Radiolysis in CANDU Coolant and its Effect on










![Chemical Additives/Dissolved Species Effect of [Fe 2+]o on [H 2 O 2]SS Ar, p. Chemical Additives/Dissolved Species Effect of [Fe 2+]o on [H 2 O 2]SS Ar, p.](https://slidetodoc.com/presentation_image_h/ac08be357a7cceabd8648712a286899b/image-29.jpg)


- Slides: 32

Radiolysis in CANDU Coolant and its Effect on Chemistry and Materials International Association for the Properties of Water and Steam Workshop May 11 & 12, 2009 Jungsook Clara Wren Department of Chemistry University of Western Ontario 1

Presentation Outline 1. Overview of NSERC/AECL Industrial Research Chair Program 2. Preliminary Results from -Radiolysis Induced Carbon Steel Corrosion at 150 o. C 3. Importance of Steady-State Radiolysis on Steel Corrosion 2

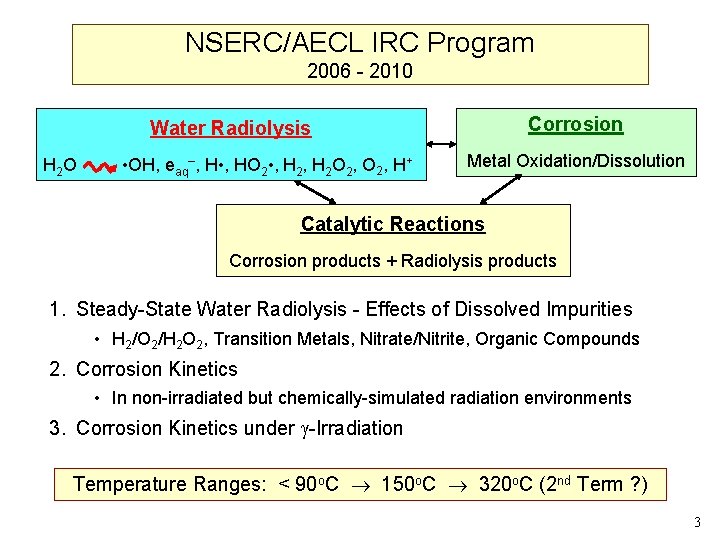
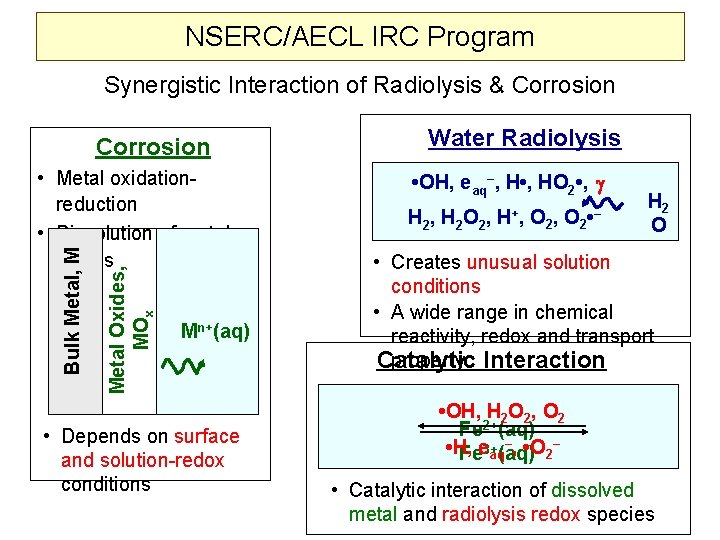
NSERC/AECL IRC Program 2006 - 2010 Corrosion Water Radiolysis H 2 O • OH, eaq–, H • , HO 2 • , H 2 O 2, H+ Metal Oxidation/Dissolution Catalytic Reactions Corrosion products + Radiolysis products 1. Steady-State Water Radiolysis - Effects of Dissolved Impurities • H 2/O 2/H 2 O 2, Transition Metals, Nitrate/Nitrite, Organic Compounds 2. Corrosion Kinetics • In non-irradiated but chemically-simulated radiation environments 3. Corrosion Kinetics under -Irradiation Temperature Ranges: < 90 o. C 150 o. C 320 o. C (2 nd Term ? ) 3

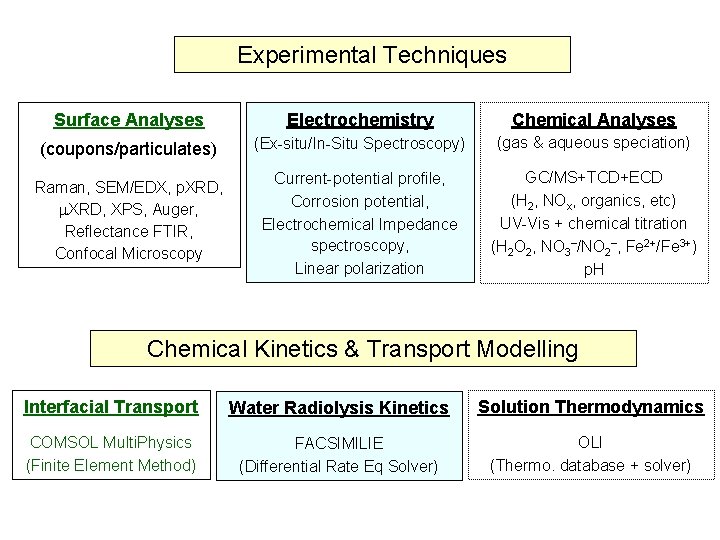
Experimental Techniques Surface Analyses Electrochemistry Chemical Analyses (coupons/particulates) (Ex-situ/In-Situ Spectroscopy) (gas & aqueous speciation) Raman, SEM/EDX, p. XRD, XPS, Auger, Reflectance FTIR, Confocal Microscopy Current-potential profile, Corrosion potential, Electrochemical Impedance spectroscopy, Linear polarization GC/MS+TCD+ECD (H 2, NOx, organics, etc) UV-Vis + chemical titration (H 2 O 2, NO 3 /NO 2 , Fe 2+/Fe 3+) p. H Chemical Kinetics & Transport Modelling Interfacial Transport Water Radiolysis Kinetics Solution Thermodynamics COMSOL Multi. Physics (Finite Element Method) FACSIMILIE (Differential Rate Eq Solver) OLI (Thermo. database + solver)

-Radiolysis on Carbon Steel Corrosion Coupon Studies at T 150 o. C Dose Rate 6. 4 k. Gy/ h These studies are performed as a function of p. H, T, irradiation time, dissolved H 2 and O 2 (cover gas), buffer 5

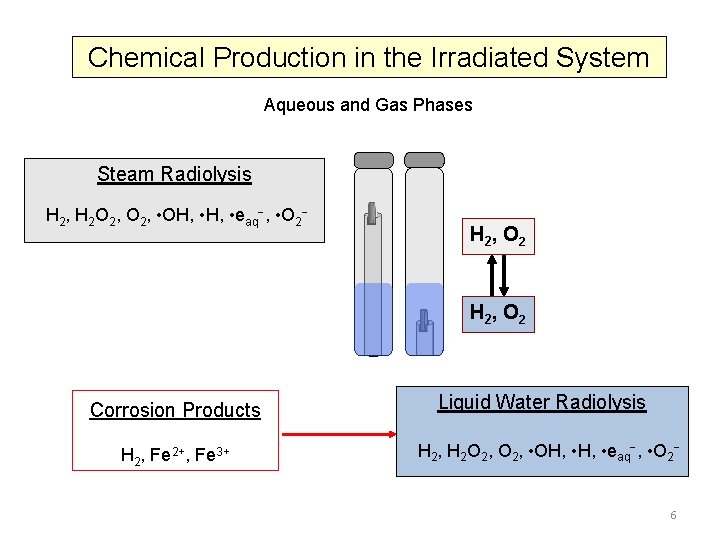
Chemical Production in the Irradiated System Aqueous and Gas Phases Steam Radiolysis H 2, H 2 O 2, • OH, • eaq , • O 2 H 2 , O 2 Corrosion Products H 2, Fe 2+, Fe 3+ Liquid Water Radiolysis H 2, H 2 O 2, • OH, • eaq , • O 2 6

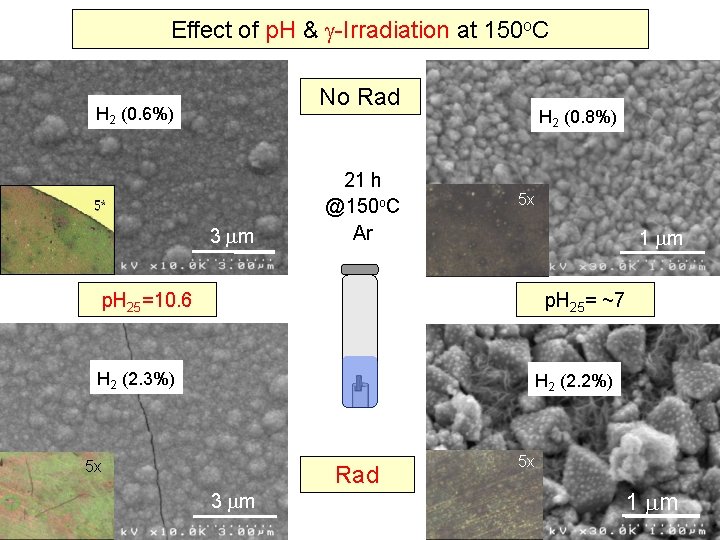
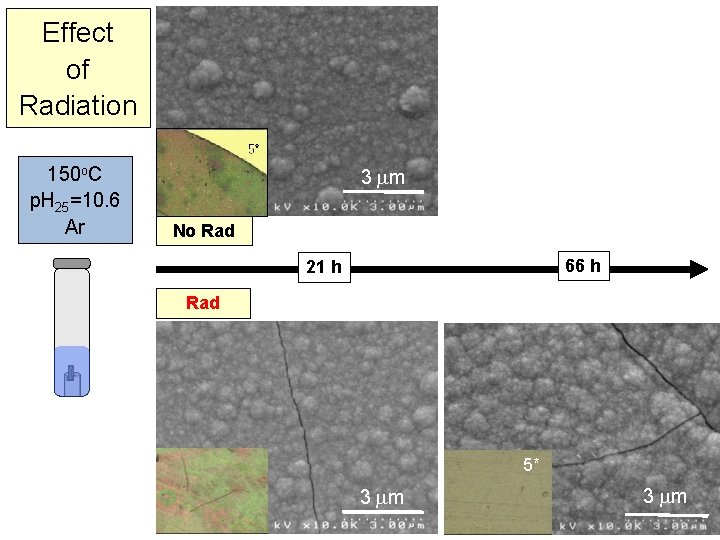
Effect of p. H & -Irradiation at 150 o. C No Rad 3 m 21 h @150 o. C Ar 5 x 1 m p. H 25=10. 6 p. H 25= ~7 5 x 3 m Rad 5 x 1 m 7

Effect of p. H & -Irradiation at 150 o. C No Rad H 2 (0. 6%) 3 m 21 h @150 o. C Ar H 2 (0. 8%) 5 x 1 m p. H 25=10. 6 p. H 25= ~7 H 2 (2. 3%) H 2 (2. 2%) 5 x 3 m Rad 5 x 1 m 8

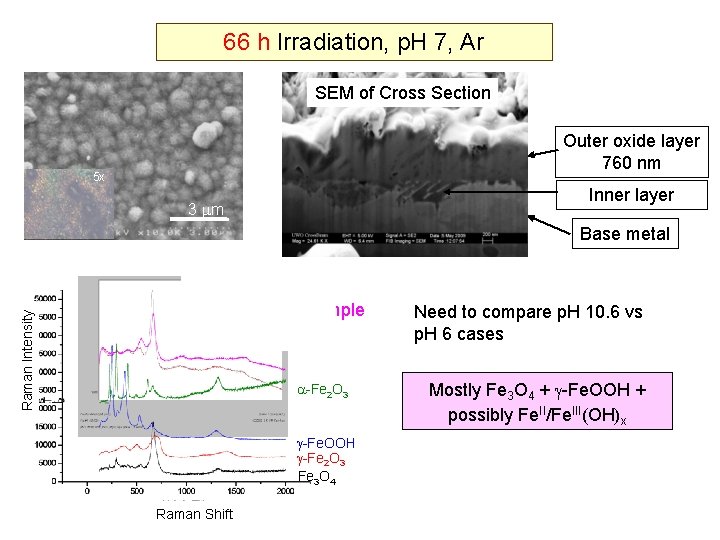
66 h Irradiation at 150 o. C, Ar Aq. Phase Cross Section p. H 25 = 10. 6 Outer oxide layer 2. 2 m 5 x Inner layer 3 m 9 Base metal 1 m Aq. Phase Cross Section p. H 25 = 7 Outer oxide layer 760 nm 5 x 3 m Inner layer 9 Base metal 200 nm

Cross Section Raman Spectroscopy 66 h Irradiation, p. H 10. 6, Ar Raman Intensity Irradiated Sample A Mixed Phase ? ? -Fe 2 O 3 -Fe. OOH -Fe 2 O 3 Fe 3 O 4 / -Fe 2 O 3 / Fe. II/Fe. III(OH)x No significant -Fe 2 O 3 Fe 3 O 4 Raman Shift Confocal Raman Microscopy, small angle XRD for depth profile are underway

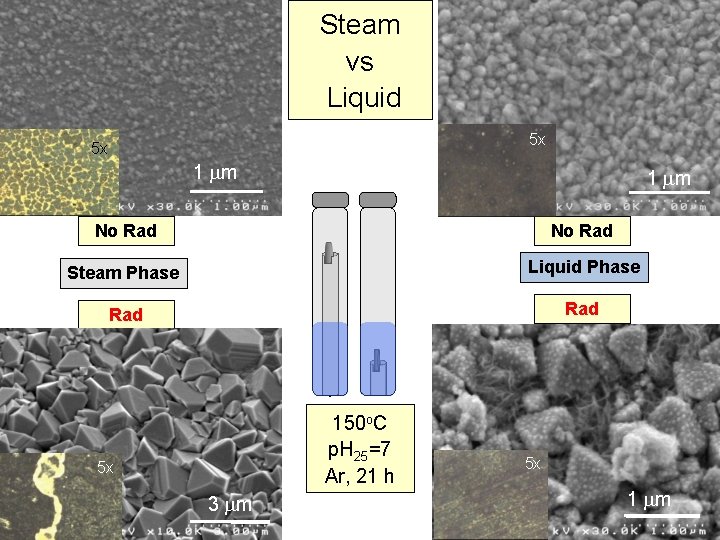
Steam vs Liquid 5 x 5 x 1 m No Rad Steam Phase Liquid Phase Rad 150 o. C p. H 25=7 Ar, 21 h 5 x 3 m 5 x 1 m 11

Preliminary Conclusions Coupon Studies at 150 o. C • Kinetics is still important at 150 o. C • Steady-state irradiation enhances surface oxide formation • The type of oxide depends on the rate of oxidation and p. H • Aqueous corrosion on CS under -irradiation is uniform, and does not show localized corrosion • At p. H 25 10. 6, -irradiation appears to promote more compact oxide • Corrosion in the steam phase is more inhomogeneous Steady-State Radiolysis affects carbon steel corrosion behaviour

Water Radiolysis Solvent Oriented Process Physical (chemical) Stage Primary Radiolysis Yields (G-values) Bulk Phase Chemistry Stage 13

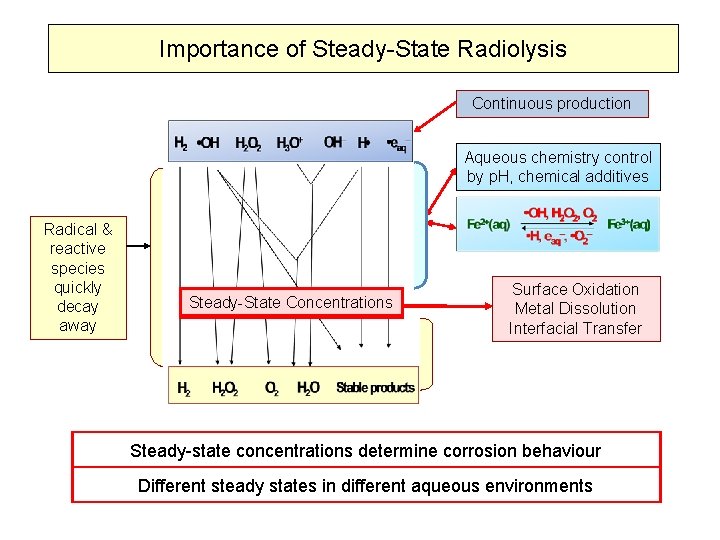
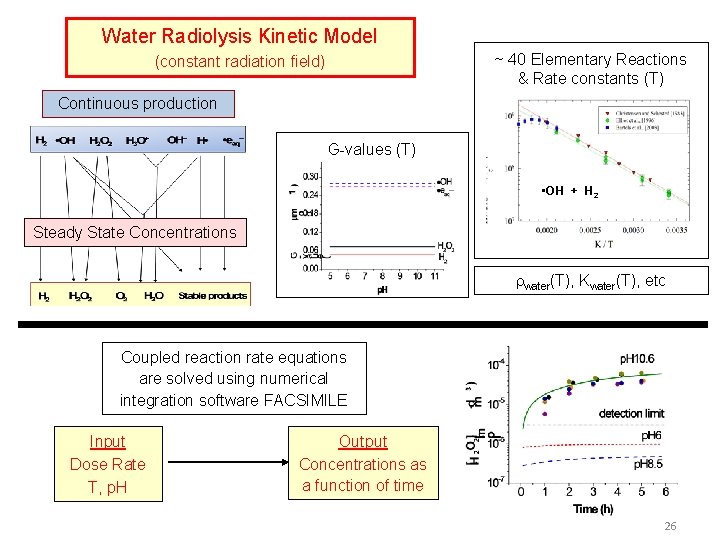
Importance of Steady-State Radiolysis Continuous production Aqueous chemistry control by p. H, chemical additives Radical & reactive species quickly decay away Steady-State Concentrations Surface Oxidation Metal Dissolution Interfacial Transfer Steady-state concentrations determine corrosion behaviour Different steady states in different aqueous environments

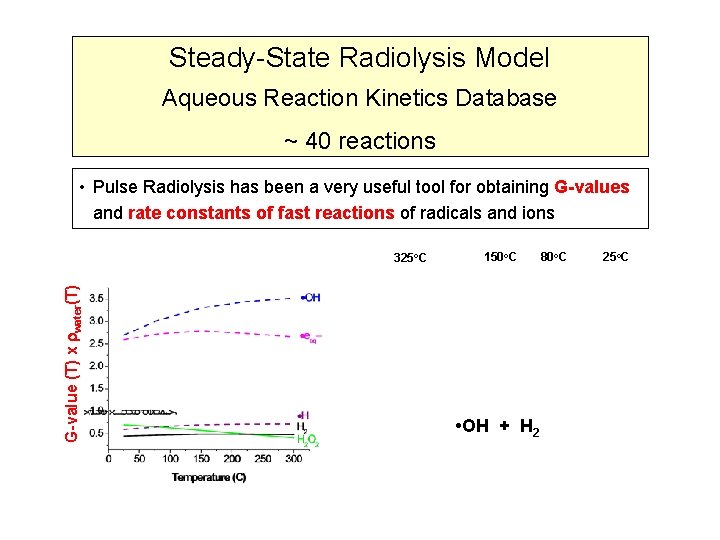
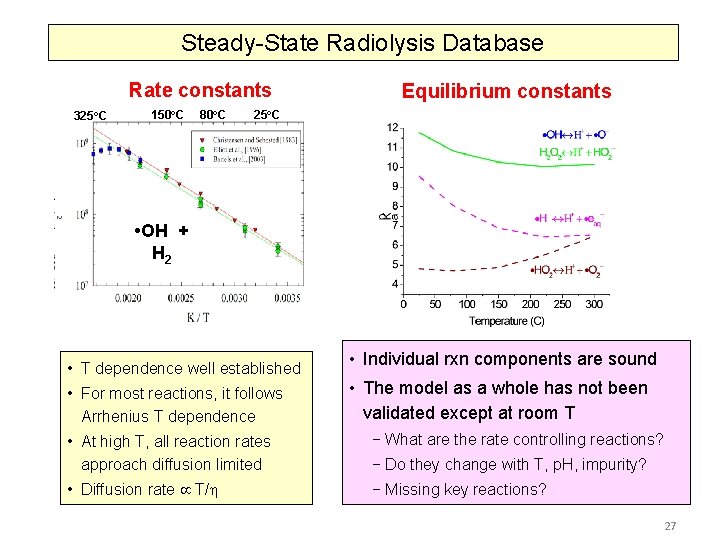
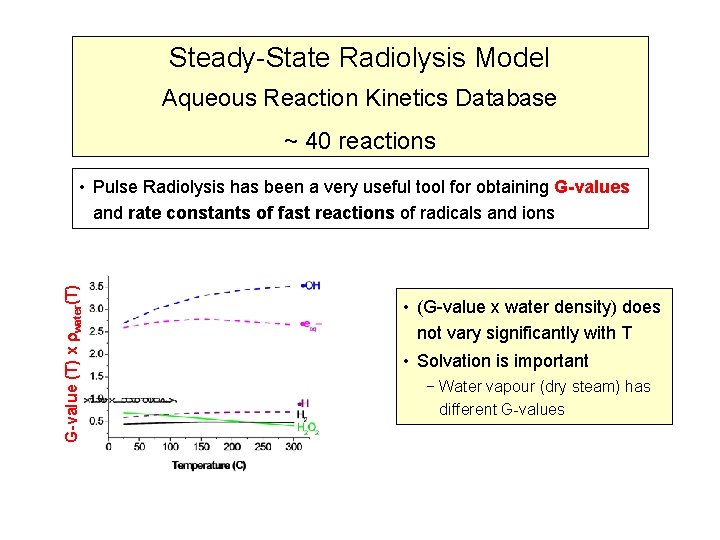
Steady-State Radiolysis Model Aqueous Reaction Kinetics Database ~ 40 reactions • Pulse Radiolysis has been a very useful tool for obtaining G-values and rate constants of fast reactions of radicals and ions G-value (T) x water(T) 325 o. C 150 o. C • OH + H 2 80 o. C 25 o. C

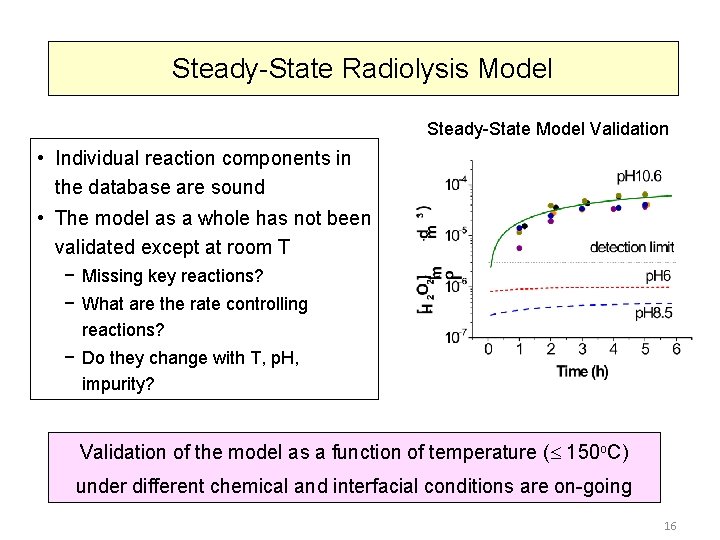
Steady-State Radiolysis Model Steady-State Model Validation • Individual reaction components in the database are sound • The model as a whole has not been validated except at room T − Missing key reactions? − What are the rate controlling reactions? − Do they change with T, p. H, impurity? Validation of the model as a function of temperature ( 150 o. C) under different chemical and interfacial conditions are on-going 16

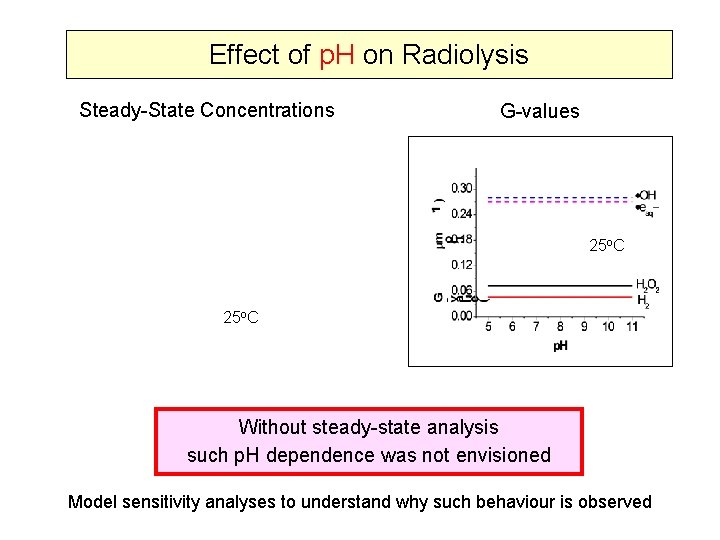
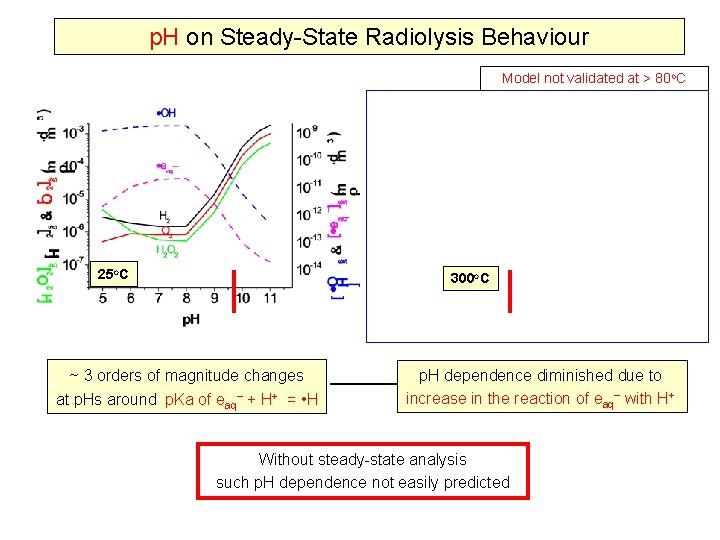
Effect of p. H on Radiolysis Steady-State Concentrations G-values 25 o. C Without steady-state analysis such p. H dependence was not envisioned Model sensitivity analyses to understand why such behaviour is observed

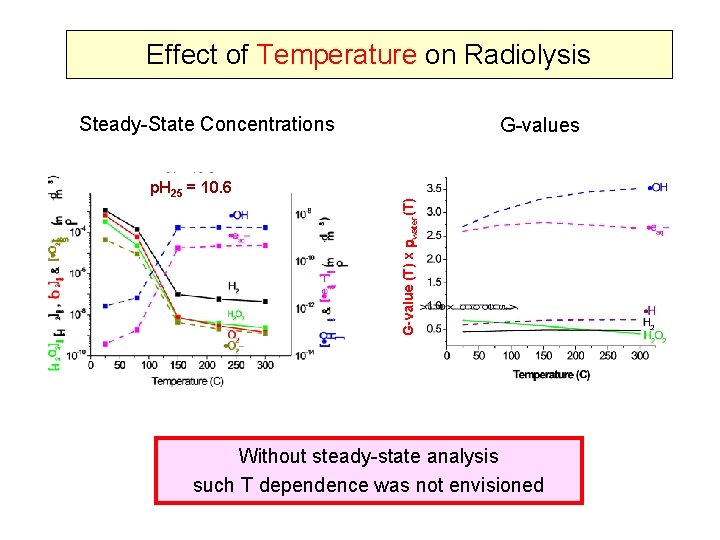
Effect of Temperature on Radiolysis Steady-State Concentrations G-value (T) x water(T) p. H 25 = 10. 6 G-values Without steady-state analysis such T dependence was not envisioned

Summary at T 150 o. C • Radiolysis affects carbon steel corrosion – Thermodynamic considerations are not sufficient – Steady-state concentrations of radiolytic products determine surface oxide formation/transformation • Steady-state radiolysis behaviour strongly depends on p. H, T, chemical additives, dose rate, etc, – These dependences are not well established – Pulse radiolysis studies are not sufficient • Molecular, not radical, products are more important for aqueous corrosion in basic solutions – The relative importance of radical species may increase with T – Significant implication in chemistry control

Acknowledgement Dr. Jamie Noel Dr. X John Zhang Dr. Peter Keech Dr. Jiju Joseph Dr. Sriya Peiris Dr. Sergey Mitlin Dong Fu Sarah Pretty Kevin Daub Katy Yazdanfar Pam Yakabuskie Susan Howett

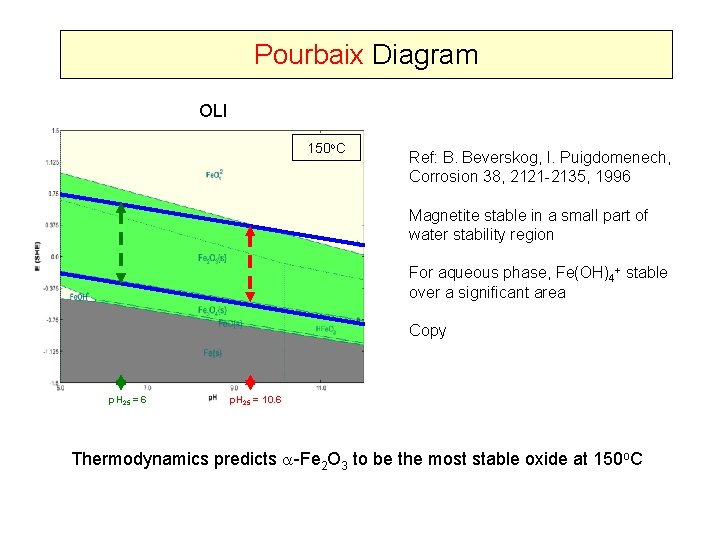
Pourbaix Diagram OLI 150 o. C Ref: B. Beverskog, I. Puigdomenech, Corrosion 38, 2121 -2135, 1996 Magnetite stable in a small part of water stability region For aqueous phase, Fe(OH)4+ stable over a significant area Copy p. H 25 = 6 p. H 25 = 10. 6 Thermodynamics predicts -Fe 2 O 3 to be the most stable oxide at 150 o. C

NSERC/AECL IRC Program Synergistic Interaction of Radiolysis & Corrosion Metal Oxides, MOx Bulk Metal, M • Metal oxidationreduction • Dissolution of metal oxides Mn+(aq) • Depends on surface and solution-redox conditions Water Radiolysis • OH, eaq–, H • , HO 2 • , H 2 , H 2 O 2 , H+ , O 2 , O 2 • – H 2 O • Creates unusual solution conditions • A wide range in chemical reactivity, redox and transport property Interaction Catalytic • OH, H 2 O 2, O 2 Fe 2+(aq) –, • O – • H, 2 Fee 3+aq(aq) • Catalytic interaction of dissolved metal and radiolysis redox species

Effect of Radiation 150 o. C p. H 25=10. 6 Ar 3 m No Rad 66 h 21 h Rad 5* 3 m 23

Effect of Radiation 5* 150 o. C p. H 25=6 Ar m 13 m No Rad 66 h 21 h Rad 5* 5* 1 m 3 m 5* 3 m 24

66 h Irradiation, p. H 7, Ar SEM of Cross Section Outer oxide layer 760 nm 5 x Inner layer 3 3 m m 25 Raman Intensity Base metal Irradiated Sample -Fe 2 O 3 -Fe. OOH -Fe 2 O 3 Fe 3 O 4 Raman Shift Need to compare p. H 10. 6 vs p. H 6 cases Mostly Fe 3 O 4 + -Fe. OOH + possibly Fe. II/Fe. III(OH)x

Water Radiolysis Kinetic Model ~ 40 Elementary Reactions & Rate constants (T) (constant radiation field) Continuous production G-values (T) • OH + H 2 Steady State Concentrations water(T), Kwater(T), etc Coupled reaction rate equations are solved using numerical integration software FACSIMILE Input Dose Rate T, p. H Output Concentrations as a function of time 26

Steady-State Radiolysis Database Rate constants 325 o. C 150 o. C 80 o. C Equilibrium constants 25 o. C • OH + H 2 • T dependence well established • For most reactions, it follows Arrhenius T dependence • Individual rxn components are sound • The model as a whole has not been validated except at room T • At high T, all reaction rates approach diffusion limited − What are the rate controlling reactions? • Diffusion rate T/ − Missing key reactions? − Do they change with T, p. H, impurity? 27

p. H on Steady-State Radiolysis Behaviour Model not validated at > 80 o. C 25 o. C 300 o. C ~ 3 orders of magnitude changes at p. Hs around p. Ka of eaq + H+ = • H p. H dependence diminished due to increase in the reaction of eaq with H+ Without steady-state analysis such p. H dependence not easily predicted
![Chemical AdditivesDissolved Species Effect of Fe 2o on H 2 O 2SS Ar p Chemical Additives/Dissolved Species Effect of [Fe 2+]o on [H 2 O 2]SS Ar, p.](https://slidetodoc.com/presentation_image_h/ac08be357a7cceabd8648712a286899b/image-29.jpg)
Chemical Additives/Dissolved Species Effect of [Fe 2+]o on [H 2 O 2]SS Ar, p. H = 10. 6 Effect of Radiolysis on Iron Solubility [Fe 2+]o = 0, 5 10 -5, 1 10 -4 M Synergistic interaction between corrosion products and radiolysis products • OH, H 2 O 2, O 2 Fe 2+(aq) • H, Fe eaq 3+–(aq) , • O 2–


Steady-State Radiolysis Model Aqueous Reaction Kinetics Database ~ 40 reactions G-value (T) x water(T) • Pulse Radiolysis has been a very useful tool for obtaining G-values and rate constants of fast reactions of radicals and ions • (G-value x water density) does not vary significantly with T • Solvation is important − Water vapour (dry steam) has different G-values

Aqueous Reaction Kinetics Database • Pulse Radiolysis has been a very useful tool for obtaining G-values and rate constants of fast reactions of radicals and ions Rate constants 325 o. C 150 o. C 80 o. C 25 o. C • T dependence well established • For most reactions, it follows Arrhenius T dependence • OH + H 2 • At high T, all reaction rates approach diffusion limits • Diffusion rate T/