Radiology The study of electromagnetic radiation the movement










- Slides: 14

Radiology The study of electromagnetic radiation, the movement of photons, and their ability to create an image.

Why do we care about Radiology? Benefits? Disadvantages?

Chapters 1 & 2: The Technical Side of Imaging Basic concepts that impact x-ray settings: Fractions, +, -, x Proportionality Units of measurement Metric prefixes 3

Math Matters! Fractions “Pieces of the pie” Numerator/Denominator Used in technique charts m. A X seconds (fraction) Multiplication Used for exposure times m. A X seconds = m. As 4

Proportionality Definition: The relationship between two numbers Two types: direct & indirect Direct 1 st number is a multiple of the 2 nd Shown as fraction or with a colon Radiology example? Medical example? 5

Proportionality Indirect 1 st number up = 2 nd number down Product stays the same, but values differ Radiology example Inverse square law Medical example? 6

Length and Time Meter is standard unit Thickness of the patient Distance of x-ray tube to the patient’s body Second is standard unit Exposure time Processing time Understand metric prefixes Needed for conversions

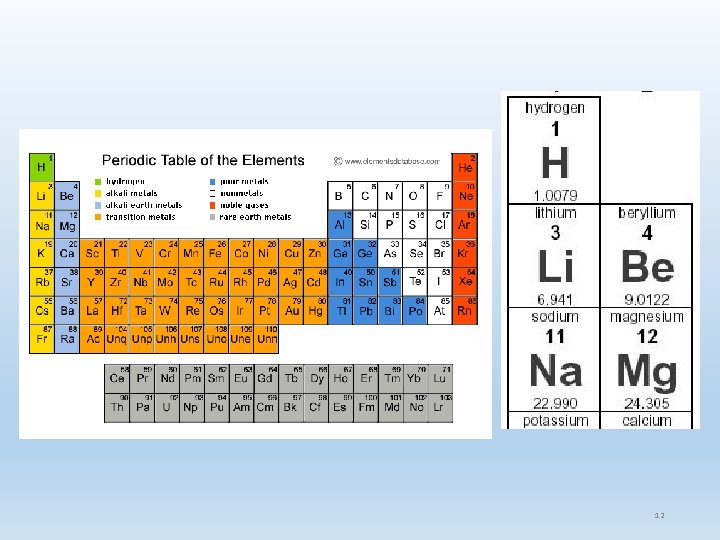
The Atom & Radioactivity Atomic Theory Atomic Structure Combining Atoms Periodic Table t igh Atomic We Organizing Matter 8

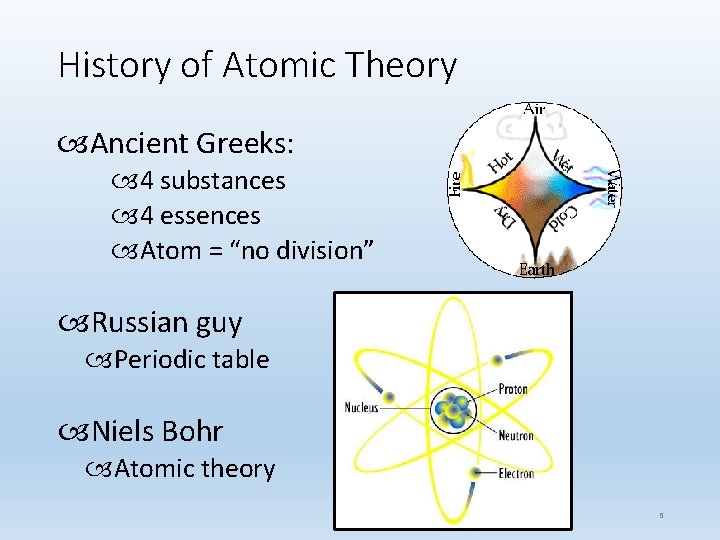
History of Atomic Theory Ancient Greeks: 4 substances 4 essences Atom = “no division” Russian guy Periodic table Niels Bohr Atomic theory 9

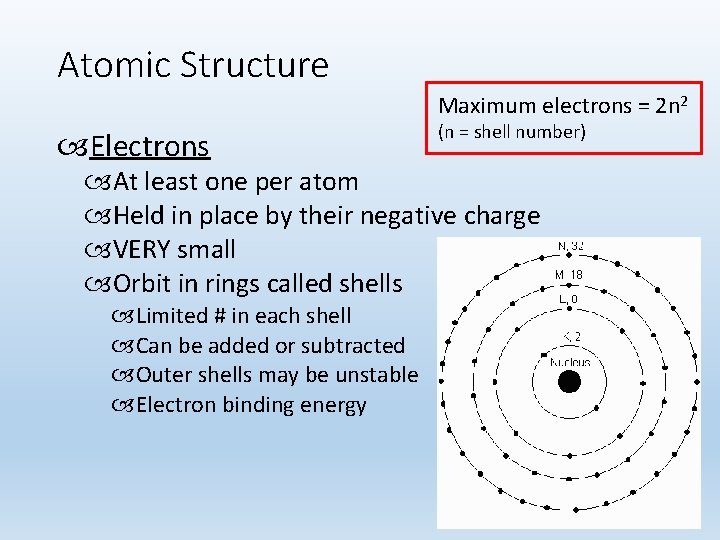
Atomic Structure Maximum electrons = 2 n 2 Electrons (n = shell number) At least one per atom Held in place by their negative charge VERY small Orbit in rings called shells Limited # in each shell Can be added or subtracted Outer shells may be unstable Electron binding energy 10

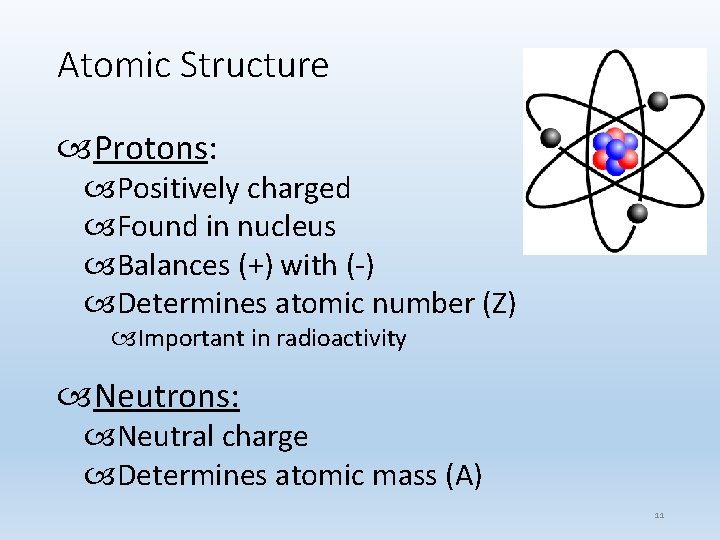
Atomic Structure Protons: Positively charged Found in nucleus Balances (+) with (-) Determines atomic number (Z) Important in radioactivity Neutrons: Neutral charge Determines atomic mass (A) 11

12

Organization of Matter Atoms elements molecules tissues Isotopes: Same atomic number but different mass Can be stable or unstable Important in imaging – contrast studies and nuclear medicine Radioactivity: Atom lives in an abnormal state of excitement …but all matter seeks stability Particles and energy are released & form new atoms 13

Organization of Matter Ionization: Lower electron binding energy leads to electrons leaving their shell imbalance in electrical charge = Atom is now ionized Radiology importance? 14