followed by low-dose RT became the standard therapy The most recent trials use risk-adapted and/or response-adapted therapy

Risk groups Division into groups based on factors shown to influence outcome Histology Clinical stage B symptoms Bulky disease

Risk groups Prognostic stratification (not uniformly agreed on): Low-risk/favorable: Stage I or II, no B symptoms, no bulky disease and disease in fewer than 3 nodal regions Intermediate-risk: Stage IB, IIB (or bulky disease and extranodal involvement) and sometimes IIIA High-risk: Stage IIIB, IVA/B

Early stage/favorable risk Goal = limit treatment-related toxicity and maintain success of therapy Efficacy of various strategies is relatively equivalent ~90% or better EFS or PFS ~95% OS Treatment: 2 -4 cycles of chemotherapy +/- involved field RT Response-adapted approach: Response to the first cycles of chemotherapy determines inclusion of additional chemo or dose of RT

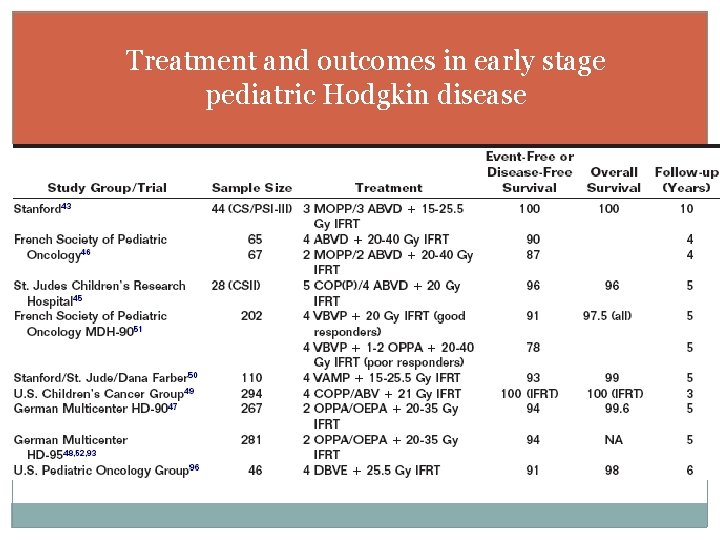
Treatment and outcomes in early stage pediatric Hodgkin disease

Early stage/favorable risk Response-adapted approach SDS group study 6, 7: single arm study; 4 cycles of VAMP followed by IFRT with RT dose determined by response to first 2 cycles PR (53%): 25. 5 Gy IFRT CR (47%): 15 Gy IFRT German HD 958, 9: OPPA or OEPA for two cycles CR (27%): observation PR with >75% reduction (53%): 25 Gy IFRT <75% reduction (~5%): 20 -30 Gy IFRT + 5 Gy boost to >50 ml residual French MDH 9010: 4 cycles of VBVP >70% response (85%): 20 Gy IFRT <70% response: 1 or 2 more cycles of OPPA and 20 or 40 Gy IFRT Despite the differences in treatment, all these studies had a EFS or PFS of 93% or better

Early stage/favorable risk Exclusion of RT POG 862511: Laparotomy-staged IA-IIIA disease 4 cycles of MOPP/ABVD OR 2 cycles of MOPP/ABVD plus 25. 5 Gy IFRT EFS (83% vs 91%) and OS (94% vs 97%) were statistically equivalent CCG 594212: Clinically staged I-II disease 4 cycles of COPP/ABV CRs randomized to observation vs. 21 Gy IFRT Stopped early after interim analysis indicated superiority of RT arm (EFS 85% vs 93%) but OS was 100% in both arms

Intermediate and advanced stage disease More intensified regimens with a combination of diverse chemotherapeutic agents Goal of minimizing treatment-related toxicity is still important but studies that reduced alkylating agents and anthracyclines with limited IFRT lead to decreased EFS 13, 14 RT continues to be standard therapy in this risk group (unless on protocol)

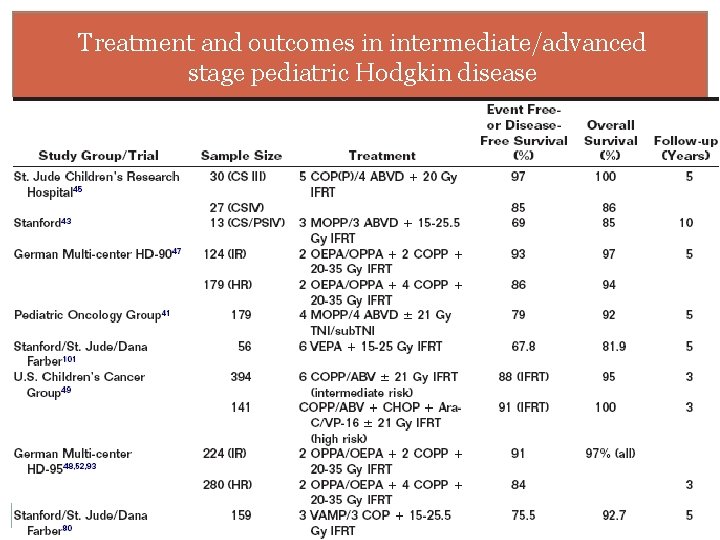
Treatment and outcomes in intermediate/advanced stage pediatric Hodgkin disease

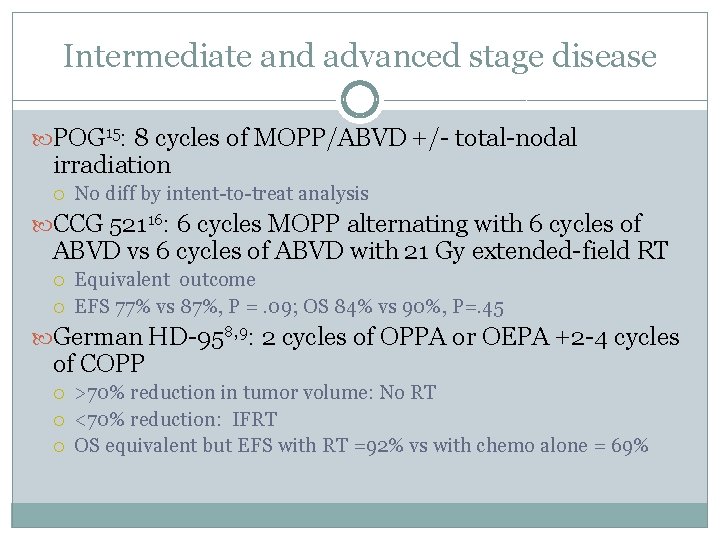
Intermediate and advanced stage disease POG 15: 8 cycles of MOPP/ABVD +/- total-nodal irradiation No diff by intent-to-treat analysis CCG 52116: 6 cycles MOPP alternating with 6 cycles of ABVD vs 6 cycles of ABVD with 21 Gy extended-field RT Equivalent outcome EFS 77% vs 87%, P =. 09; OS 84% vs 90%, P=. 45 German HD-958, 9: 2 cycles of OPPA or OEPA +2 -4 cycles of COPP >70% reduction in tumor volume: No RT <70% reduction: IFRT OS equivalent but EFS with RT =92% vs with chemo alone = 69%

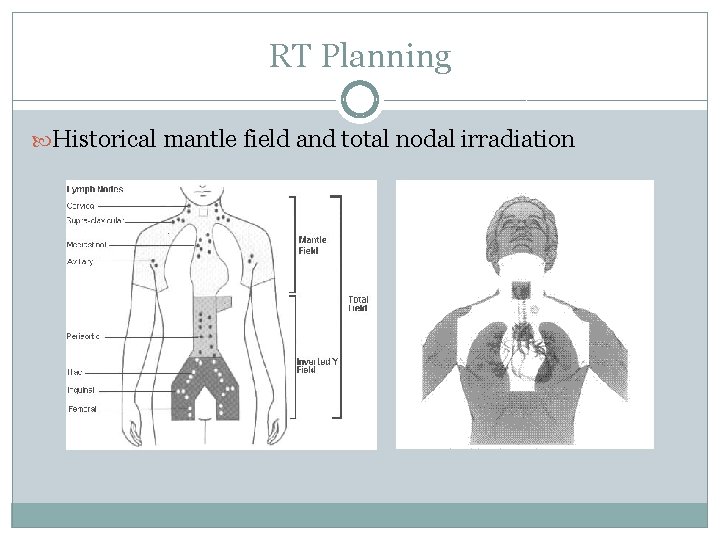
RT Planning Historical mantle field and total nodal irradiation

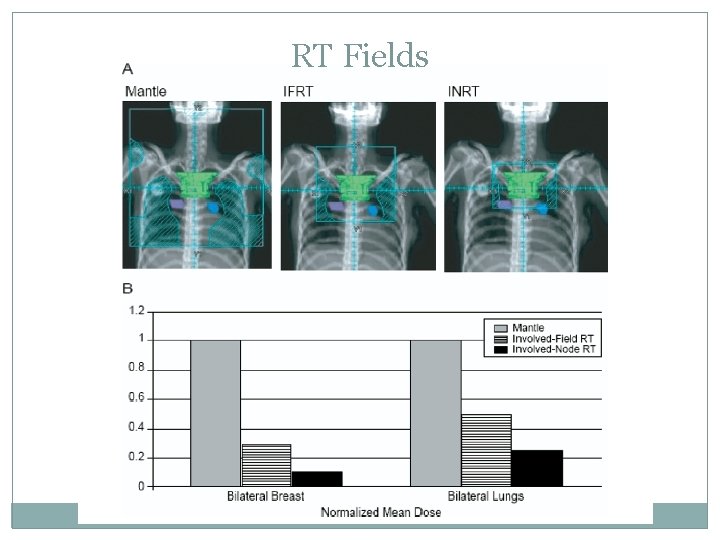
RT Fields

IFRT requires careful evaluation of pre- and post- chemotherapy volumes CTV encompasses post-chemo mediastianal width laterally and pre-chemo extent in sup/inf direction An anterior laryngeal block can be used if it does not shield involved nodes If the axillae are to be treated humeral head blocks are used CT based planning allows evaluation of adequate CTV coverage and normal tissue dose

IFRT Whole-heart irradiation indications: pericardial involvement/invasion Splenic irradiation is indicated in pts with splenic involvement but renal dose must be limited to mean <10. 5 Gy or keep 2/3 rds of the kidney to <15 Gy If the pelvis needs to be treated the ovaries should be relocated and the dose should be limited to <3 Gy When treating a male, ensure on a daily basis that the scrotum is not in the pelvic field

Late Toxicity of Radiotherapy Growth abnormalities 17 Bone and soft-tissue hypoplasia in prepubertal children Thyroid sequela 18, 19 Hypothyroidism Hyperthyroidism Benign and malignant thyroid nodules 17% of children treated with RT dose <26 Gy had thyroid abnormalities compared to 78% with >26 Gy

Late Toxicity Cardiovascular disease 20, 21 Atherosclerotic heart disease Valvular dysfunction Pericardial disease Pulmonary toxicity 22 Decrease in pulmonary function tests Sterility/Infertility: limit dose to ovaries to 3 Gy Increase incidence of secondary cancers 23 -25 Late effects study group: 30 yr cumulative incidence of SC = 26. 3% in pts dx’ed before age 16 Breast cancer was most elevated solid cancer

Late Toxicity Toxicities of higher dose RT are well documented but it is less clear what toxicities will exist with 15 -25 Gy bc many toxicities are dose and volume dependent Second solid cancer risk appears to be dose dependent with patients w/ <23 Gy mediastinal RT with lower risk of developing breast cancer 26

Future Directions Improve the technique of response-adapted therapy Incorporate functional imaging into evaluating treatment response and RT planning Improve upon late toxicities AND determine the effects of decreased dose IFRT on late toxicities Refine risk categories Improve treatment regimen for high-risk disease

References 1. 2. 3. 4. 5. 6. 7. Devita VT Jr et al. Combination chemotherapy in the treatment of advanced HD. Ann Intern Med 73: 881 -95. 1970 Bonadonna G et al. Combination chemotherapy of HD with adriamycin, bleomycin, vinblastine, and imidazole vs MOPP. Cancer 36: 252 -9, 1975. Donaldson SS et al. HD: Treatment with low dose radiation and chemotherapy. Front Radiat Ther Oncol 16: 122 -33, 1981. Hunger SP et al. ABVD/MOPP and low-dose IFRT in pediatric HD. J Clin Oncol 12: 2160 -6, 1994. Weiner MA et al. Intensive chemotherapy and low-dose RT for the treatment of advanced-stage HD in pediatric patients: A POG study. J Clin Oncol 9: 1591 -98, 1991. Donaldson SS et al. VAMP and low-dose, IFRT for children and adolescents with favorable, early-stage HD: results of a prospective clinical trial. J Clin Oncol 20: 3081– 3087, 2002. Donaldson SS et al. Final results of a prospective clinical trial with VAMP and low -dose IFRT for children with low-risk HD. J Clin Oncol 25: 332– 337, 2007.

References 8. Ruhl U et al. Response adapted RT in the treatment of pediatric HD: an interim 9. 10. 11. 12. 13. 14. report at 5 years of the German GPOH-HD 95 trial. IJROBP, 51: 1209– 1218, 2001. Ruhl U et al. Abstract at ASTRO, 46 th annual meeting: German GPOH-HD 95 trial: Treatment results and analysis of failures in pediatric HD using combination chemotherapy with and without RT. IJROBP 60: S 131, 2004. Landman-Parker Jet al. Localized childhood HD: response-adapted chemotherapy with etoposide, bleomycin, vinblastine, and prednisone before low-dose RT-results of the French MDH 90. J Clin Oncol 18: 1500– 1507, 2000. Kung FH et al. POG 8625: a randomized trial comparing chemotherapy with chemoradiotherapy for children and adolescents with stages I, IIA, IIIA 1 HD: a report from the COG. J Pediatr Hematol Oncol 28: 362– 368, 2006. Nachman JB et al. Randomized comparison of IFRT and no RT for children with HD who achieve a complete response to chemotherapy. J Clin Oncol 20(18): 3765– 3771, 2002. Hudson MM et al. Risk-adapted, combined-modality therapy with VAMP/COP and response-based, IFRT for unfavorable pediatric HD. J Clin Oncol 22: 4541– 4550, 2004. Friedmann AM et al. Treatment of unfavorable childhood HD with VEPA and lowdose, involved-field radiation. J Clin Oncol 20: 3088– 3094, 2002.

References 15. 16. 17. 18. 19. 20. 21. 22. Weiner MA et al. Randomized study of intensive MOPP-ABVD with or without lowdose total-nodal RT in the treatment of HD in pediatric patients: a POG study. J Clin Oncol 15: 2769– 79, 1997. Fryer CJ et al. Efficacy and toxicity of 12 courses of ABVD chemotherapy followed by low-dose regional RT in advanced HD in children: a report from the Children’s Cancer Study Group. J Clin Oncol 8(12): 1971– 1980, 1990. Willman KY, Cox RS, Donaldson SS: Radiation induced height impairment in pediatric HD. IJROBP 28(1): 85– 92, 1994. Constine LS , et al. Thyroid dysfunction after radiotherapy in children with Hodgkin’s disease. Cancer 53: 878 -883, 1984. Sklar C, et al. Abnormalities of the thyroid in survivors of HD: Data from the Childhood Cancer Survivor Study. J Clin Endocrinol Metab 85: 3227 -3232, 2000. Hancock SL, et al. Factors affecting late mortality from heart disease after treatment of Hodgkin’s disease. JAMA 270: 1949 -1955, 1993. Hull MC, et al. Valvular dysfunction and carotid, subclavian, and coronary artery disease in survivors of HD treated with RT. JAMA 290: 2831 -2837, 2003. Villani F, et al. Late pulmonary effects in favorable stage I and IIA HD treated with radiotherapy alone. Am J Clin Oncol 23: 18 -21, 2000.

References 23. Bhatia S et al. Second cancers after pediatric Hodgkin’s disease. J Clin Oncol 16(7): 2570– 2572, 1998. 24. Bhatia S, et al. High risk of subsequent neoplasms continues with extended followup of childhood HD: Report from the Late Effects Study Group. J Clin Oncol 21: 4386 -4394, 2003. 25. Metayer C, et al. Second cancers among long-termsurvivors of Hodgkin’s disease diagnosed in childhood and adolescence. J Clin Oncol 18: 2435 -2443, 2000. 26. Travis LB, et al. Cumulative absolute breast cancer risk for young women treated for Hodgkin lymphoma. J Natl Cancer. Inst 97: 1428 -1437, 2005.