radioisotopes Chemical bonds Ionic Radioisotopes These are radioactive

radioisotopes Chemical bonds (Ionic)

Radioisotopes These are radioactive isotopes. They have an unstable nucleus and will emit subatomic particles or energy as they decay into more stable atoms. Radioactive emissions include: alpha particles (α) - a helium nucleus - can be stopped by a sheet of paper beta particles (β) - high speed electrons - can be stopped by a sheet of lead - energetic electromagnetic radiation gamma rays (γ) - can be stopped by a brick of lead α- paper sheet What can stop them? β- lead sheet γ- lead brick http: //www. physics. umd. edu/lecdem/services/demosp 4/p 4 -01 a. mpg http: //www. physics. umd. edu/lecdem/services/demosp 4/p 4 -01 c. mpg http: //www. physics. umd. edu/lecdem/services/demosp 4/p 4 -01 f. mpg

Radioisotopes Examples of radioisotopes include: Radioisotope Uses To trace the movement of carbon through biological pathways, like respiration and photosynthesis. To measure the rate of bone formation. Has a half-life of 1. 3 billion years (decays into 40 Ar). The ratio of 40 K : 40 Ar can be used to date fossils. Iodine is taken up by the thyroid gland. The gland can therefore be imaged to detect abnormalities. Emits radiation that can destroy living cells. Can be used to treat cancer tumors. Figure 1: Scientific and medical uses for some radioisotopes.

Chemical Bonds Chemical bonds are formed between atoms by the interaction of their respective electrons. There are three types of intramolecular bonds (bonds that molecule in a that hold aatoms together): molecule Ionic Bonds Covalent Bonds Polar Covalent Bonds
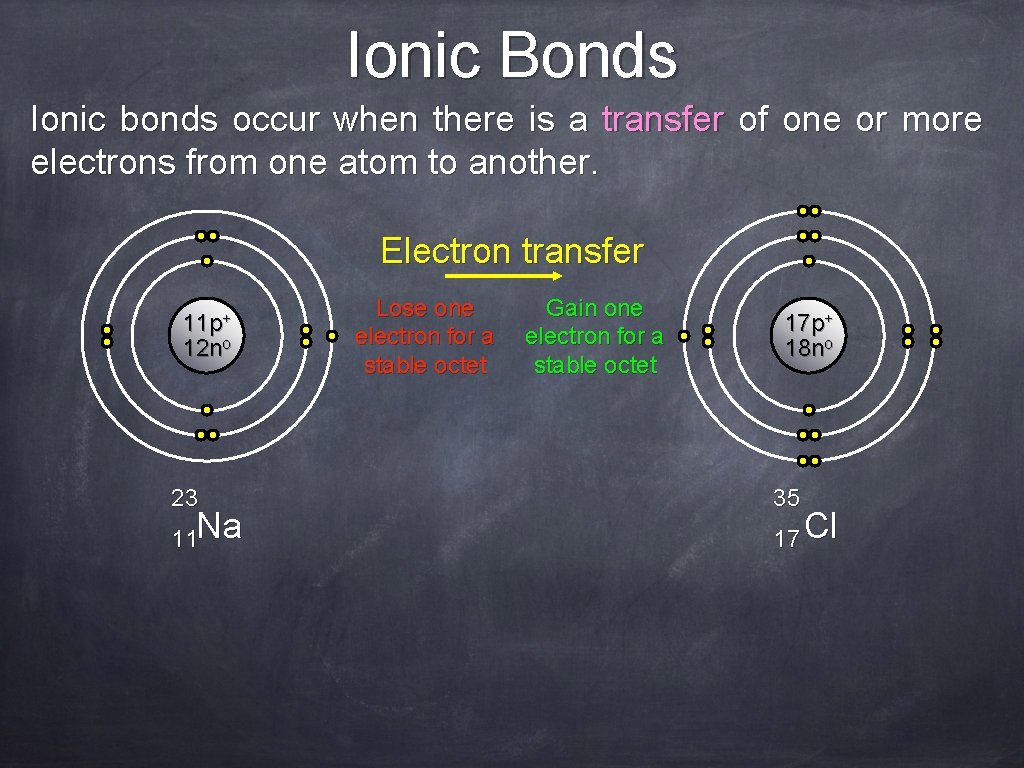
Ionic Bonds Ionic bonds occur when there is a transfer of one or more electrons from one atom to another. Electron transfer 11 p+ 12 no 23 11 Na Lose one electron for a stable octet Gain one electron for a stable octet 17 p+ 18 no 35 17 Cl
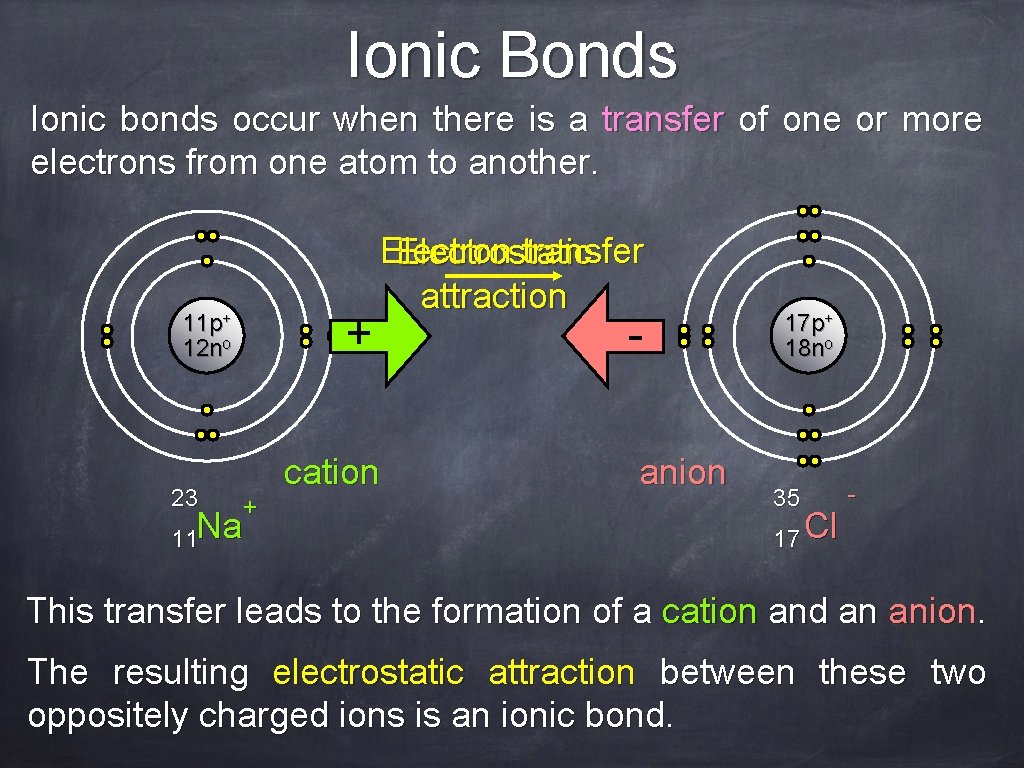
Ionic Bonds Ionic bonds occur when there is a transfer of one or more electrons from one atom to another. + 11 p+ 12 no 23 Electron transfer Electrostatic attraction cation + 11 Na - anion 17 p+ 18 no 35 17 Cl - This transfer leads to the formation of a cation and an anion. The resulting electrostatic attraction between these two oppositely charged ions is an ionic bond.
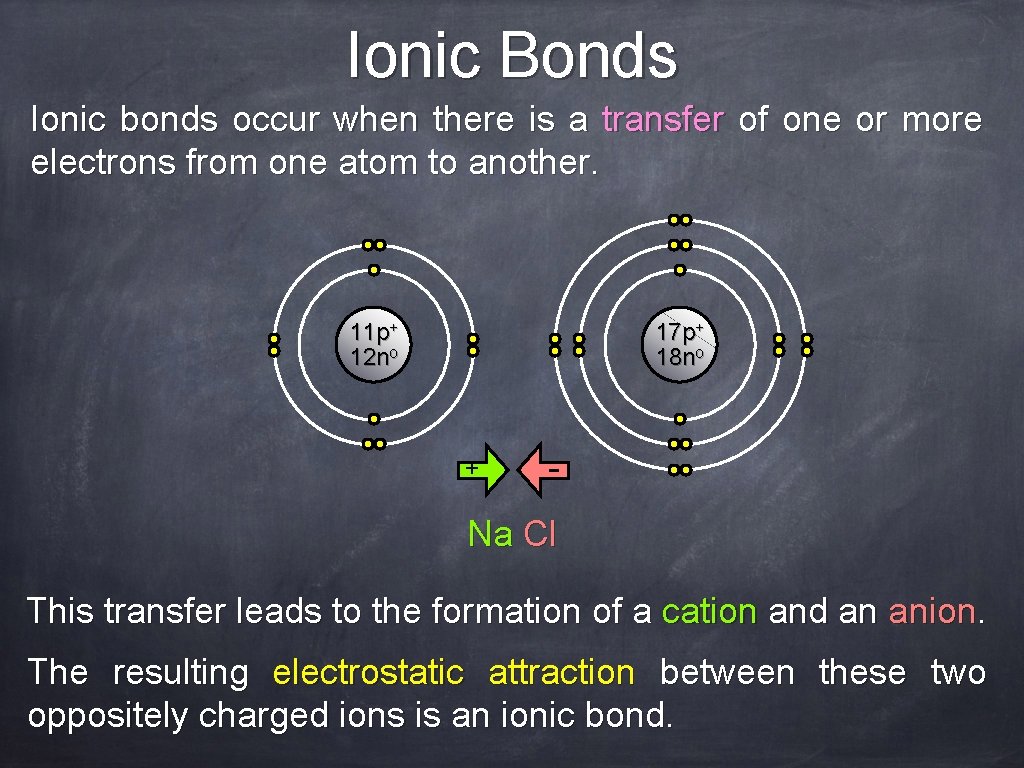
Ionic Bonds Ionic bonds occur when there is a transfer of one or more electrons from one atom to another. 11 p+ 12 no 17 p+ 18 no + - Na Cl This transfer leads to the formation of a cation and an anion. The resulting electrostatic attraction between these two oppositely charged ions is an ionic bond.

Ionic Bonds Ionic bonds occur when there is a transfer of one or more electrons from one atom to another. Let’s see that again (Nelson Biology 12 animation) This transfer leads to the formation of a cation and an anion. The resulting electrostatic attraction between these two oppositely charged ions is an ionic bond.

Study Table 1: Common Ions in Living Systems (booklet) Read pages 10 -12 Do Q 3 2 -3 page 18
- Slides: 9