Radiometric Dating clocks in rocks Absolute Dating Gives

Radiometric Dating “clocks in rocks”

Absolute Dating Gives a numerical age n Works best with igneous rocks & fossils n Uses isotopes n

Isotopes- different number of neutrons n Carbon 12 (C) – Atomic mass is on periodic table – stable n Carbon 14 (14 C) – 2 “extra” neutrons – radioactive

Common dating isotopes Parent Daughter Half life Potassium 40 Argon 40 1. 3 by Rubidium 87 Strontium 87 48. 8 by Uranium 235 Lead 207 . 7 by Carbon 14 Nitrogen 14 6, 000 years Sodium 22 Aluminum 27 15 hours

Radiation Radioactive Atom Ionizing Radiation Alpha Particle Neutron Particle Beta Particle Gamma Ray (X Ray)

radioactivity n n Nuclei break apart Emit particles or waves (radiation)

The average annual dose to the general population from natural background and man-made sources is 360 mrem. Terrestrial Sources Radon Cosmic Radiation Radon Internal Sources Other
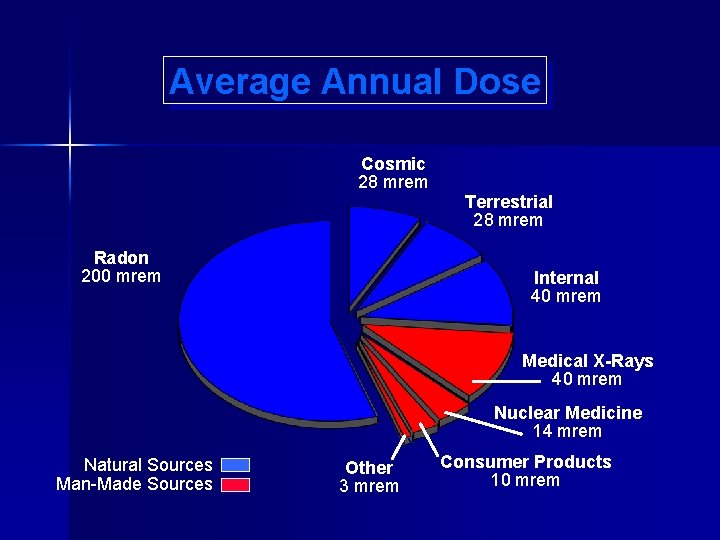
Average Annual Dose Cosmic 28 mrem Radon 200 mrem Terrestrial 28 mrem Internal 40 mrem Medical X-Rays 40 mrem Nuclear Medicine 14 mrem Natural Sources Man-Made Sources Other 3 mrem Consumer Products 10 mrem
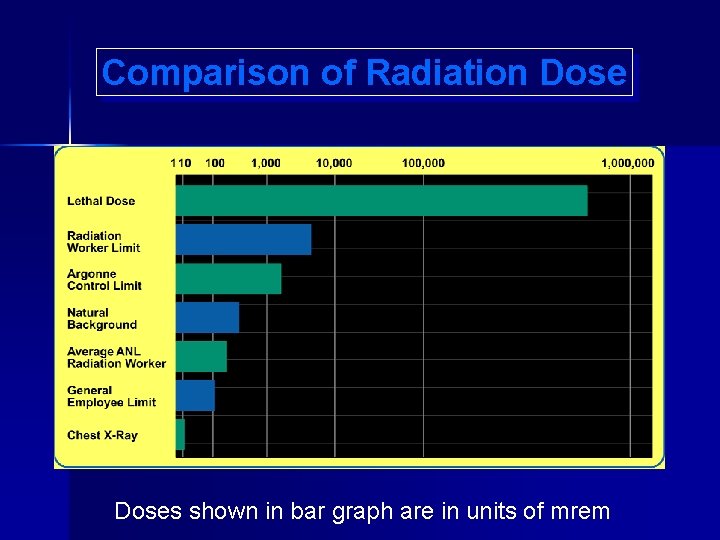
Comparison of Radiation Doses shown in bar graph are in units of mrem
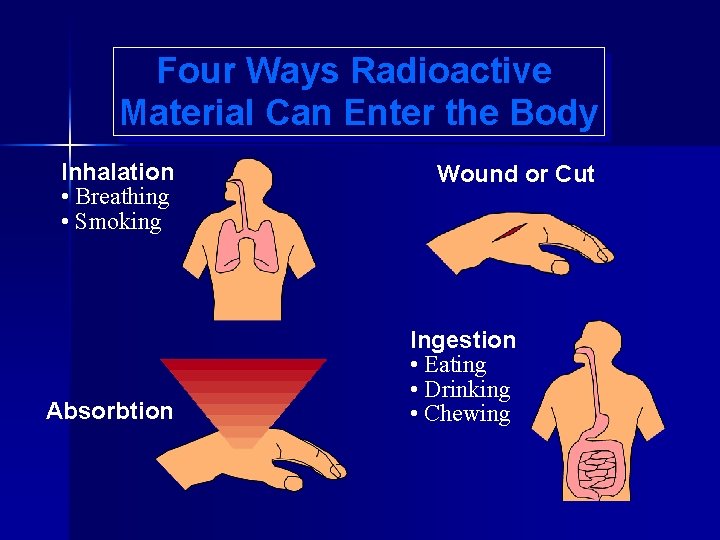
Four Ways Radioactive Material Can Enter the Body Inhalation • Breathing • Smoking Absorbtion Wound or Cut Ingestion • Eating • Drinking • Chewing
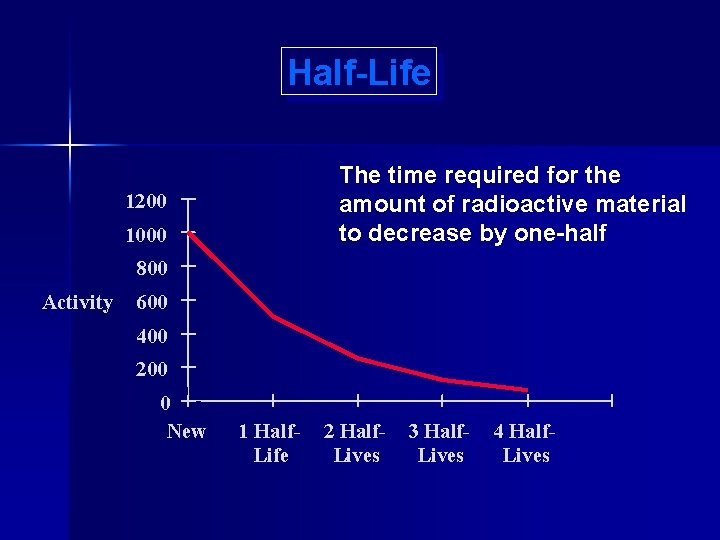
Half-Life The time required for the amount of radioactive material to decrease by one-half 1200 1000 800 Activity 600 400 200 0 New 1 Half. Life 2 Half. Lives 3 Half. Lives 4 Half. Lives
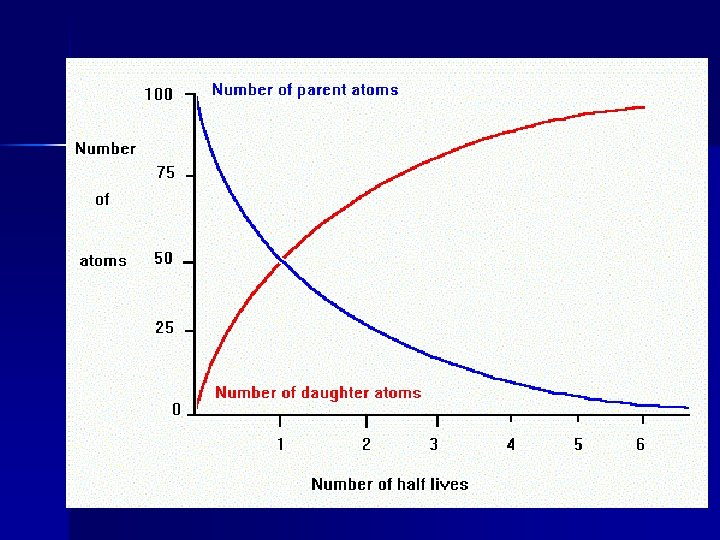
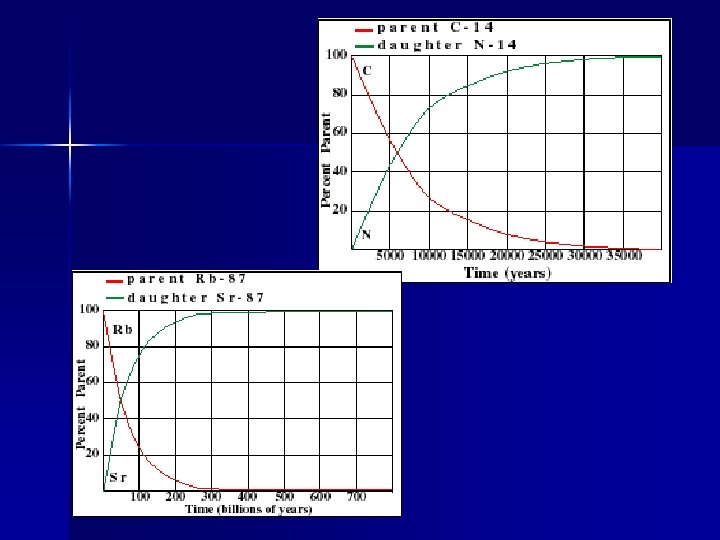

Half-lives Parent Daughter Half life Potassium 40 Argon 40 1. 3 by Rubidium 87 Strontium 87 48. 8 by Uranium 235 Lead 207 . 7 by Carbon 14 Nitrogen 14 6, 000 years Sodium 22 Aluminum 27 15 hours

Half life problem n If you begin with 80 grams of 14 C after a time, 20 grams are left. How old is the sample?

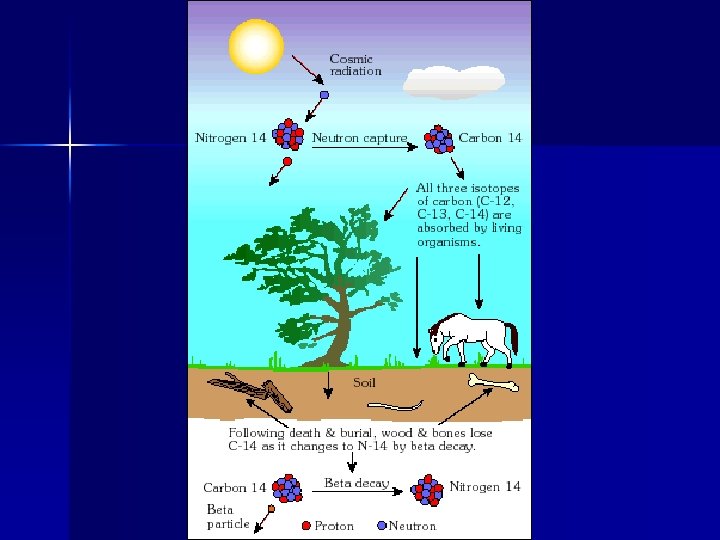
14 Carbon Only accurate for ages less than 100, 000 years n Parent gets too small to accurately measure n

Mass spectrometer measures D/P ratio
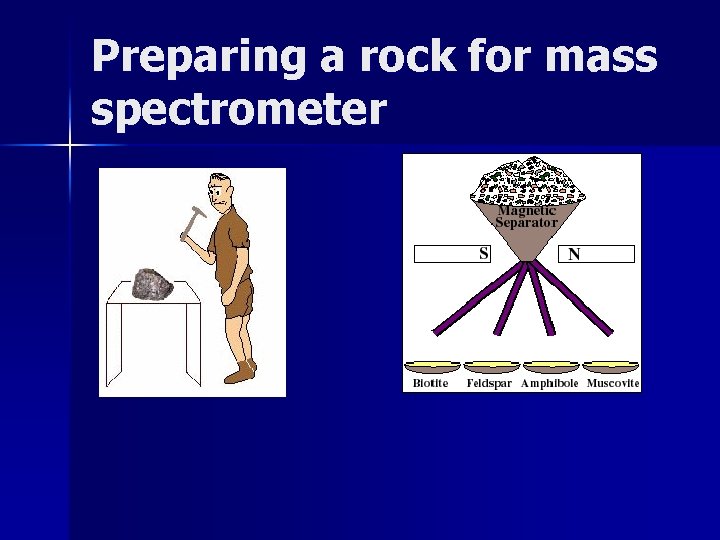
Preparing a rock for mass spectrometer

Using daughter/parent ratio 1) D/P ratio n 2) figure out number of half-lives n – Use graph n 3) multiply number of half-lives by the time of one half-live n Example: 240 g 14 N ; 8 g 14 C

Another half-life problem n 1, 000 grams of radioactive element is in a rock when it is formed. The element’s half-life is 2 million years. After a time, 125 grams of the original element remain. How old is the rock?
- Slides: 21