Radionuclides Production Alkhair Adam Khalil B Pharm M

Radionuclides Production Alkhair Adam Khalil, B. Pharm, M. Pharm College of Pharmacy – Karary University Department of Pharmaceutical Chemistry

• Radionuclides are required in both diagnostic and therapeutic nuclear medicine procedures. • The principle difference between nuclear medicine and x-ray procedures is that nuclear medicine relies on image formation by photon emission from the object while x -ray procedures rely on transmission through the patient. • Diagnostic and therapeutic procedures in nuclear medicine utilize radionuclides for this purpose.

• Naturally occurring radionuclides are generally not suitable for diagnostic and therapeutic procedures due to their typically long half-lives or less than ideal physical or chemical characteristics; therefore appropriate radionuclides need to be produced • Radionuclides decay exponentially according to its half-life. As a result, naturally occurring radionuclides tend to have very long half-lives, often thousands of years, or are stable.

The common methods of radionuclide production for nuclear medicine include: 1. Fission, 2. Neutron activation, 3. Cyclotrons, 4. Generators.

1. Fission • Fission is simply splitting the nuclei into two parts; those two parts become new smaller nuclides. • The fission nuclides are generally uranium-235 (235 U) or plutonium-239 (239 Pu). • While this can occur spontaneously in unstable nuclei, in the nuclear reactor we add ‘energy’ by way of a neutron. Given that a neutron has no charge (i. e. no repulsion), they can penetrate a target nucleus without being accelerated to high energies (unlike protons).
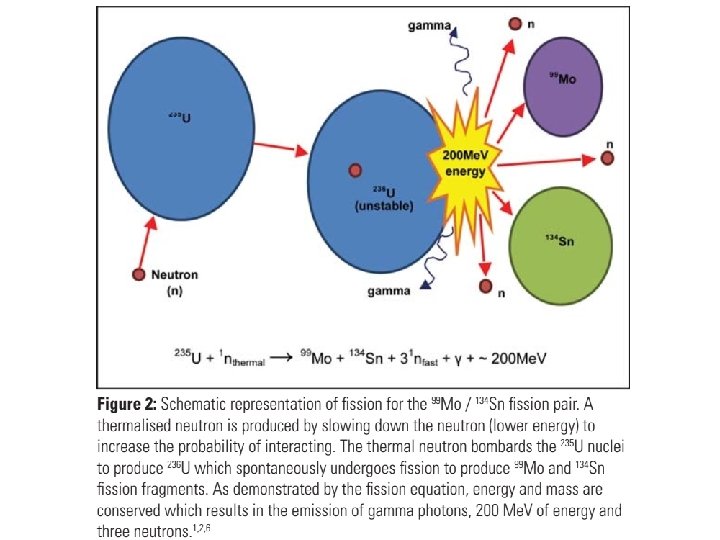

• There is a very wide variety of nuclides that might be produced during fission with mass numbers in the range 70 to 160. • In reality, a single fission reaction is uncommon and, thus, a large number of new atoms might be simultaneously produced in a reactor. We know, however, that iodine-131(131 I) and cesium-137 (137 Cs) are fission pairs.
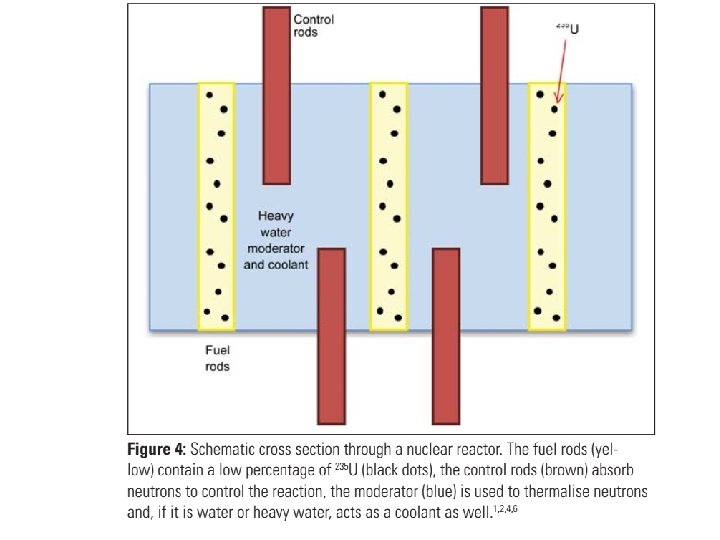

Fill in the missing parts

2. Neutron activation • Neutron activation is also undertaken in the nuclear reactor. The high neutron flux associated with the fission reactions in the reactor can be used to bombard other stable nuclides (target) if they are placed in the reactor. • It is possible to produce radionuclides that are elementally different (that could be separated) from the target using neutron, proton (n, p) and neutron, alpha (n, α) particle reactions. • Unfortunately, the target is not entirely transformed which means there is carrier, or residual target, present.

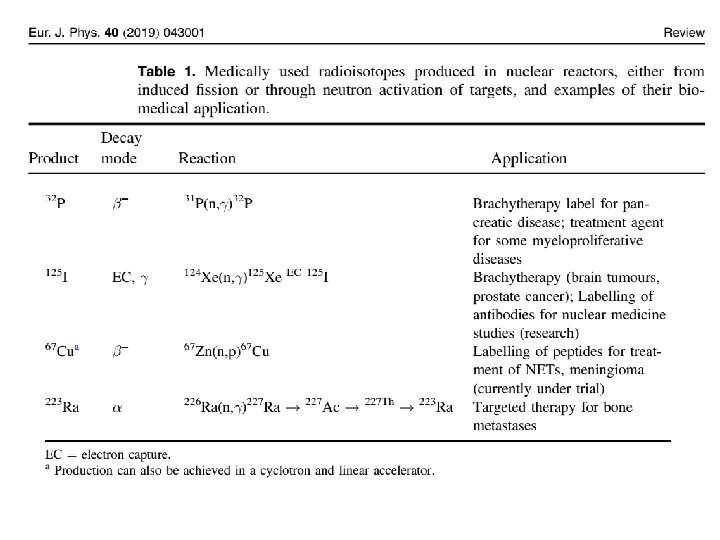
• Similarly, any impurities in the target may result in impurities in the product. • Removal of carrier and impurities is an expensive process and contributes to the high cost of these products. • For this reason, many clinically useful medical radioisotopes that could be created through neutron activation are instead acquired through nuclear fission to maximize the specific activity, e. g. 99 Mo (the parent of 99 m. Tc).

• In some instances the product following a (n, γ) reaction will decay to a daughter product that is of medical interest, which can then be separated from the target, e. g. 125 Xe (54) is produced by neutron activation of 124 Xe, which later decays (by electron capture) to 125 I(53), a medically used radioisotope that can be extracted.
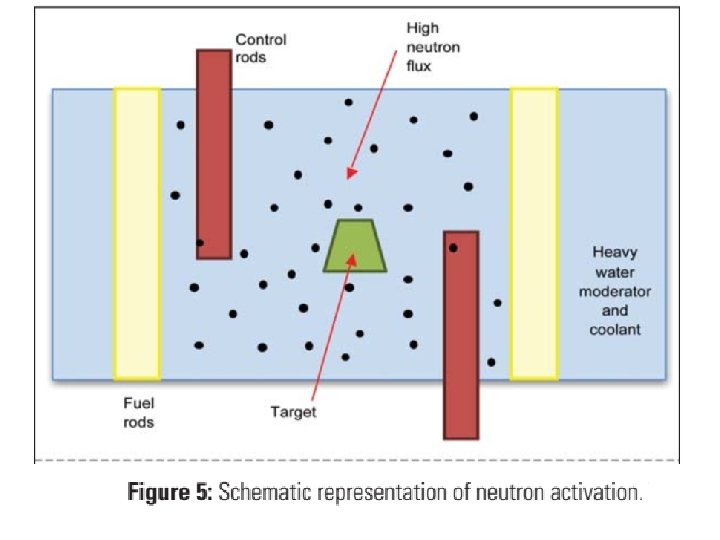


3. Cyclotrons • The cyclotron bombards a stable nuclei with high energy charged particles; protons (p), deuterons (d) (= 2 H) , triton (t) (=3 H)and even alpha (α) (=4 H) particles to penetrate the nucleus of the target, the particles must be accelerated to very high energies (to overcome proton repulsion in nuclei). • A cyclotron can be thought of as a linear accelerator but using a spiral to overcome the long linear distance that would be required to reach the energies required.
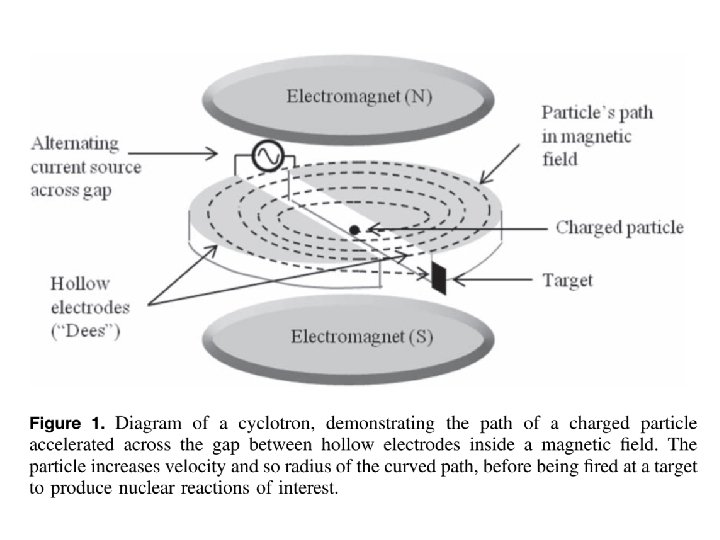

• The radioisotopes produced in cyclotrons will be neutron deficient, as opposed to those produced in a reactor which have a neutron excess, and as such will decay via different means, namely electron capture and positron emission. • Some of the more common cyclotron reactions:
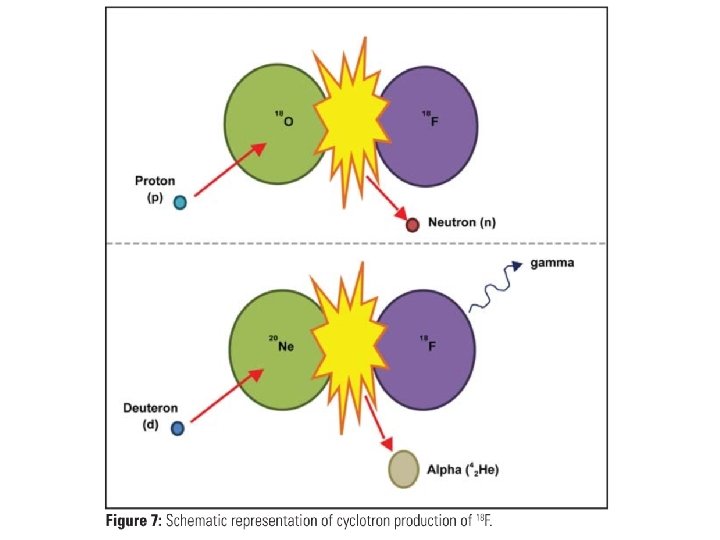

• Cyclotrons are rated based on the energy of the accelerated charged particle. • Small cyclotrons (9 -11 Me. V) are typically limited to producing fluorine-18 (18 F) for positron emission tomography (PET). • Medium sized cyclotrons (in the order of 15 Me. V) can produce a larger array of PET radionuclides (13 N, 11 C, 15 O). • Larger cyclotrons (30 Me. V) are capable of producing other nuclear medicine radionuclides like 67 Ga, 201 Tl, 123 I.


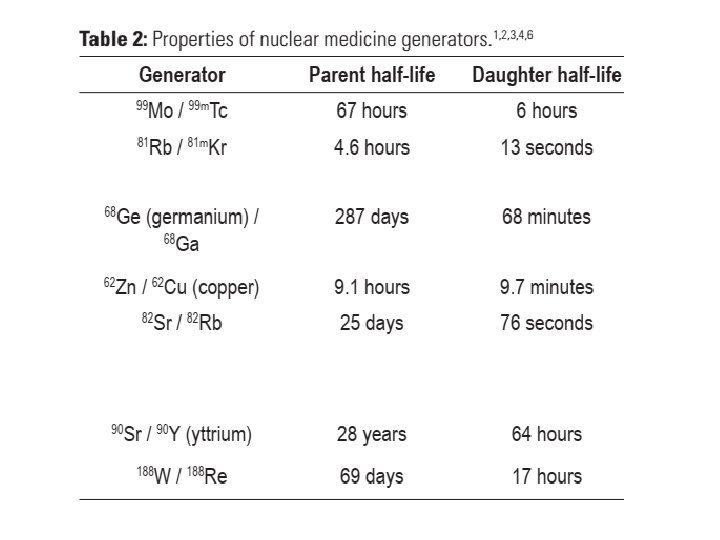
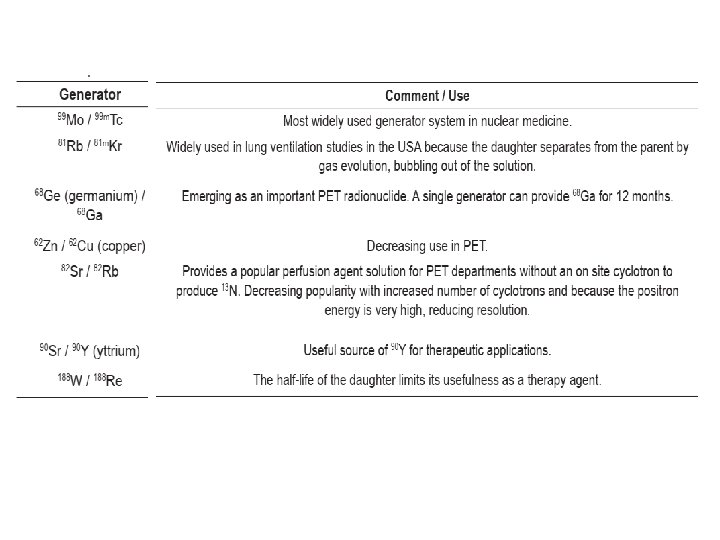
4. Generators • Some radionuclides have sufficiently short half-lives that daily manufacture, quality control and shipping is impractical. • 99 m. Tc is the most widely used radionuclide in nuclear medicine. • A radionuclide generator is a device that allows a weekly supply of a short lived radionuclide to be available on site. • The 99 Mo / 99 m. Tc generator is the most common.

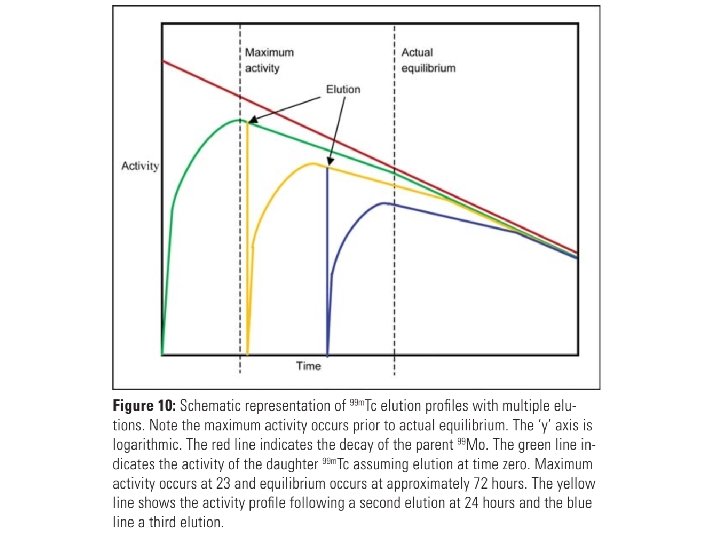
• A radionuclide generator is comprised of a parent/daughter radionuclide pair. • The short lived daughter (e. g. 99 m. Tc) is paired with a longer lived parent (e. g. 99 Mo). • Depending on the half-life of each, regular separation and extraction of the daughter from the parent can occur; elution. • Elution is also referred to as ‘milking the generator’. Elution can be repeated because the parent replenishes the daughter by decay.

• Separation of the parent and daughter is generally based on different physical or chemical properties. • 99 Mo has a 67 hour half-life and it is produced by fission and loaded onto an aluminum column as ammonium molybdenate. • 99 m. Tc has different chemical properties to 99 Mo and so, as the parent decays to the daughter, the 99 m. Tc is less tightly bound to the aluminium column (99 Mo. O 42 - versus 99 m. Tc. O 4 -).
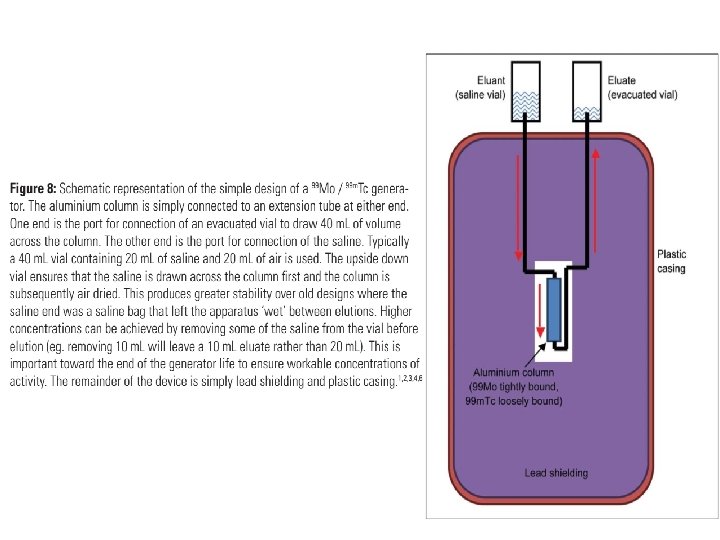

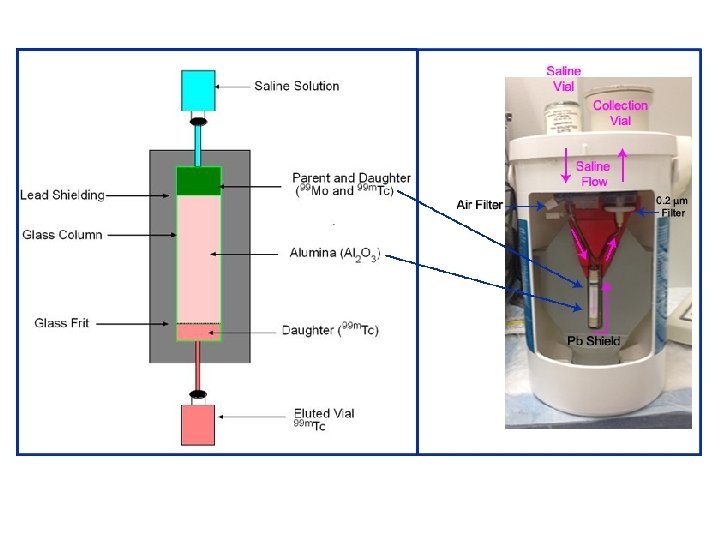
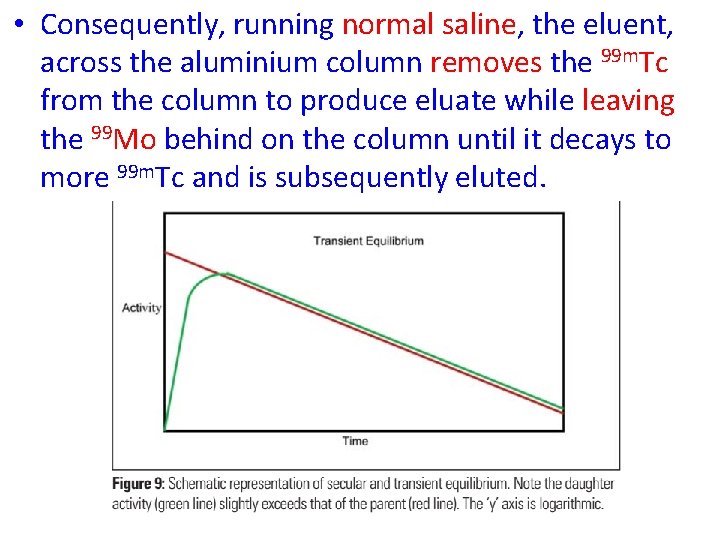
• Consequently, running normal saline, the eluent, across the aluminium column removes the 99 m. Tc from the column to produce eluate while leaving the 99 Mo behind on the column until it decays to more 99 m. Tc and is subsequently eluted.

- Slides: 32