Radiology Pathology Pancreatic Pathology Before You Begin This

Radiology - Pathology Pancreatic Pathology

Before You Begin This module is intended primarily for pre-clinical students learning or reviewing pathophysiology. Please note that this series will focus on how pathology presents in imaging studies. It assumes familiarity with fundamental anatomy. If you need to learn or review this core concept, please visit the “Anatomy” section of our website. If material is repeated from another module, it will be outlined as this text is so that you are aware

Pancreatic Imaging Evaluation • Ultrasound: • Quick, cheap • No radiation • Often obscured by bowel gas • CT: • Quick. • Pancreatic protocol for lesion detection and characterization • MRI: Best characterization of ductal anatomy and variants disease as well as characterization of masses • Respiratory/Patient motion sensitive • Expensive • Contraindications (i. e. implanted metal)
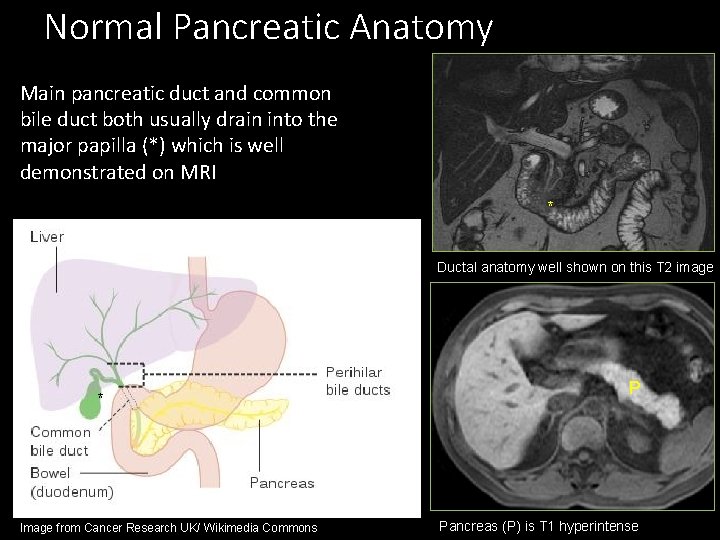
Normal Pancreatic Anatomy Main pancreatic duct and common bile duct both usually drain into the major papilla (*) which is well demonstrated on MRI * Ductal anatomy well shown on this T 2 image * Image from Cancer Research UK/ Wikimedia Commons P Pancreas (P) is T 1 hyperintense
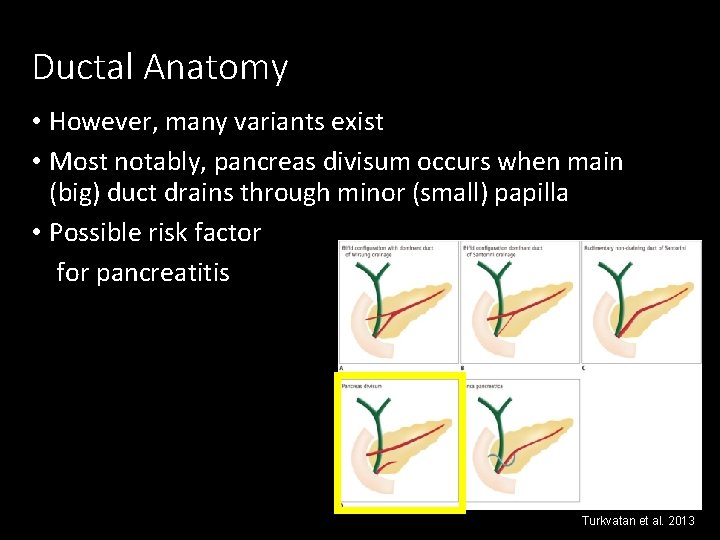
Ductal Anatomy • However, many variants exist • Most notably, pancreas divisum occurs when main (big) duct drains through minor (small) papilla • Possible risk factor for pancreatitis Turkvatan et al. 2013
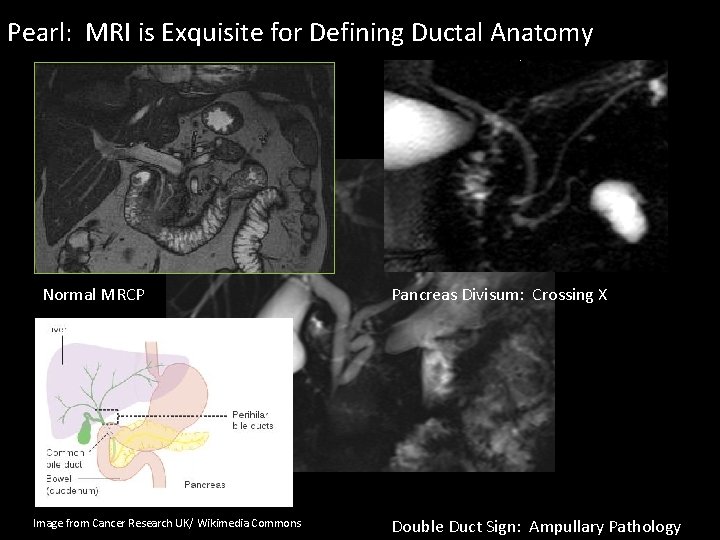
Pearl: MRI is Exquisite for Defining Ductal Anatomy Normal MRCP Image from Cancer Research UK/ Wikimedia Commons Pancreas Divisum: Crossing X Double Duct Sign: Ampullary Pathology
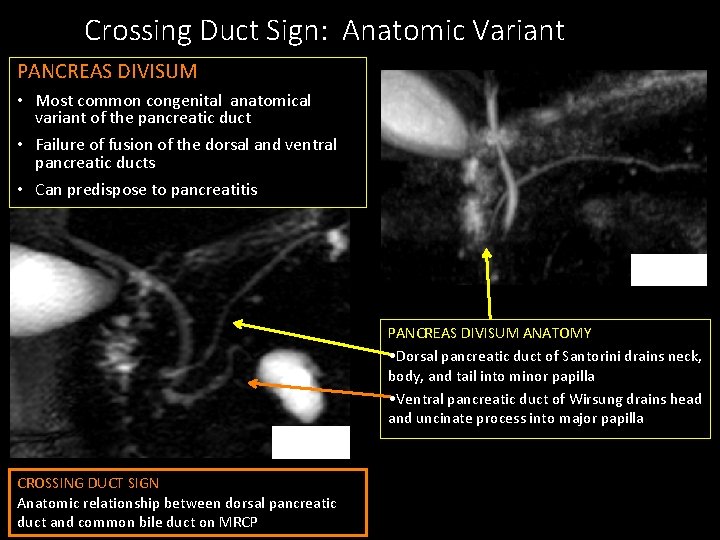
Crossing Duct Sign: Anatomic Variant PANCREAS DIVISUM • Most common congenital anatomical variant of the pancreatic duct • Failure of fusion of the dorsal and ventral pancreatic ducts • Can predispose to pancreatitis MRCP PANCREAS DIVISUM ANATOMY • Dorsal pancreatic duct of Santorini drains neck, body, and tail into minor papilla • Ventral pancreatic duct of Wirsung drains head and uncinate process into major papilla MRCP CROSSING DUCT SIGN Anatomic relationship between dorsal pancreatic duct and common bile duct on MRCP
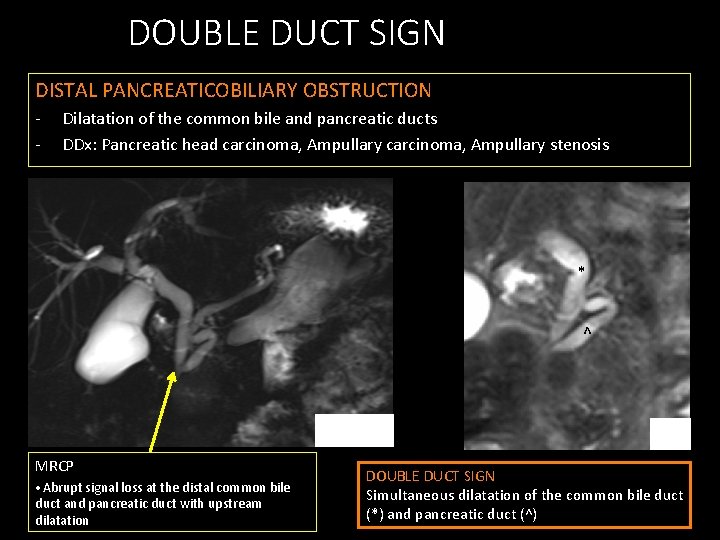
DOUBLE DUCT SIGN DISTAL PANCREATICOBILIARY OBSTRUCTION - Dilatation of the common bile and pancreatic ducts DDx: Pancreatic head carcinoma, Ampullary stenosis * ^ MRCP • Abrupt signal loss at the distal common bile duct and pancreatic duct with upstream dilatation T 2 DOUBLE DUCT SIGN Simultaneous dilatation of the common bile duct (*) and pancreatic duct (^)
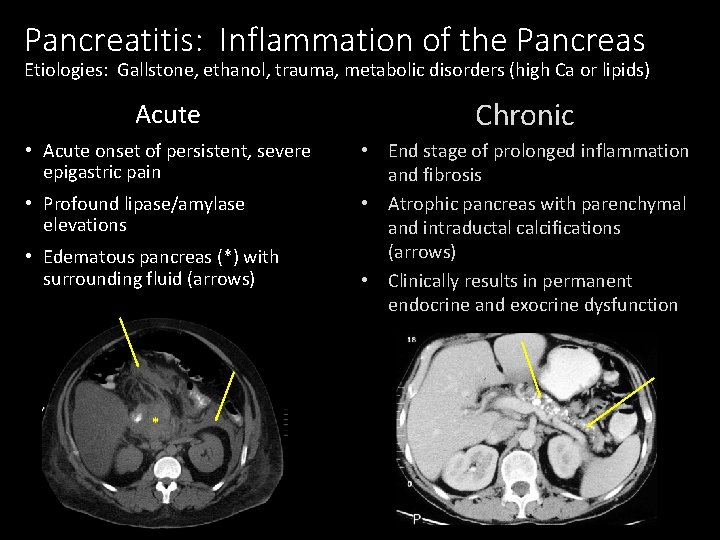
Pancreatitis: Inflammation of the Pancreas Etiologies: Gallstone, ethanol, trauma, metabolic disorders (high Ca or lipids) Acute Chronic • Acute onset of persistent, severe epigastric pain • End stage of prolonged inflammation and fibrosis • Atrophic pancreas with parenchymal and intraductal calcifications (arrows) • Clinically results in permanent endocrine and exocrine dysfunction • Profound lipase/amylase elevations • Edematous pancreas (*) with surrounding fluid (arrows) *
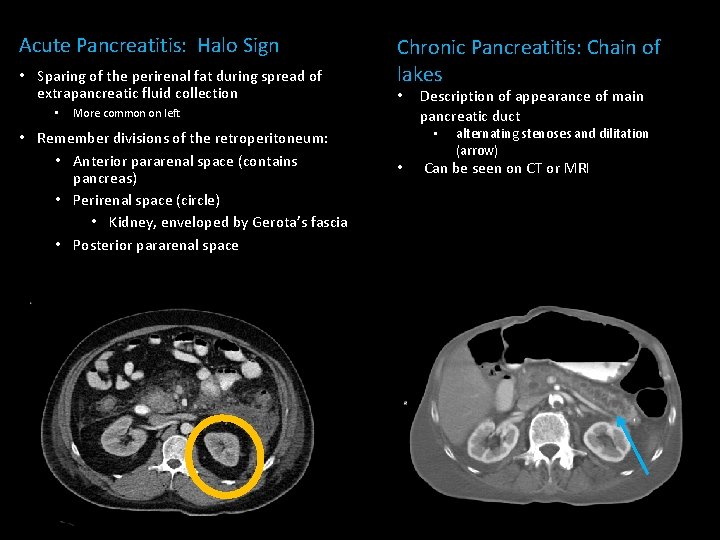
Acute Pancreatitis: Halo Sign • Sparing of the perirenal fat during spread of extrapancreatic fluid collection • Chronic Pancreatitis: Chain of lakes • More common on left • Remember divisions of the retroperitoneum: • Anterior pararenal space (contains pancreas) • Perirenal space (circle) • Kidney, enveloped by Gerota’s fascia • Posterior pararenal space Description of appearance of main pancreatic duct • • alternating stenoses and dilitation (arrow) Can be seen on CT or MRI

Pancreatic Lesion Characterization Now that we have some of the basics, it is time to characterize the lesion. – Solid or cystic? – Ductal dilation? – Involvement of adjacent vessels/structures? – Extra-pancreatic metastases?

Cyst or Cystic lesion • Simple cyst T 2 hyperintense (MRI), water density (CT) • Septations or mural nodularity (solid component that may enhance) make it cystic and not a simple cyst Solid lesion • Less bright than water on T 2. • Post contrast enhancement on CT and MRI Pearl: An initially solid lesion like pancreatic carcinoma can start as a solid lesion and undergo necrosis making it look cystic

Solid Pancreatic Masses Adenocarcinoma Neuroendocrine (Islet Cell) • Highly lethal, with average survival 5 -8 mo. • Metastases often go to liver, lungs, nodes, and peritoneal cavity. • Imaging evaluation of resectability is critical • Can be functional (hormoneproducing) or nonfunctional • Functional tumors vary in their malignant potential from high (80% for glucagonoma) to low (10% insulinoma) • Nonfunctional are larger and frequently (80%) malignant. – Surgery is only treatment, but associated with high morbidity and modest survival benefit
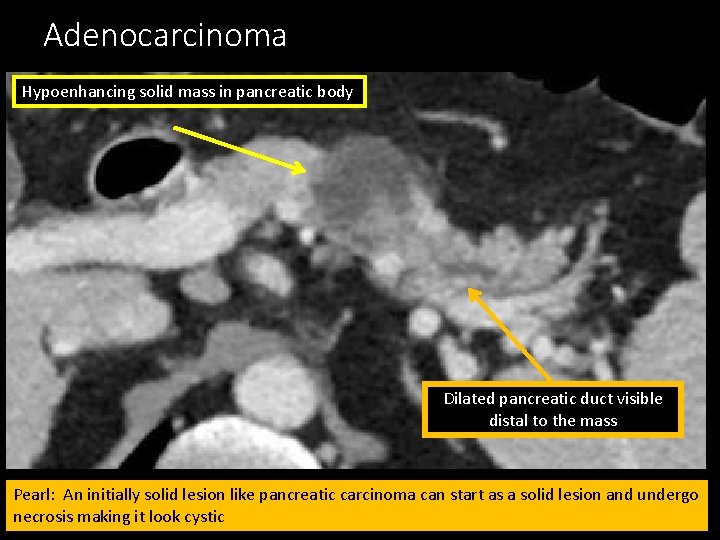
Adenocarcinoma Hypoenhancing solid mass in pancreatic body Dilated pancreatic duct visible distal to the mass Pearl: An initially solid lesion like pancreatic carcinoma can start as a solid lesion and undergo necrosis making it look cystic
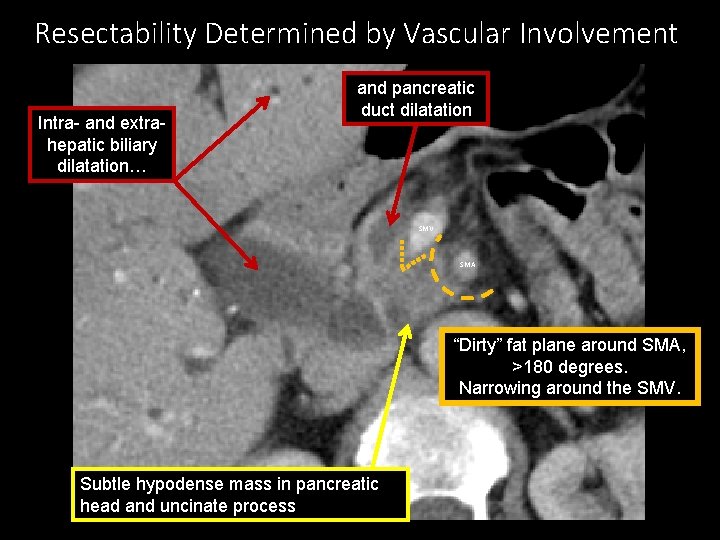
Resectability Determined by Vascular Involvement Intra- and extrahepatic biliary dilatation… and pancreatic duct dilatation SMV SMA “Dirty” fat plane around SMA, >180 degrees. Narrowing around the SMV. Subtle hypodense mass in pancreatic head and uncinate process

Resectability Criteria • Tricky cases hinge on determining degrees of tumor-vessel interface (TVI) Vessel Resectable Borderline Nonresectable SMA Not involved < 180 o ≥ 180 o Celiac Trunk Not involved < 180 o ≥ 180 o Portal system < 180 o ≥ 180 o Unreconstructable occlusion CHA Not involved Reconstructable Unreconstructable occlusion • A reconstructable vessel is one with normal vessel proximal and distal to involvement, long enough to reconnect after resection Pearl: Distant metastases also preclude resection.
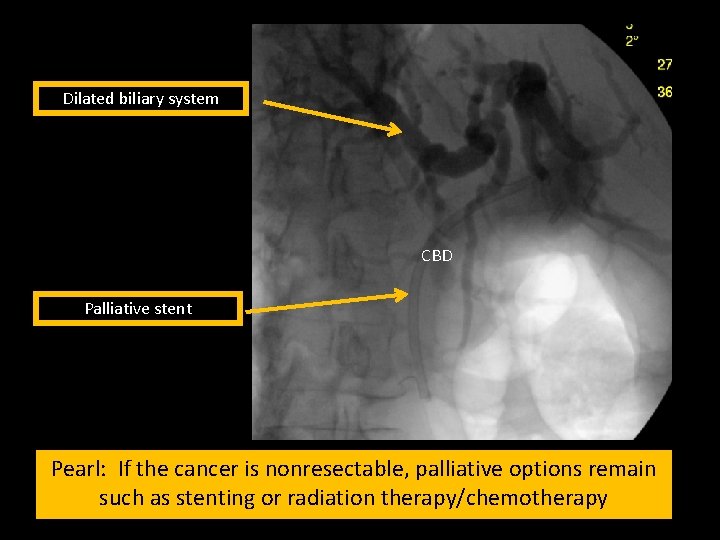
Dilated biliary system CBD Palliative stent Pearl: If the cancer is nonresectable, palliative options remain such as stenting or radiation therapy/chemotherapy
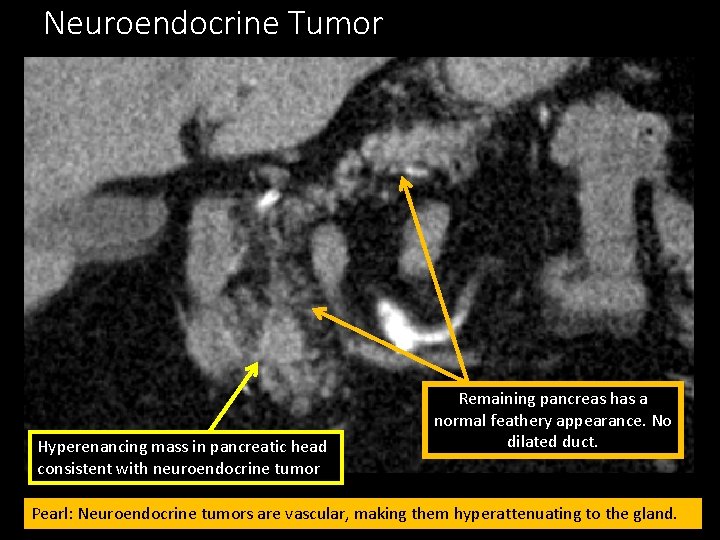
Neuroendocrine Tumor Hyperenancing mass in pancreatic head consistent with neuroendocrine tumor Remaining pancreas has a normal feathery appearance. No dilated duct. Pearl: Neuroendocrine tumors are vascular, making them hyperattenuating to the gland.
Cystic Lesion Characterization • The soft tissue contrast provided by MR imaging has made it the modality of choice for evaluating these lesions and their discerning characteristics: • • Type of fluid Cyst morphology and size Hemorrhage Septation/scar Soft-tissue components/nodularity Communication with pancreatic duct Surrounding pancreatic parenchyma • Finally, patient demographics and clinical history are invaluable to further fine tune the differential diagnosis
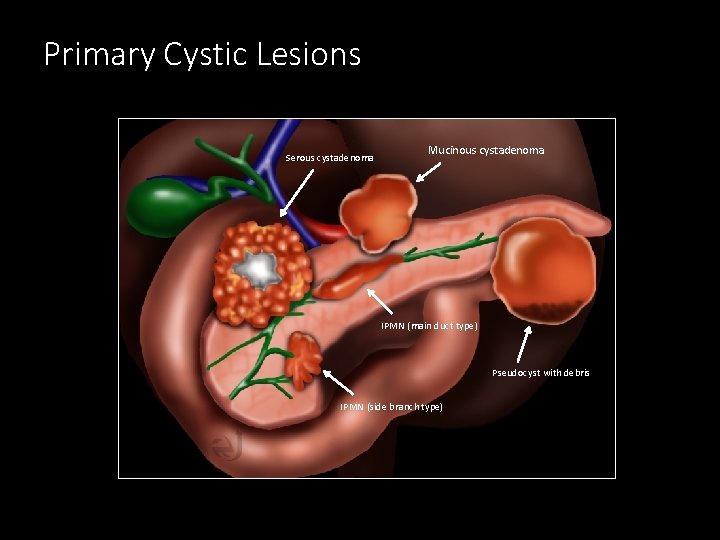
Primary Cystic Lesions Serous cystadenoma Mucinous cystadenoma IPMN (main duct type) Pseudocyst with debris IPMN (side branch type)

Pancreatic Cystic Lesions Pathological Classification of Cystic Lesions • Injury /inflammatory related cysts • Pseudocysts • Neoplastic cysts • Ductal lineage • Serous cystadenoma • Mucinous cystadenomas/carcinomas • Intraductal papillary mucinous neoplasms • Ductal adenocarcinoma with cystic features • Endocrine lineage • Cystic neuroendocrine tumor Pearl: Pancreatic pseudocysts related to pancreatitis are very common (see slides on pancreatitis)

Pancreatic Pseudocysts Background • Most common, >75% of all pancreatic cystic lesions • Occur in the setting of pancreatitis • A result of hemorrhagic fat necrosis with encapsulation of the pancreatic secretions by granulation tissue and a fibrous capsule • No epithelial lining Management • 4 -6 week Follow-up • Can spontaneously resolve • Should evolve over short intervals, whereas other types of cysts often persist without significant change • Complications: hemorrhage, infection, rupture or masseffect on adjacent organs • If complicated or symptomatic may require surgical, percutaneous or endoscopic drainage
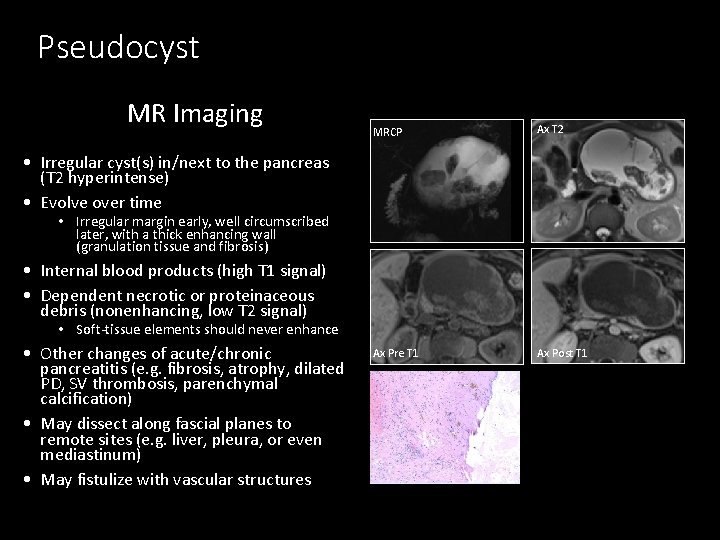
Pseudocyst MR Imaging MRCP Ax T 2 Ax Pre T 1 Ax Post T 1 • Irregular cyst(s) in/next to the pancreas (T 2 hyperintense) • Evolve over time • Irregular margin early, well circumscribed later, with a thick enhancing wall (granulation tissue and fibrosis) • Internal blood products (high T 1 signal) • Dependent necrotic or proteinaceous debris (nonenhancing, low T 2 signal) • Soft-tissue elements should never enhance • Other changes of acute/chronic pancreatitis (e. g. fibrosis, atrophy, dilated PD, SV thrombosis, parenchymal calcification) • May dissect along fascial planes to remote sites (e. g. liver, pleura, or even mediastinum) • May fistulize with vascular structures

Pancreatic cystic neoplasms Next group of slides will show a barrage of cystic pancreatic neoplasms. Don’t sweat the details. Look at the radiology and pathology images and get a sense of why this is a cystic lesion and some of the unique imaging characteristics.
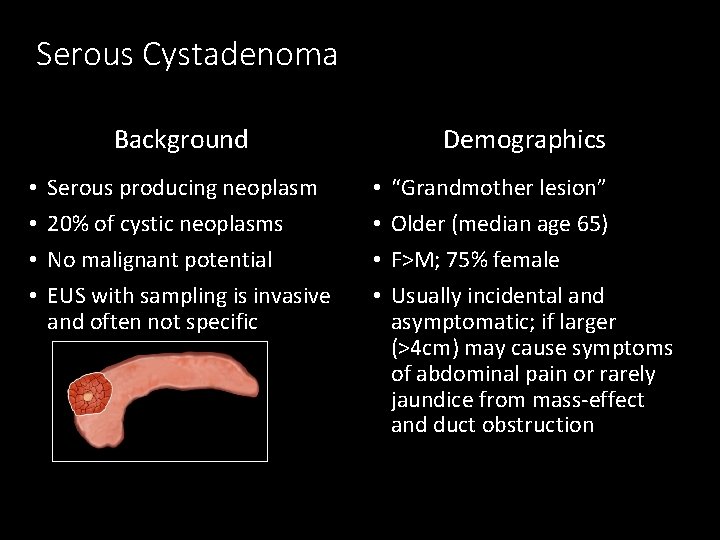
Serous Cystadenoma Background • • Serous producing neoplasm 20% of cystic neoplasms No malignant potential EUS with sampling is invasive and often not specific Demographics • • “Grandmother lesion” Older (median age 65) F>M; 75% female Usually incidental and asymptomatic; if larger (>4 cm) may cause symptoms of abdominal pain or rarely jaundice from mass-effect and duct obstruction
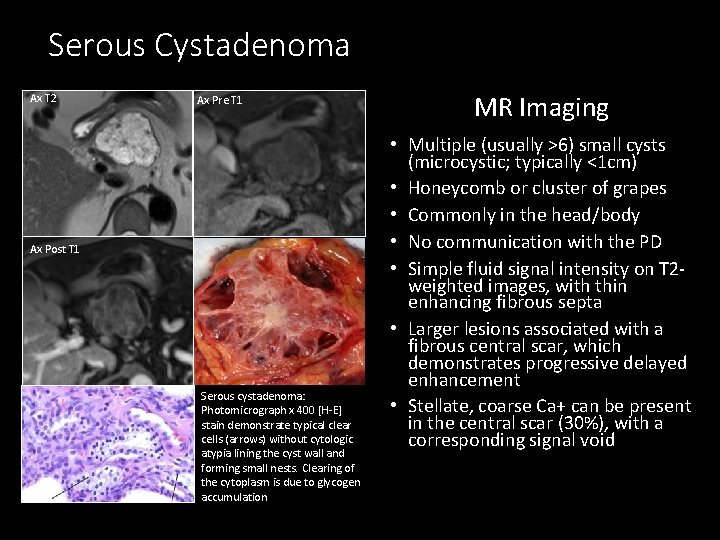
Serous Cystadenoma Ax T 2 Ax Pre T 1 Ax Post T 1 Serous cystadenoma: Photomicrograph x 400 [H-E] stain demonstrate typical clear cells (arrows) without cytologic atypia lining the cyst wall and forming small nests. Clearing of the cytoplasm is due to glycogen accumulation MR Imaging • Multiple (usually >6) small cysts (microcystic; typically <1 cm) • Honeycomb or cluster of grapes • Commonly in the head/body • No communication with the PD • Simple fluid signal intensity on T 2 weighted images, with thin enhancing fibrous septa • Larger lesions associated with a fibrous central scar, which demonstrates progressive delayed enhancement • Stellate, coarse Ca+ can be present in the central scar (30%), with a corresponding signal void
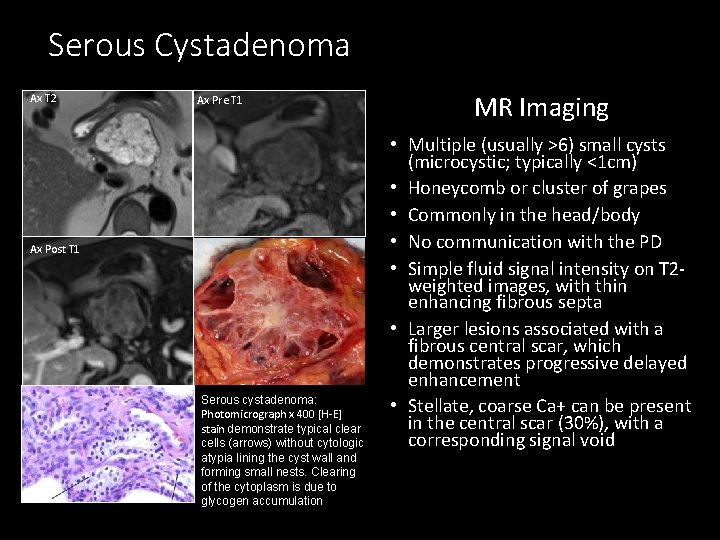
Serous Cystadenoma Ax T 2 Ax Pre T 1 Ax Post T 1 Serous cystadenoma: Photomicrograph x 400 [H-E] stain demonstrate typical clear cells (arrows) without cytologic atypia lining the cyst wall and forming small nests. Clearing of the cytoplasm is due to glycogen accumulation MR Imaging • Multiple (usually >6) small cysts (microcystic; typically <1 cm) • Honeycomb or cluster of grapes • Commonly in the head/body • No communication with the PD • Simple fluid signal intensity on T 2 weighted images, with thin enhancing fibrous septa • Larger lesions associated with a fibrous central scar, which demonstrates progressive delayed enhancement • Stellate, coarse Ca+ can be present in the central scar (30%), with a corresponding signal void

Mucinous Cystadenoma Background Demographics • 10 -40% of cystic neoplasms • “Mother Lesion” • Mucin-producing neoplasm lined with thick columnar epithelium, surrounded by ovarian-type stroma • Mean Age 40 -50 • >95% are female • Nonspecific symptoms when large; 20 -30% are • All have malignant potential asymptomatic and incidentally • Spectrum of low, moderate, discovered or high-grade dysplasia, +/features of invasive carcinoma • Surgical resection recommended
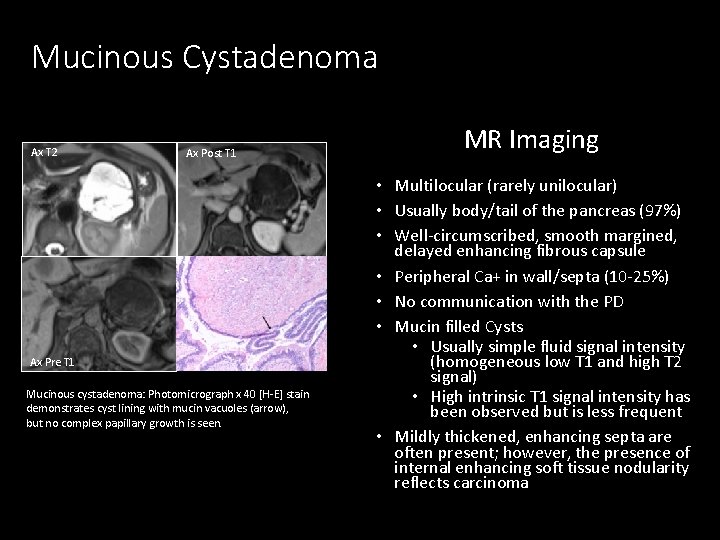
Mucinous Cystadenoma Ax T 2 Ax Post T 1 Ax Pre T 1 Mucinous cystadenoma: Photomicrograph x 40 [H-E] stain demonstrates cyst lining with mucin vacuoles (arrow), but no complex papillary growth is seen. MR Imaging • Multilocular (rarely unilocular) • Usually body/tail of the pancreas (97%) • Well-circumscribed, smooth margined, delayed enhancing fibrous capsule • Peripheral Ca+ in wall/septa (10 -25%) • No communication with the PD • Mucin filled Cysts • Usually simple fluid signal intensity (homogeneous low T 1 and high T 2 signal) • High intrinsic T 1 signal intensity has been observed but is less frequent • Mildly thickened, enhancing septa are often present; however, the presence of internal enhancing soft tissue nodularity reflects carcinoma
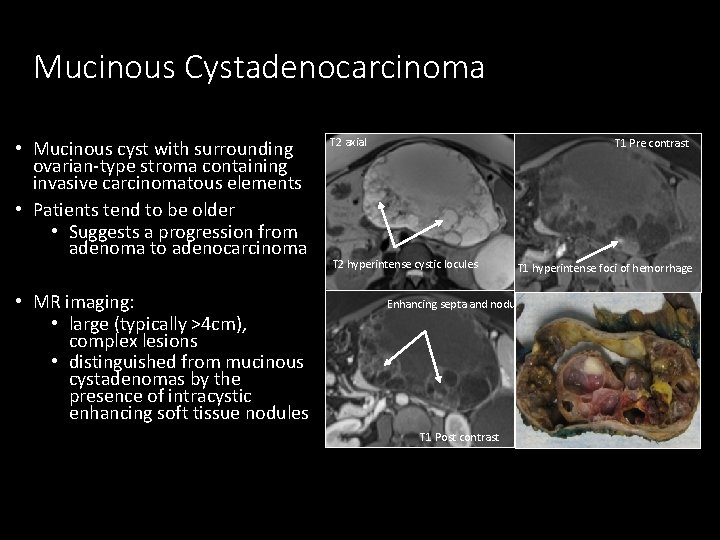
Mucinous Cystadenocarcinoma • Mucinous cyst with surrounding ovarian-type stroma containing invasive carcinomatous elements • Patients tend to be older • Suggests a progression from adenoma to adenocarcinoma • MR imaging: • large (typically >4 cm), complex lesions • distinguished from mucinous cystadenomas by the presence of intracystic enhancing soft tissue nodules T 2 axial T 1 Pre contrast T 2 hyperintense cystic locules T 1 hyperintense foci of hemorrhage Enhancing septa and nodularity T 1 Post contrast
Intraductal Papillary Mucinous Neoplasms Background Demographics • Metaplasia of mucin producing columnar epithelium within pancreatic ducts, commonly with papillary projections • All have malignant potential • “Grandfather Lesion” • Spectrum of noninvasive neoplasms with varying degrees of dysplasia to invasive carcinoma • 20% of cystic pancreatic neoplasms • ERCP demonstrates spillage of mucin from the ampulla of Vater • Mean Age is 65 • >60% of patients are male • Usually symptomatic • Abdominal pain • Weight loss • Recurrent pancreatitis
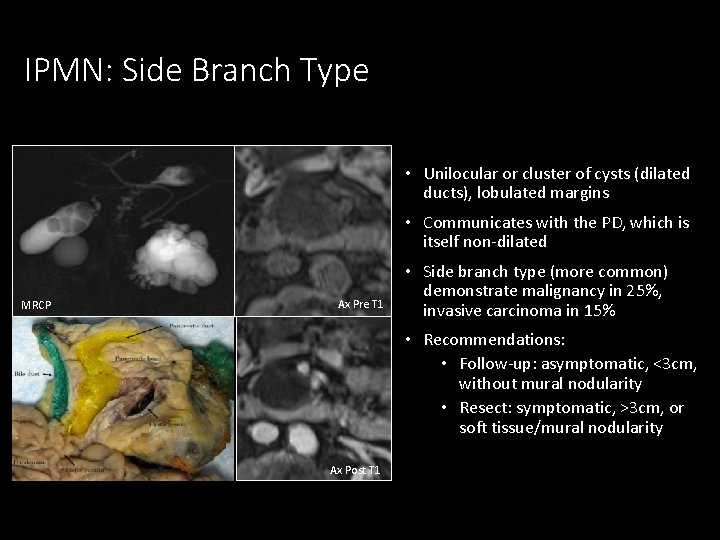
IPMN: Side Branch Type • Unilocular or cluster of cysts (dilated ducts), lobulated margins • Communicates with the PD, which is itself non-dilated MRCP Ax Pre T 1 • Side branch type (more common) demonstrate malignancy in 25%, invasive carcinoma in 15% • Recommendations: • Follow-up: asymptomatic, <3 cm, without mural nodularity • Resect: symptomatic, >3 cm, or soft tissue/mural nodularity Ax Post T 1
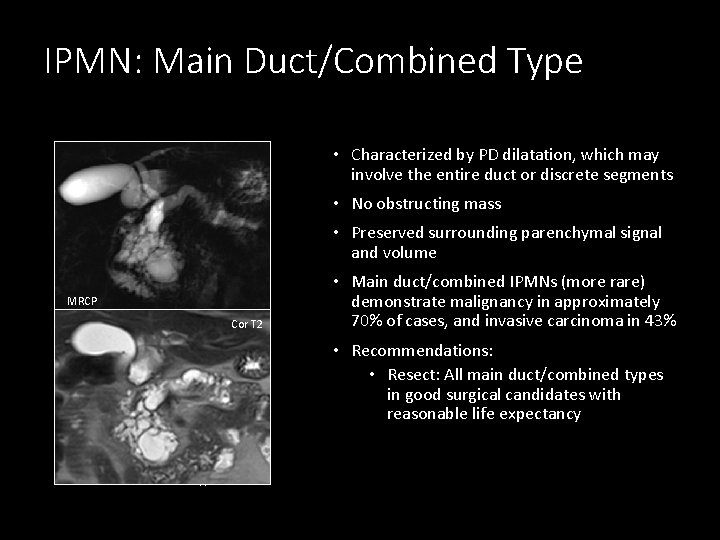
IPMN: Main Duct/Combined Type • Characterized by PD dilatation, which may involve the entire duct or discrete segments • No obstructing mass • Preserved surrounding parenchymal signal and volume MRCP: Side Branch Type Cor T 2 • Main duct/combined IPMNs (more rare) demonstrate malignancy in approximately 70% of cases, and invasive carcinoma in 43% • Recommendations: • Resect: All main duct/combined types in good surgical candidates with reasonable life expectancy MRCP: Combined Type
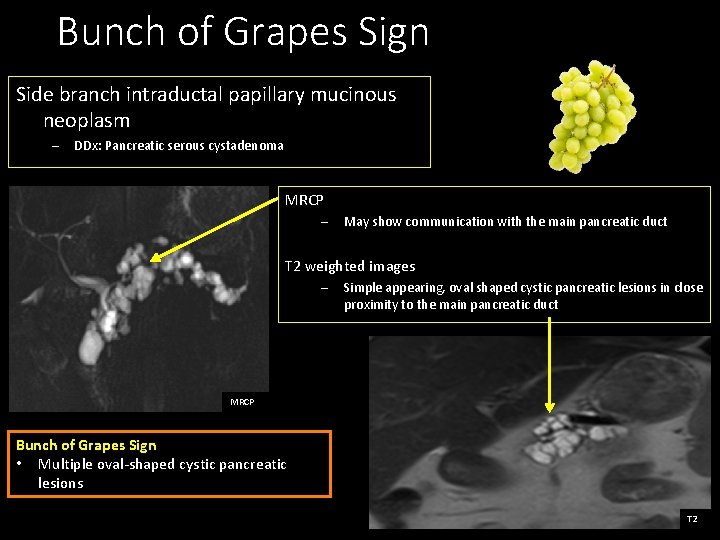
Bunch of Grapes Sign Side branch intraductal papillary mucinous neoplasm – DDx: Pancreatic serous cystadenoma MRCP – May show communication with the main pancreatic duct T 2 weighted images – Simple appearing, oval shaped cystic pancreatic lesions in close proximity to the main pancreatic duct MRCP Bunch of Grapes Sign • Multiple oval-shaped cystic pancreatic lesions T 2
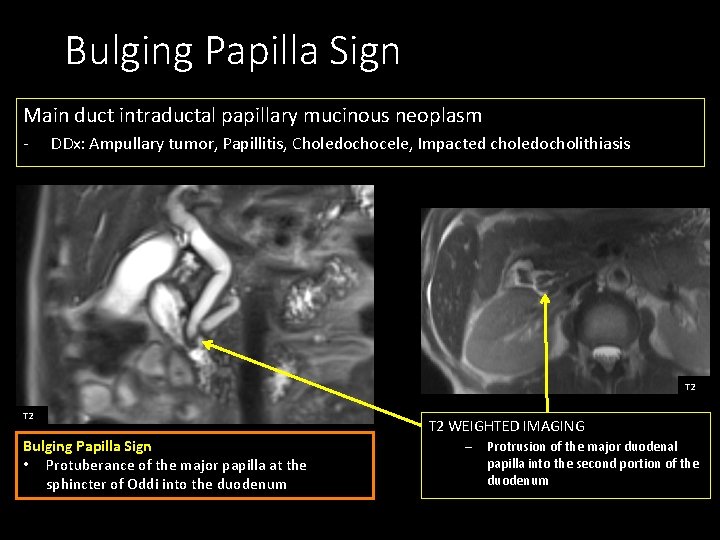
Bulging Papilla Sign Main duct intraductal papillary mucinous neoplasm - DDx: Ampullary tumor, Papillitis, Choledochocele, Impacted choledocholithiasis T 2 Bulging Papilla Sign • Protuberance of the major papilla at the sphincter of Oddi into the duodenum T 2 WEIGHTED IMAGING – Protrusion of the major duodenal papilla into the second portion of the duodenum
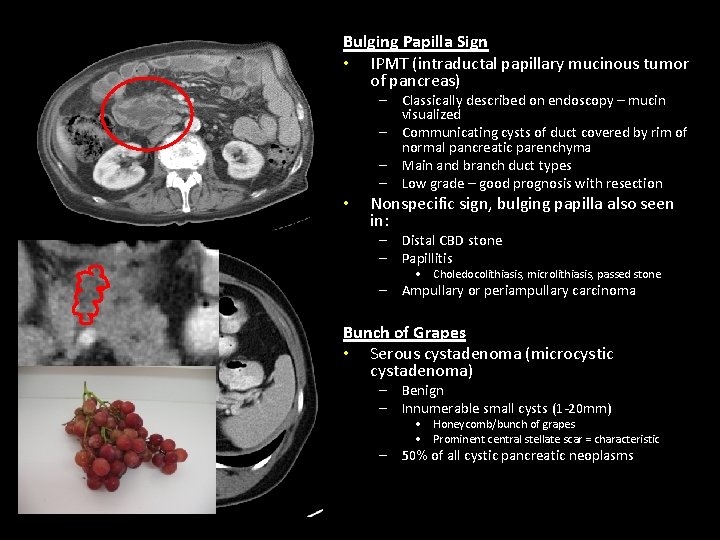
Bulging Papilla Sign • IPMT (intraductal papillary mucinous tumor of pancreas) • – Classically described on endoscopy – mucin visualized – Communicating cysts of duct covered by rim of normal pancreatic parenchyma – Main and branch duct types – Low grade – good prognosis with resection Nonspecific sign, bulging papilla also seen in: – Distal CBD stone – Papillitis • Choledocolithiasis, microlithiasis, passed stone – Ampullary or periampullary carcinoma Bunch of Grapes • Serous cystadenoma (microcystic cystadenoma) – Benign – Innumerable small cysts (1 -20 mm) • Honeycomb/bunch of grapes • Prominent central stellate scar = characteristic – 50% of all cystic pancreatic neoplasms

Conclusions • Pancreatic pathology can be divided into solid and cystic lesions • Pancreatic adenocarcinoma is the most common solid lesion and has a poor prognosis. CT and MRI evaluation are imperative for surgical planning • Cystic lesions will be T 2 hyperintense • MR Imaging is diagnostic solving tool for detection and characterization of a wide range of pancreatic cystic lesions because of its unsurpassed soft tissue contrast and its ability to evaluate pancreatic duct • Pseudocysts related to pancreatitis are the most common cystic lesion/collection • Cystic lesions such as serous cystadenoma, IPMN and mucinous cystadenoma/carcinoma can be characterized with MRI

References • Bordeianou L, Vagefi PA, Sahani D, et al. Cystic pancreatic endocrine neoplasms: a distinct tumor type? J Am Coll Surg 2008; 206: 1154– 1158. • Figueiras R, Martin C, et al. The Spectrum of Cystic Masses of the Pancreas: Imaging Features and Diagnostic Difficulties. Current Problems in Diag Radio 2007; 36: 199 -212 • Kalb B, Sarmiento JM, Kooby DA et-al. MR imaging of cystic lesions of the pancreas. Radiographics. 2009; 29 (6): 1749 -65 • Kim YH, Saini S, Sahani D, Hahn PF, Mueller PR, Auh YH. Imaging diagnosis of cystic pancreatic lesions: pseudocyst versus nonpseudocyst. Radio. Graphics 2005; 25: 671– 685 • Lewis R. Cystic pancreatic tumors. AIRP 2011 -2012; Vol 1: 211 -217 • Macari M, Megibow AJ. Focal Cystic Pancreatic Lesions: Variability in Radiologists' Recommendations for Follow-up Imaging. Radiology. 2011; 259 (1): 20 -3 • Sahani DV, Kadavigere R, Saokar A, Fernandez-del Castillo C, Brugge WR, Hahn PF. Cystic pancreatic lesions: a simple imagingbased classification system for guiding management. Radio. Graphics 2005; 25: 1471– 1484 • Tanaka M , Chari S , Adsay V , et al. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas. Pancreatology 2006; 6 ( 1 -2 ): 17 – 32 • Turkvatan A, Erden A, Mehmat AT, Yener O. Congenital Variants and Anomalies of the Pancreas and Pancreatic Duct: Imaging by MRCP and Multidetector CT. Korean Journal of Radiology 2013; 14(6): 905 -913

END
- Slides: 39