Radioactivity What you dont see can hurt you















































- Slides: 62

Radioactivity What you don’t see. . . can hurt you!.

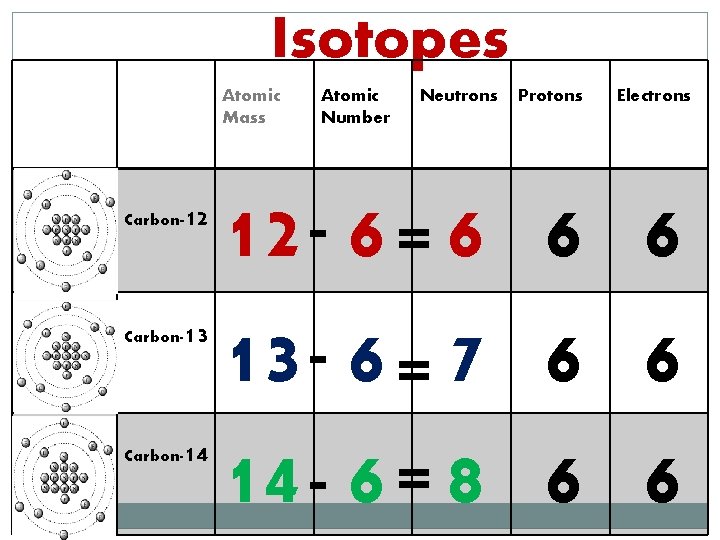
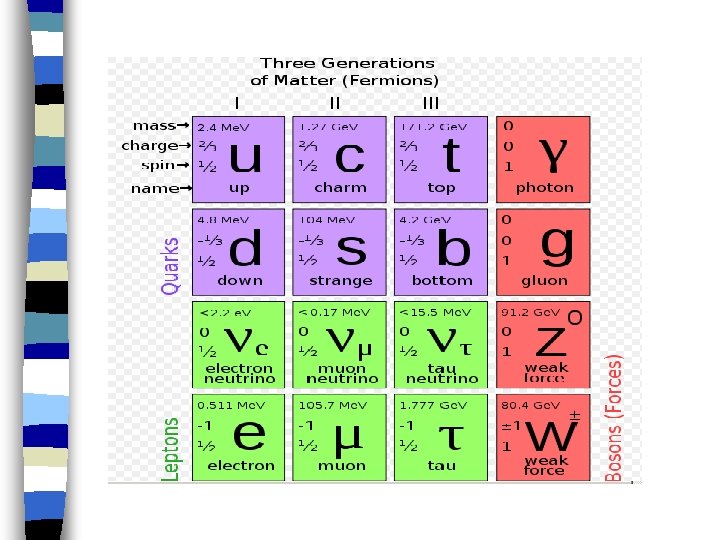
Isotopes Atomic Mass Carbon- 12 Carbon- 13 Carbon- 14 Atomic Number Neutrons Protons Electrons 12 - 6 = 6 6 6 13 - 6 = 7 6 6 14 - 6 = 8 6 6

The extra neutron causes 235 U to produce the unstable 236 U. Thus causing a chain reaction that eventually leads to a big BOOM!!!

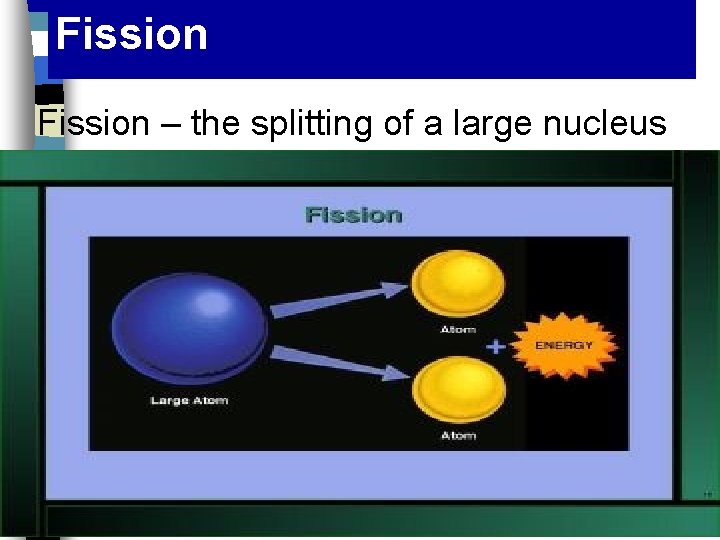
Fission The splitting of a nucleus accompanied by the release of neutrons and a large amount of energy

Fusion The process of combining nuclei to produce a nucleus of greater mass

History Only 2 nuclear weapons have ever been used in war. Approx. 200, 000 Japanese deaths (mostly Civilians)

Effects of the Nuclear explosion

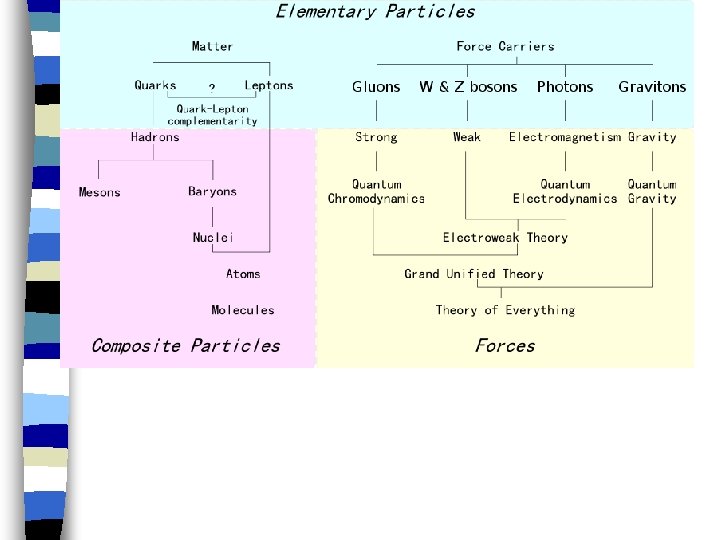
n Nuclear Reaction any change that involves nucleus

n 1896 - Antoine Henri Becquerel accidently discovered uranium ores emit invisible rays. Radioactivity

his 2 students, Marie Curie and Pierre Curie, isolated several radioactive elements. n Marie Curie named the process by which uranium gives off rays of radioactivity – radioactive decay n

The Atomic Nucleus Made of nucleons. . + proton & neutral - neutron. held by“strong nuclear force. ”

STRONG NUCLEAR FORCE = force that holds the nucleus together As size of nucleus increases, a stronger force needed to hold nucleus

Radioisotope – unstable isotope which undergoes radioactive decay

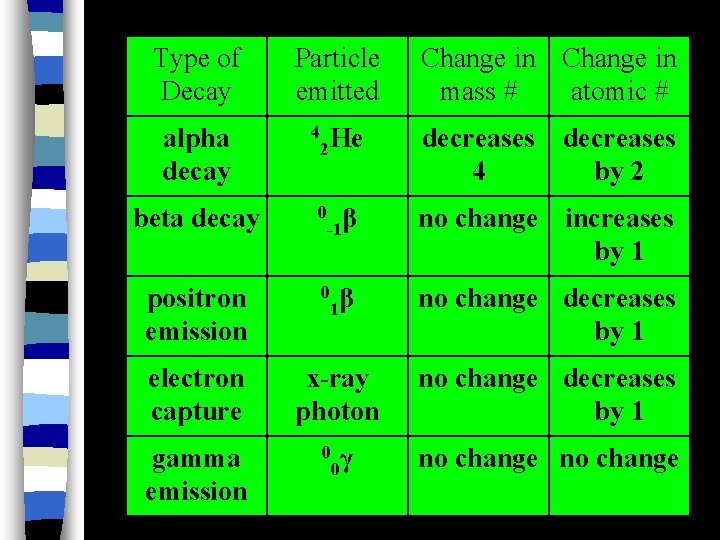
Type of Decay Particle emitted Change in mass # atomic # 2 He decreases 4 by 2 0 β -1 no change increases by 1 1β no change decreases by 1 electron capture x-ray photon no change decreases by 1 gamma emission 0 γ 0 no change alpha decay beta decay positron emission 4 0

Radioactive Decay Nucleus unstable when too many Protons & undergoes Radioactive decay

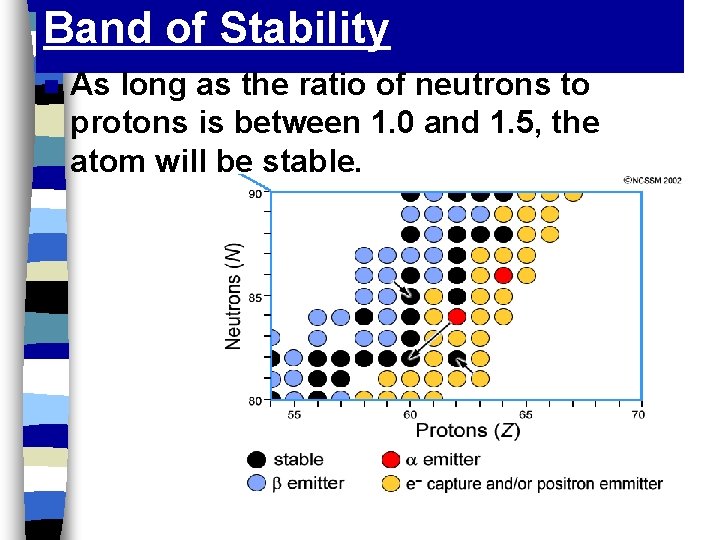
Band of Stability n As long as the ratio of neutrons to protons is between 1. 0 and 1. 5, the atom will be stable.

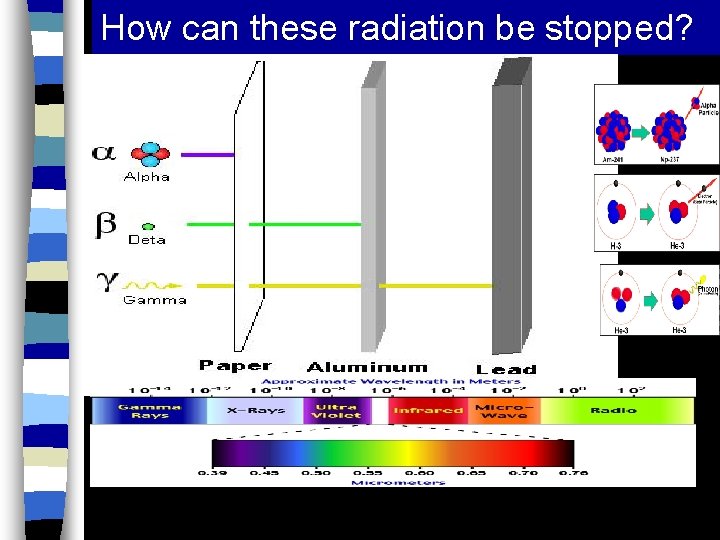
How can these radiation be stopped?

Alpha Radiation Radium - 226 decays into radon 222 and alpha particles. 226 --> 222 + 4 Ra Rn n 88 86 2 4 = e 4 n 2 2 n Alpha particles - helium nuclei n Use “Radioisotope, Radio Activity &Radioactive Decay” to describe the above Nuclear Reaction The Radioisotope, Radium 226, undergoes radioactive decay to form Radon. The type of radioactivity emitted is Alpha radiation

Beta Radiation 38 90 Sr --> 39 90 Y + - = e 0 - -1 Beta particles - high speed e-

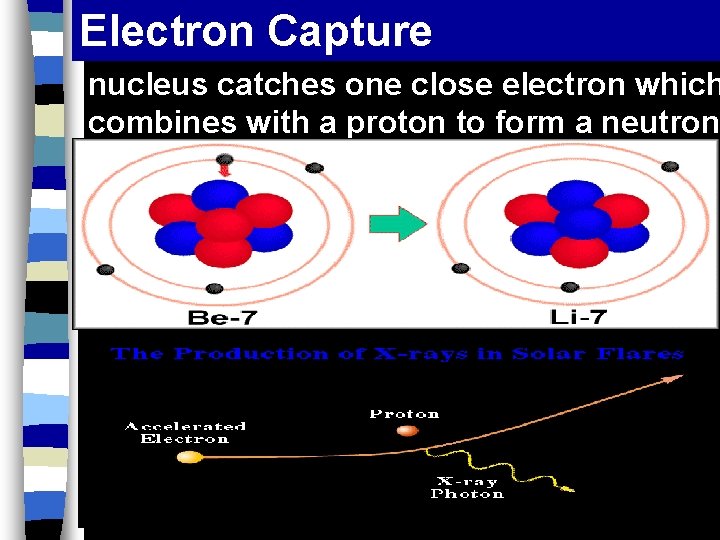
Electron Capture nucleus catches one close electron which combines with a proton to form a neutron

Beta Radiation 90 --> 90 + Sr Y n 38 39 - n High speed e- from nucleus? How is mass the same for Y 90 but the charge is greater!

Beta Radiation 90 --> 90 + Sr Y n 38 39 - n Consider neutron decay. . . 1 n 0 n --> ? + - 1 1 + - n 0 n --> 1 p

* Isotopes chemically alike as protons & electrons determine chemical properties

Gamma Radiation n Iron - 60 decays to Co-60 & gamma particles. 60 --> 60 + + Fe Co n 26 27 = electromagnetic rad. n 0 n high energy photons.

Where is Radiation in our Life n Carbon – 14 dating n Nuclear Waste n X-rays n Radiation therapy for cancer

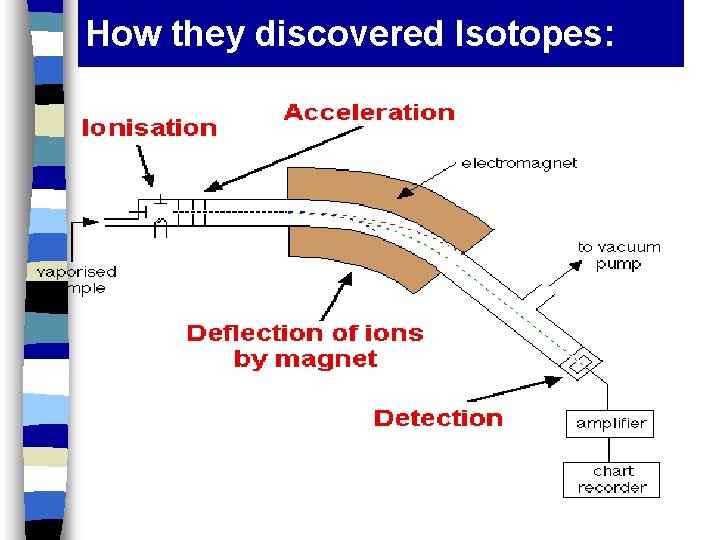
How they discovered Isotopes:

Hydrogen –Hydrogen has 3 isotopes • Hydrogen-1 = “Protium” 11 H • Hydrogen-2 = “Deuterium” 21 H • Hydrogen-3 = “Tritium” 31 H

Nuclear Fusion Nuclear fusion - 2 small nuclei COMBINE larger element. Fusion releases more energy than fission because only @ very high temp 40, 000 o. C fusion of hydrogen into helium keeps the sun burning.

Fusion n Fusion is how the sun continually produces energy. 4 11 H + 2 0 -1β 42 He + a lot of energy

Fusion – the combining of atomic nuclei

Fission – the splitting of a large nucleus

Fission (cont. ) n Fission is what nuclear power plants use for energy. n U – 235 + a neutron = a chain reaction of neutrons and energy

The Thorium Decay Series Pg 97 & 97 b 232 --> 4 + 228 Th Ra n 90 2 88 228 --> + 228 Ra Ac n 88 - 89 228 --> - + 90 Th n 89 Ac 228 4 224 --> 2 + 88 Ra n 90 Th 224 --> 4 + 220 Ra Rn n 88 2 86 220 --> 4+ 216 Rn Po n 86 2 84

The Thorium Decay Series 220 4 216 --> 2 + 84 Po n 86 Rn 216 --> 4 + 212 Po Pb n 84 2 82 212 --> + 212 or Pb Bi n 82 - 83 212 --> 4 + 208 Bi Tl n 83 2 81 208 --> + 208 Tl Pb n 81 - 82 208 is stable n 82 Pb

n. Chernobyl n. Hurt



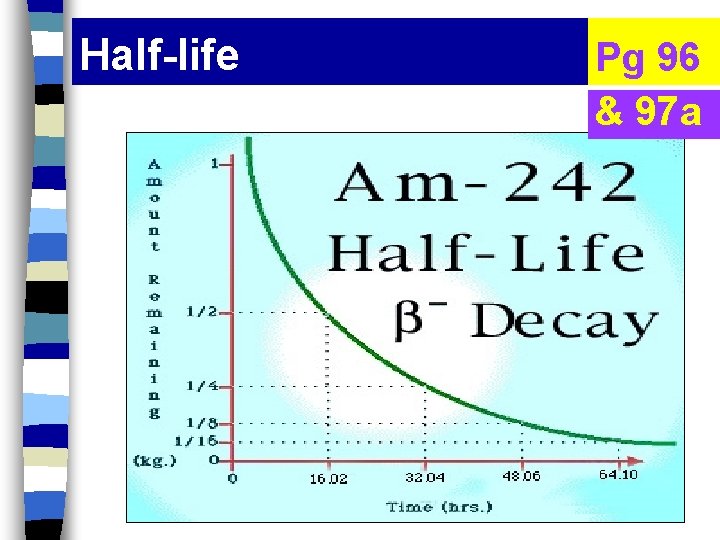
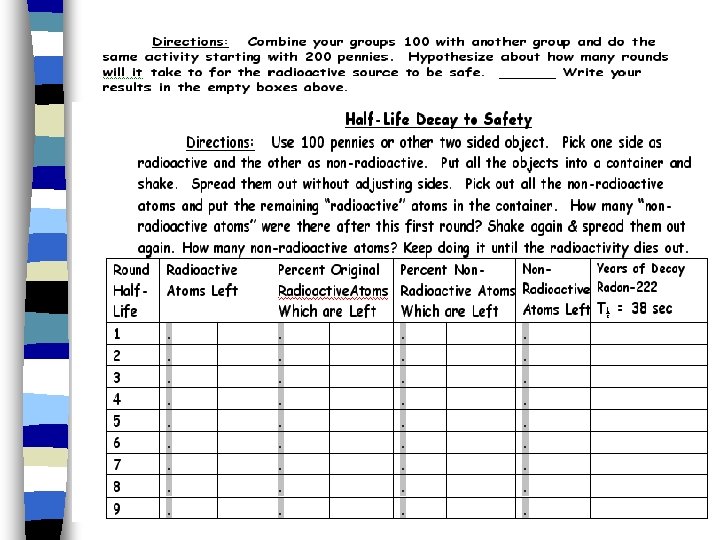
Half-life Pg 96 & 97 a


If you have 100 g of Uranium, How many half lives are needed to reach ~ 3% of the original radioactivity. 50 g 25 g 12. 5 g 100 g 1 ½ st life 6. 25 g 5 th ½ life 2 nd ½ life 3. 125 g 3 rd life 4 th ½ life

Half – Life Problems: 1) If I have 50 g of Pb-208 and it has decayed during 2 half-lives, how much is remaining? 2) If I have 25 g of U-235 and it has a half-life of one day, how much will be left after a week?

Critical Mass & The Enola Gay Nuclear Activities Directions: Each student will hold 3, 6 or 9 ping pong balls. Divide the room into two sections. Throw one ping pong ball. The rule is that when a student is hit by one ping pong ball, three must be released randomly. 1. What happened? Why? Directions: Now put the entire class within a 3 meter radius and try again. 2. What happened? Why?

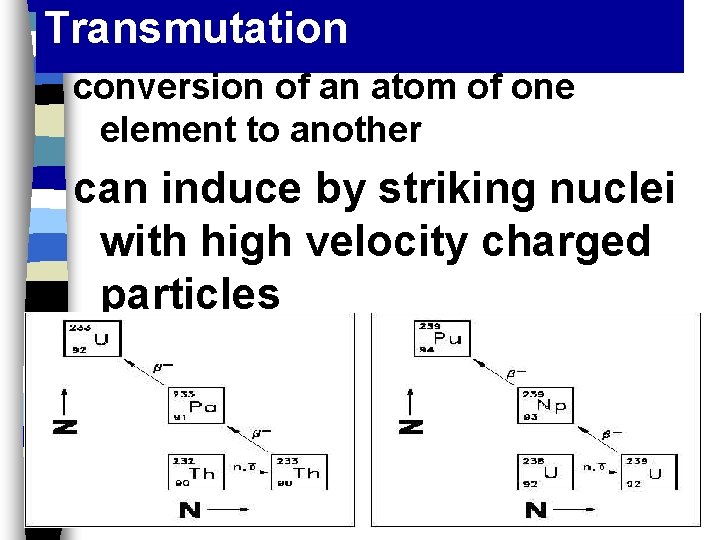
Transmutation conversion of an atom of one element to another can induce by striking nuclei with high velocity charged particles

n. Neutron-Proton Example nuclides. Ratio with magic numbers of nucleons and Nuclear Stability These proton and neutron numbers includes: are called “Magic Numbers. ” Magic numbers are: 2 8 20 28 50 82 126 44


Half-life n time needed for 1/2 of the particles to decay.

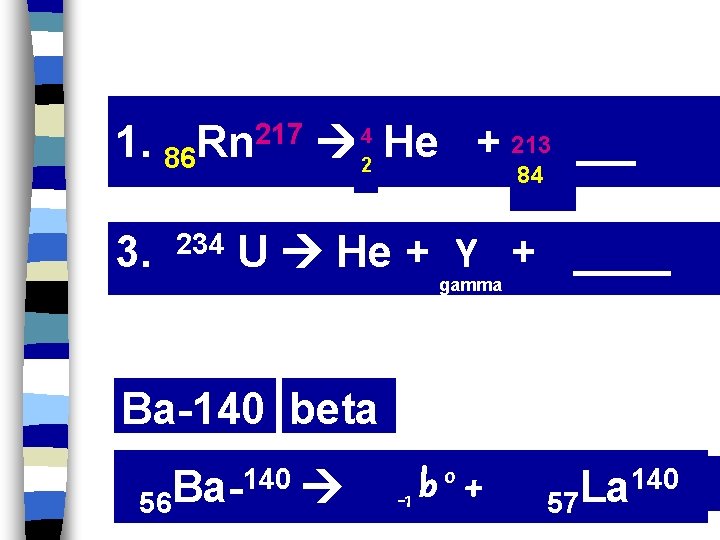
1. 86 3. Rn 217 234 He + 213____ 4 2 84 U He + Y + ____ gamma Ba-140 beta 56 Ba-140 o+ b -1 140 La 57

Radiation Practice 1) *U – 235 + → U – 235 2) Ag – 107 + → In – 111 3) N – 12 + → O -12 4) Fe – 57 + → Ni - 61

Pg 108 7. Show the equation for the fission of Th-232 with one fission product being Mo-96 plus the release of 2 neutrons 232 90 Th 96 Mo + 1 0 n If I had 20 g of U-235 & it decayed over 3 half-lives, how much would I have left?

1) If I have 10 g of U-235 and it has decayed over 7 half-lives, how much U-235 will I have remaining? 2) If I have 40 g of Pb-208 and it has a half life of 15 seconds, how much will remain after 1 minute? 3) If I have 20 g of Pt- 144 remaining after it decayed for a week. Given that it has a half-life of a day, how much did I have originally?

Fusion vs. Fission n There are two processes that make use of the enormous amount of energy in a nucleus: Fusion and Fission

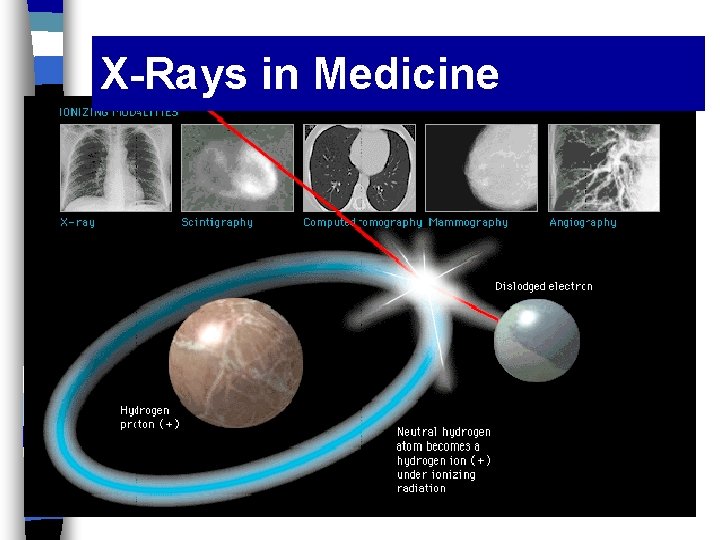
Ionizing Radiation n Ionizing radiation = radiation with enough energy to knock an electron off an atom and create an ion. n X-rays and gamma rays will ionize almost any molecule or atom - -

X-Rays in Medicine

n Transuranium Elements are all elements with an atomic number of 93 or greater –all radioactive –all man-made (SYNTHETIC)


Leptons e+ νe μ + νμ τ + ντ Up quarks Charm quarks 1 u 3|Vud c 3|Vcd| 2 2 d | d 1 u 3|Vus| cs 3|Vcs| 2 2 s 1 u 3|Vub c 3|Vcb| 2 2 b | b


n. Example 26 -1: Calculate the Nuclear Stability and mass deficiency for 39 K. The Binding Energy actual mass of 39 K is 39. 32197 amu per atom. 58

Fission & Fusion Demos Directions: Blow up a balloon. Pinch and twist in the center. Nuclear Fission! Directions: Put 2 drops of water on the overhead projector. Nuclear Fusion! Questions: 1. Why do Doctors use radioactive isotopes which have short half lives? 2. What happens to the nucleus during Nuclear Fission? 3. How many half-lives does Radon need to live out to get rid of about 87. 5% of the radioactivity?



End here