Radioisotopes in Medicine Introduction Stable subatomic particles in

Radioisotopes in Medicine • Introduction • Stable subatomic particles in an atom are proton, neutron and electron. • Isotopes are the same element with the same atomic number (protons) but different mass number (varying number of neutrons)

: Radioactivity • Isotopes may be stable or unstable (radioactive). • The nucleus of a radioactive isotope degrades spontaneously with consequent emission of rays or particles and gets transformed to another element. Such a process is called radioactivity and the degradation is called radioactive decay. • The radioactive elements emit 3 types of radiation – 1) alpha (α) 2) beta (β) and 3) gamma (γ).

Applications of radioisotopes in medicine • Are three fold • 1) research • 2) clinical diagnosis and • 3) treatment.

1) Research applications • tracer technique • Almost all the metabolic pathways are discovered by using tracers. For e. g. 14 Clabeled acetoacetic acid can be shown to be incorporated into palmitic acid. • Other uses are determination of turnover rate, pool size of a substance in the body and volumes of body compartments containing body fluids (e. g. extra-cellular volume).
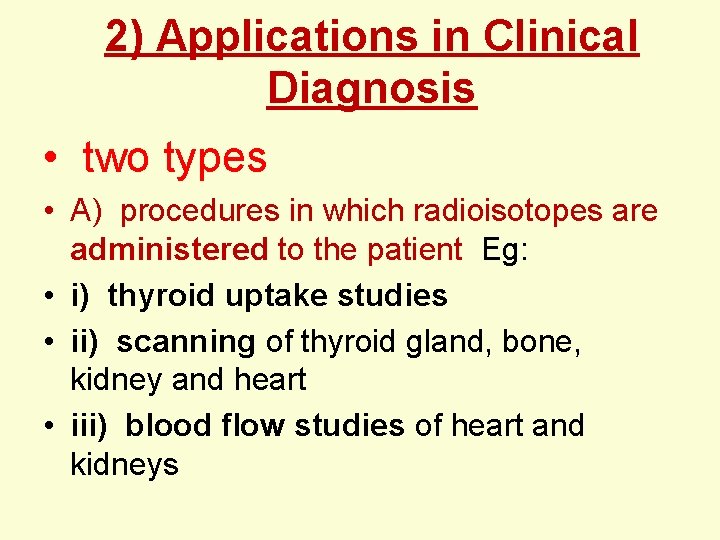
2) Applications in Clinical Diagnosis • two types • A) procedures in which radioisotopes are administered to the patient Eg: • i) thyroid uptake studies • ii) scanning of thyroid gland, bone, kidney and heart • iii) blood flow studies of heart and kidneys

• B) As reagents, radioisotopes are used • i) to quantitate hormones, tumor markers and other biological substances present in blood in very small quantities [e. g. radioimmunoassay (RIA)] • ii) in DNA analysis techniques e. g. Southern blot technque.

3) Applications in Treatment • i) Radioactivity is used mainly in the treatment of cancer. • Radiotherapy mainly affects cells in the division phase. Cancer tissue is preferentially affected by radiation, it contains more dividing cell than normal tissue. • ii)Radioactivity is also used for treatment of hyperthyroidism.
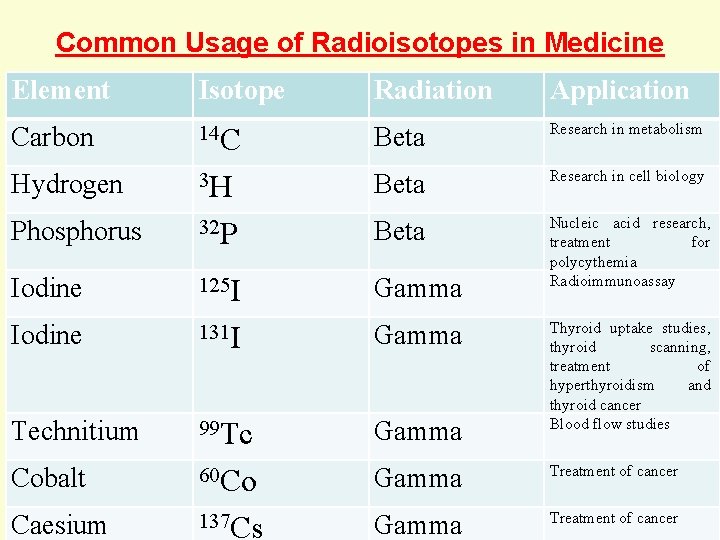
Common Usage of Radioisotopes in Medicine Radiation Element Isotope Application Carbon 14 C Beta Research in metabolism Hydrogen 3 H Beta Research in cell biology Phosphorus 32 P Beta Iodine 125 I Gamma Nucleic acid research, treatment for polycythemia Radioimmunoassay Iodine 131 I Gamma Technitium 99 Tc Gamma Cobalt 60 Co Gamma Treatment of cancer Caesium 137 Cs Gamma Treatment of cancer Thyroid uptake studies, thyroid scanning, treatment of hyperthyroidism and thyroid cancer Blood flow studies

• Questions on radioisotopes in medicine • 1. Define radioactivity? Name the uses of a) 131 I b) 60 Co. (3) • 2. Mention the clinical applications of Radioactive Isotopes. (3) • 3. Mention two isotopes and mention their application in medicine (3) •

Radioimmunoassay (RIA) and Enzyme-Linked Immunosorbant Assay (ELISA) HP Kedilaya

Radioimmunoassay (RIA) • A Clinical Biochemical Assay • for Determination of Concentration of Antigenic Substances in Body Fluids • by Antigen-Antibody Reaction • Using an Appropriate Antibody and Radio. Labeled Antigen.

Radioimmunoassay (RIA) • Thus, Combines the Advantages of • Specificity -- of Antigen-Antibody Interaction and • Sensitivity -- of Measurement of Radio-labeled Compounds.

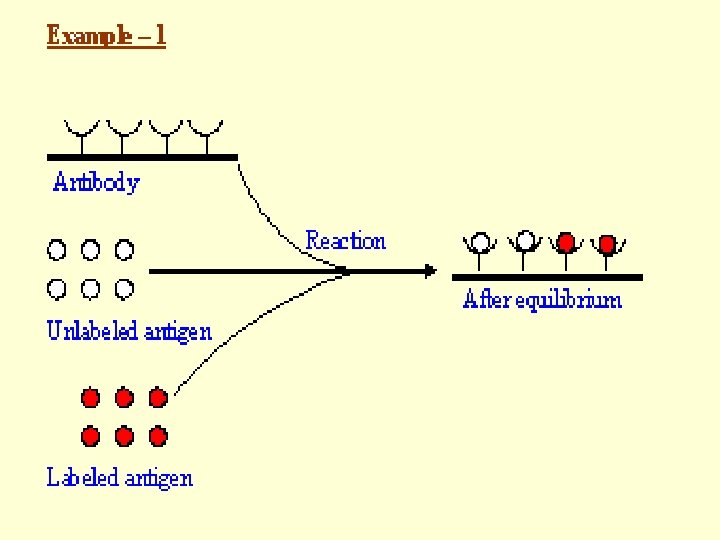
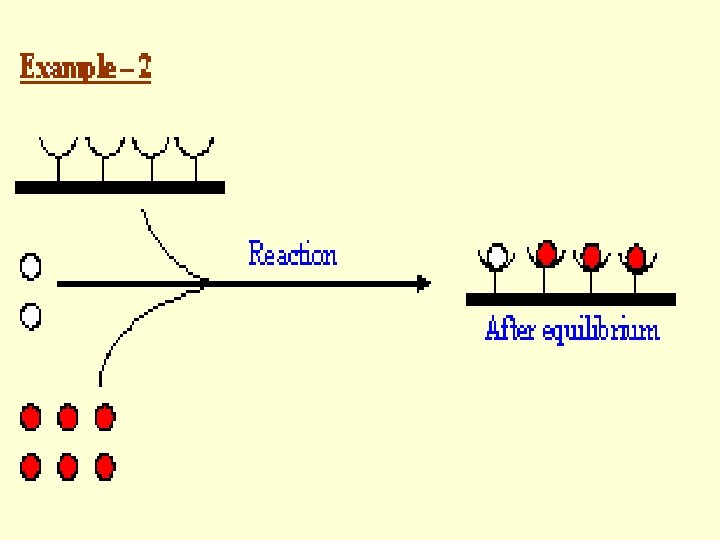
Principle • Based on Competitive Binding of Radio-labeled Antigen and Unlabeled Antigen (Present in the Sample to be Assayed) to Antibody, • Where, Radio-labeled Antigen -- Excess but Fixed Quantity Antibody -- Limited Quantity Unlabeled Antigen (Present in the Sample to be Assayed) -- Unknown Quantity

Principle • Frequently Used Radioisotopes in Radioimmunoassays for Labeling 125 I and 131 I
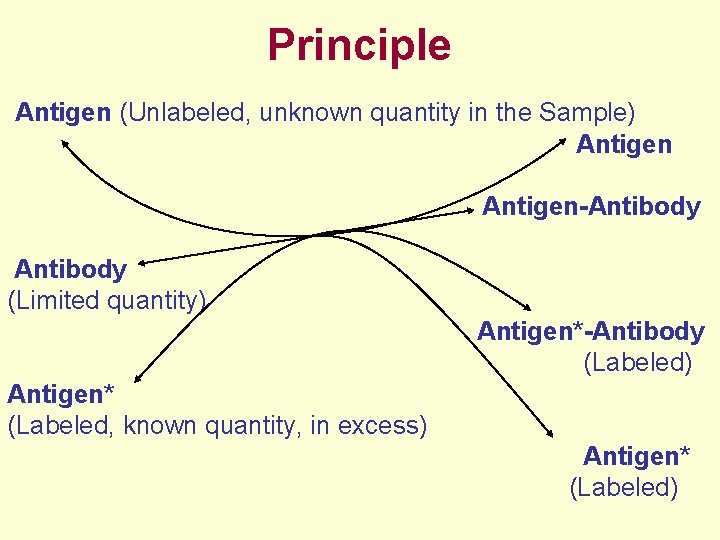
Principle Antigen (Unlabeled, unknown quantity in the Sample) Antigen Antigen-Antibody (Limited quantity) Antigen*-Antibody (Labeled) Antigen* (Labeled, known quantity, in excess) Antigen* (Labeled)

Principle • After Reaching Equilibrium in the Antigenantibody Reaction, Antibody-bound Antigen is Separated from Free Antigen (Labeled and Unlabeled). • The Amount of Labeled Antigen Present in the Antibody-bound Antigen is Determined by Measuring the Radioactivity. • The Radioactivity Measured Will be Inversely Proportional to the Concentration of Antigen Present in the Sample.

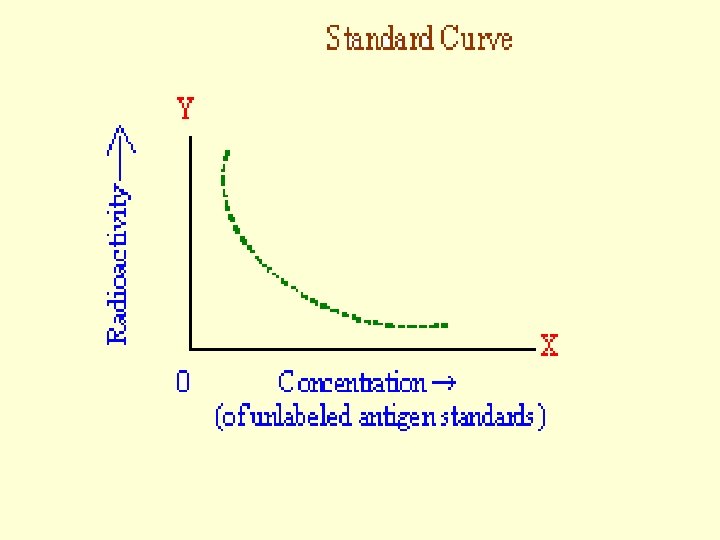
Principle A Standard Curve is Drawn by Using • Different Known Concentrations of Unlabeled Antigen and • Same Quantities of Antibody and Labeled Antigen. This Can be Used, by Comparison, to Derive Antigen Concentrations in Samples.

Applications • RIA is Used in Clinical Diagnosis in the Estimation of Substances that Exhibit Antigenic Property Either as Such or by Chemical Modification. • Substances that are Estimated by RIA are: Hormones, Tumor Markers (Peptides, Proteins, Steroids, etc), Vitamins, Drugs, etc, • Thus, Radioimmunoassay has Applications in the Diagnosis of Hormonal Disorders, Cancers and Therapeutic Monitoring of Drugs. • Also Useful in Biomedical Research.

Enzyme-Linked Immunosorbant Assay (ELISA) • Non-isotopic Immunoassay for both: Detection and Quantitation of Antigens or Antibodies in Body Fluids for Clinical Diagnosis • An Enzyme is Used as a Label in ELISA in Place of Radioactive Isotope Employed in RIA. • As Specific as RIA (Though Less Sensitive) • In Addition, No Risk of Radiation Hazards) in ELISA (as is the Case with RIA)

Principle • Used for both Detection and Quantitation of: Antigens or Antibodies in Body Fluids for Clinical Diagnosis

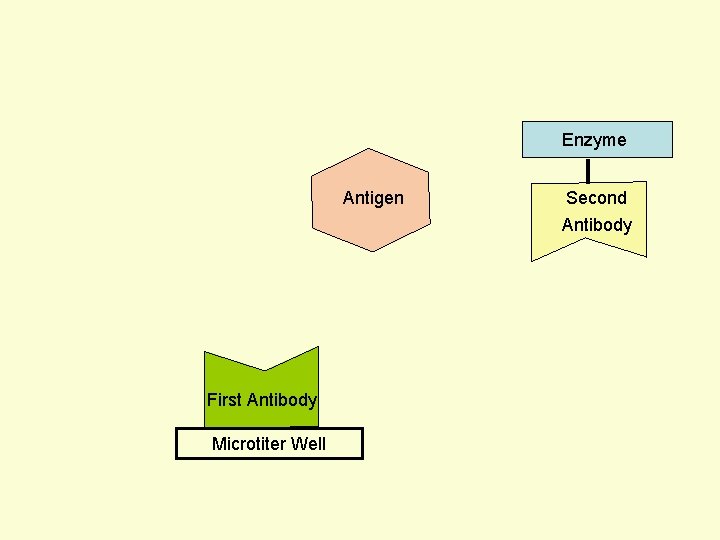
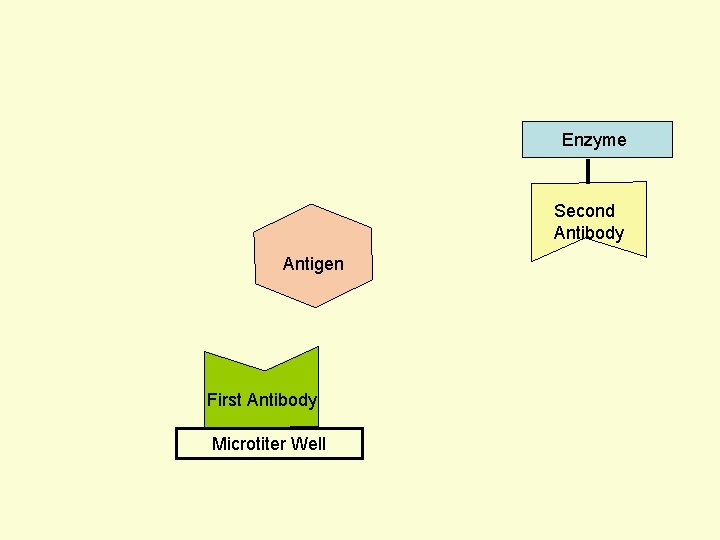
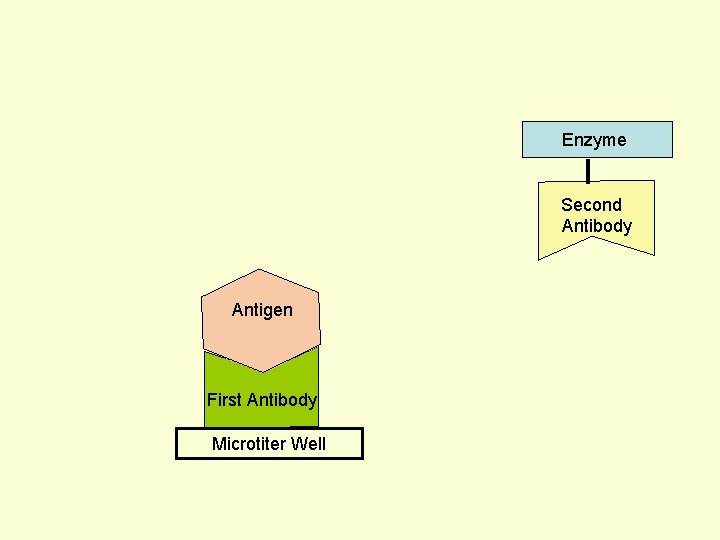
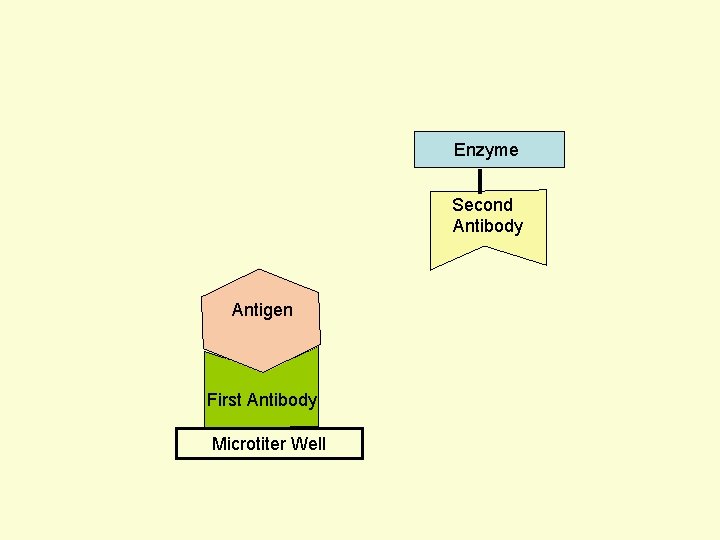
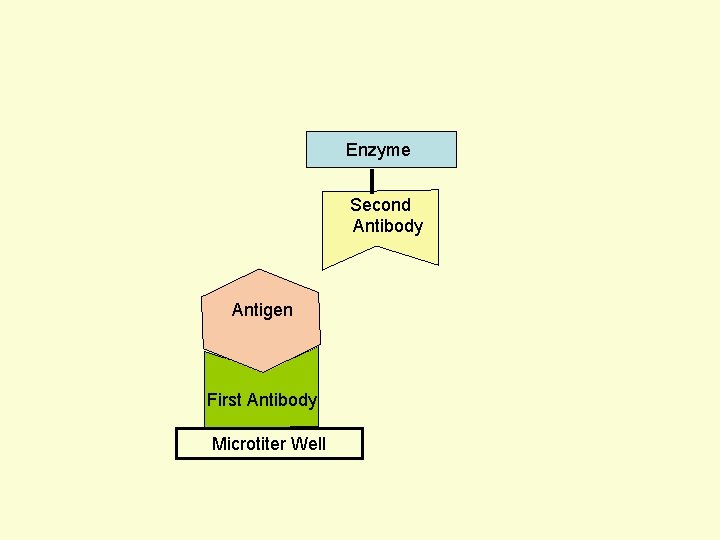
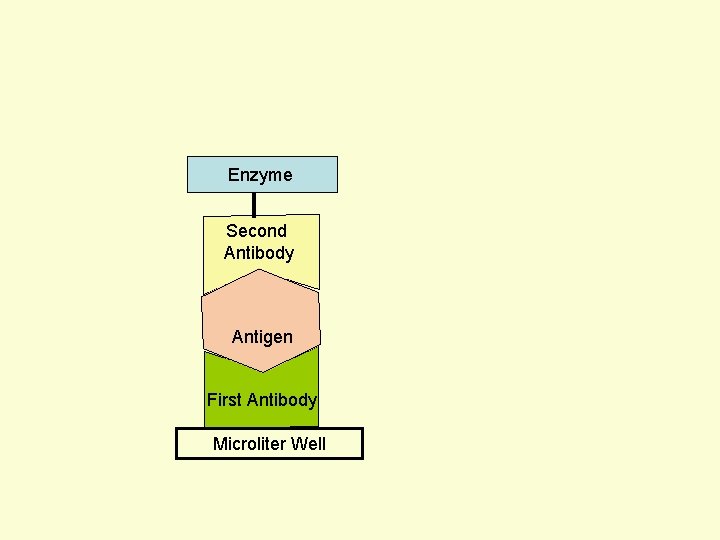
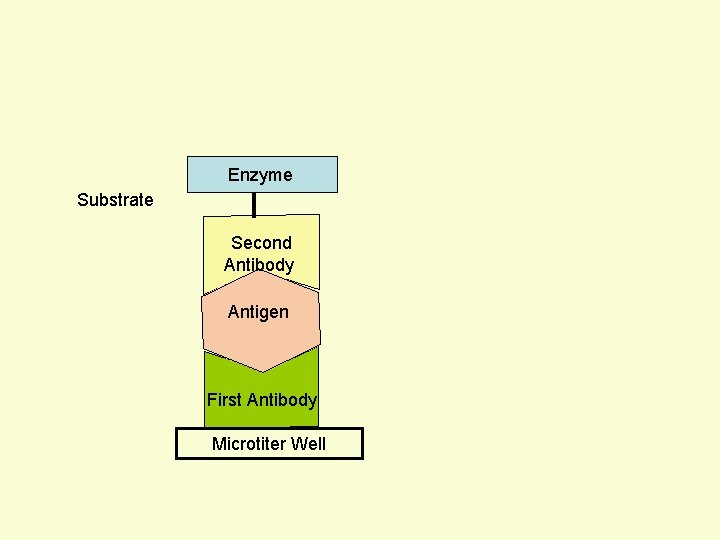
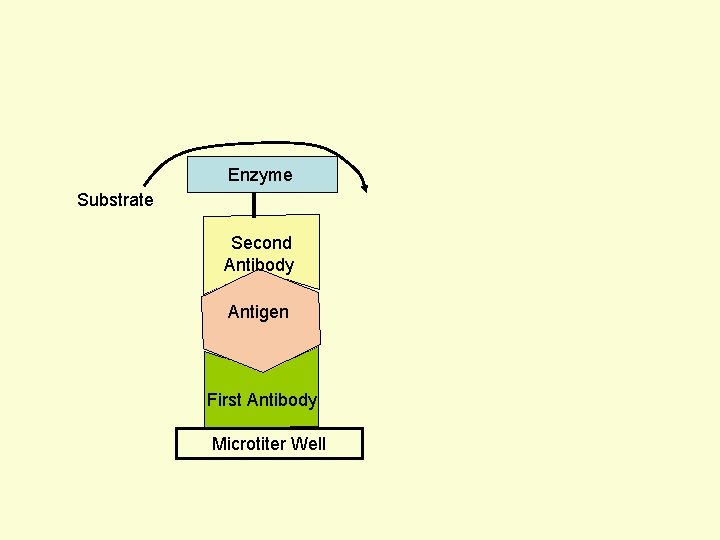
Principle Assay for Antigens • Antibody to the Antigen to be Assayed is Fixed to a Solid Surface (Microtiter Well). • Then Antigen (to be Assayed Present In Serum) Binds to the Antibody. • And then Enzyme-labeled Second Antibody Binds to the Antigen. • Thus Antigen to be Assayed is Sandwiched Between to Antibodies. • The Amount of Enzyme-linked Antibody Getting Fixed to the Well will be Directly Proportional to the Amount of Antigen Present in the Serum. • The Enzyme Selected to Label is Such that it Can Catalyze Formation of a Colored Substance (Chromogen) from a Substrate. • Intensity of Color Developed, Measured Colorimetrically, Will be Directly Proportional to Quantity of the Enzyme-labeled Antibody Fixed to the Well, which in turn is Directly Proportional to Quantity of Antigen Present in the Serum.
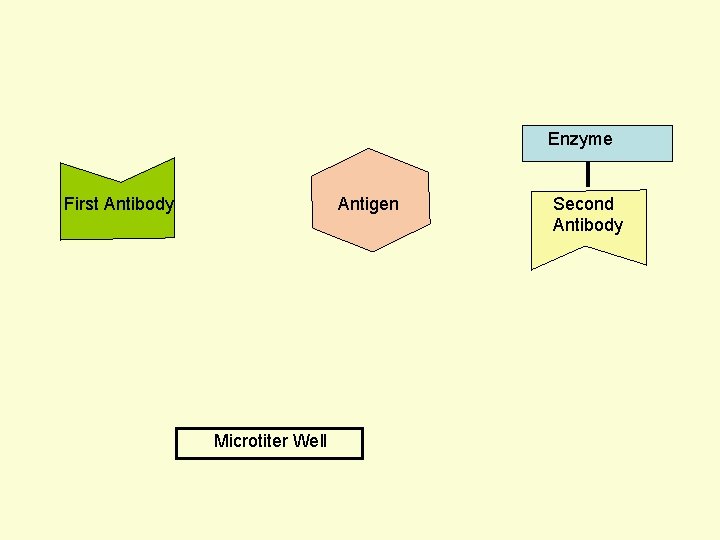
Enzyme First Antibody Antigen Microtiter Well Second Antibody
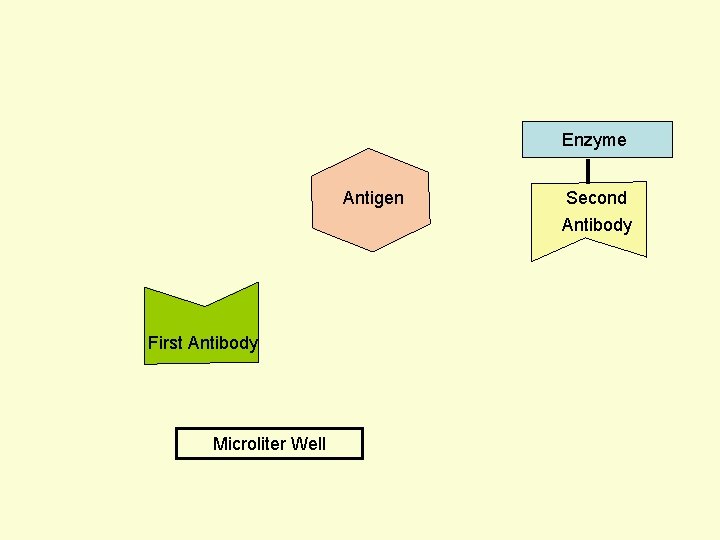
Enzyme Antigen Second Antibody First Antibody Microliter Well
Enzyme Antigen Second Antibody First Antibody Microtiter Well
Enzyme Second Antibody Antigen First Antibody Microtiter Well
Enzyme Second Antibody Antigen First Antibody Microtiter Well
Enzyme Second Antibody Antigen First Antibody Microtiter Well
Enzyme Second Antibody Antigen First Antibody Microtiter Well
Enzyme Second Antibody Antigen First Antibody Microliter Well
Enzyme Substrate Second Antibody Antigen First Antibody Microtiter Well
Enzyme Substrate Second Antibody Antigen First Antibody Microtiter Well
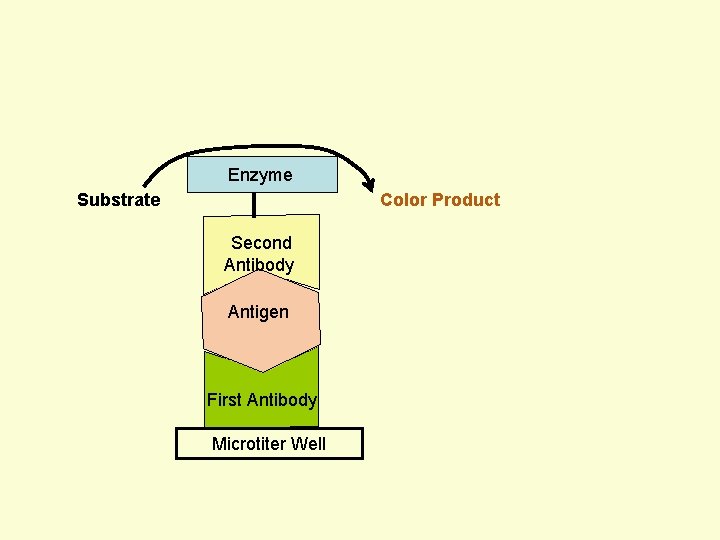
Enzyme Substrate Color Product Second Antibody Antigen First Antibody Microtiter Well

Principle (Cont’d) Assay for Antibody • In this Antibody to be Assayed is Sandwiched between an Antigen and Enzyme-Labeled Antibody to the Antibody to be Assayed.

Applications • In Clinical Practice ELISA is used for Quantitation and Detection of Proteins including: Antigens, Antibodies, Hormones and Tumor Markers. • Examples: Antibodies – e. g. Antibodies Against HIV for Test for AIDS Antigens – e. g. Detection of HIV Antigen Hormones – e. g. Human Chorionic Gonadotropin (h. CG) for Pregnancy Test.

Questions on RIA and ELISA • • RIA ELISA Radioimmunoassay Principles and applications of RIA Principles and applications of ELISA Applications of RIA (5) (5) (5) (3)

- Slides: 39