Radioactivity Types of Reaction Types of Radiation Nuclear














































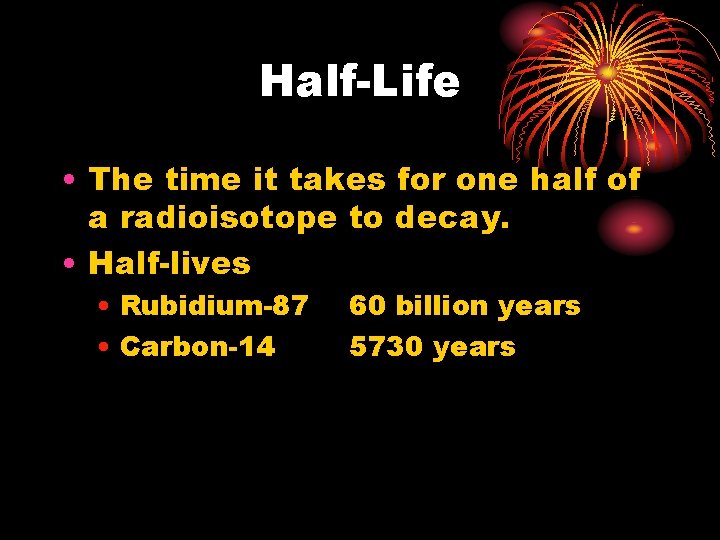




- Slides: 52

Radioactivity Types of Reaction Types of Radiation Nuclear Equations

Reactions • Chemical reaction- atoms interact only through outer electrons • New substances are formed but the atoms identity remains the same. • Nuclear reaction- change the composition of an atom’s nucleus.

Reactions • Chemical vs Nuclear • Both nonrenewable • Nuclear produces far more energy then the same amount of chemical

Radioactivity • Discovered in 1896 • Spontaneous emission of radiation from an element- radioactive decay • Fundamental changes in an atom • Transmutation- One element changes into another

Nuclear Reaction • Radioactive atoms emit radiation to try and stabilize their unstable nuclei • Three different kinds of radiation can be emitted • Alpha (α) particle • Beta (β) particle • Gamma (γ) ray • http: //www. remm. nlm. gov/ionizing radsvideo. htm

Alpha • 2 protons and 2 neutrons • +2 charge • Identical to He-4

Alpha • Symbol

Alpha • Low penetrating power • Travel only short distances • Stopped by paper or clothing • Not much of a health risk

Beta • -1 charge • Mass = 0

Beta • A neutron changes into a proton and an electron • The proton stays in the nucleus and the electron (beta particle) is propelled out of the nucleus at high speed.

Beta • 100 x more penetrating than alpha radiation • Able to penetrate through clothing and damage skin.

Beta • Symbol

Gamma • Similar to x-rays • Much more penetrating then alpha or beta • Able to penetrate deeply into solid materials

Gamma • Stopped by lead or concrete • Very dangerous

Gamma • Symbol

Characteristics of Alpha, Beta, and Gamma Radiation Symbol Type Mass (amu) Alpha 4 Beta 1/1840 Gamma 0 Charge 0

What makes one atom radioactive and the other not? • The ratio of protons to neutrons that an atom contains

Radioactive? • Most stable nuclei have almost equal number of protons and neutrons. • Nuclei need increasingly more neutrons then protons to be stable.

Radioactive? ? • When the atomic number of an atom exceeds 83, the number of neutrons is insufficient. • So, all atoms with atomic # greater then 83 are radioactive. • Ex. Radium, Uranium, Francium

Radioactive? ? • Nuclei can also be unstable if they have too many neutrons.

How can protons exist so close to each other? • Nucleons- the protons and neutrons of a nucleus. • Electrostatic Repulsion- (like charges repel) • In order for the nucleus to stay together, there must be a attraction stronger then the repulsion.

Strong Nuclear Force • Holds together all protons and neutrons of a stable nucleus • Only works over short distances • Strong force > electrostatic repulsion

How does an atom not fall apart? • Strong force travels over short distances and repulsion forces can travel long distance, the forces balance each other out for nuclear stability. • If they are not balanced, the nucleus falls apart. • www. youtube. com/watch? v=0 B 69 RHq. Afj 8

Radioactive Decay • An atom emits one of the types of radiation. • Original nucleus decomposes, or decays, to form a new nucleus releasing radiation in the process.

Nuclear Reactions • Nuclear Fusiontwo smaller nuclei fuse to form one larger nuclei. Lots of energy is released. • Occurs in stars

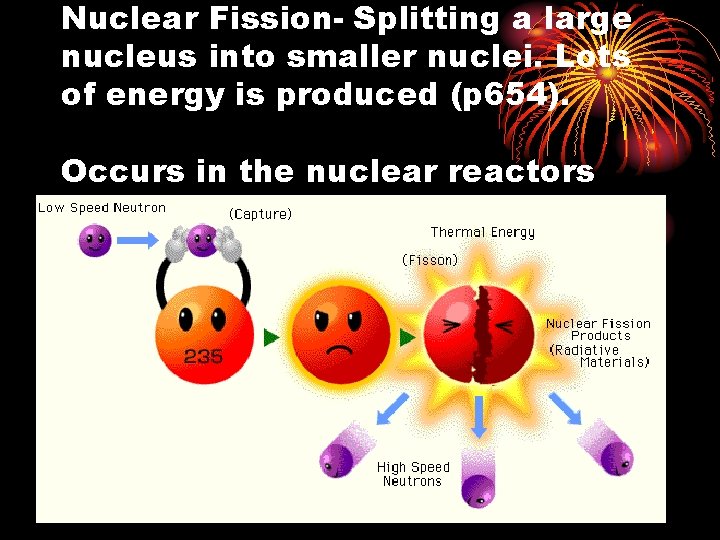
Nuclear Fission- Splitting a large nucleus into smaller nuclei. Lots of energy is produced (p 654). Occurs in the nuclear reactors

Nuclear Equations • Alpha decay of an isotope of radium-228.

Nuclear Equations • Beta decay of Iodine-131.

Nuclear Equations • Practice • Alpha decay of Gold-185. • Beta decay of Sodium-24.

Nuclear Equations • 9 4 Be + • 37 1 1 H → _____ + 19 K → _____ + • 241 95 Am + 4 0 4 2 He +1 e 1 n + _____ He → 2 2 0

Where does all the energy come from? • When protons and neutrons come together to form a nucleus energy is released. • Nucleons Nucleus + energy

Energy? • Mass of any atom is LESS then the combined individual parts. 7 amu

Mass Defect • The difference in the mass of the whole vs the mass of the individuals. • E=mc 2 mass can be converted to energy

Energy • Nuclear Binding Energy- energy released when the protons and neutrons come together. • The energy released is ENORMOUS compared to a chemical reaction.

Pros to nuclear power • Emits relatively low amounts of carbon dioxide (CO 2): contribution of to global warming is therefore relatively little (less than one hundreth the Greenhouse Gases of Coal or Gas fired power stations) • This technology is readily available. • Generates a high amount of electrical energy

Cons of nuclear power • The waste is extremely dangerous and it has to be carefully looked after for several thousand years • High risks: Despite high standards, accidents can still happen. The consequences of an accident can be absolutely devastating. • Nonrenewable

Nuclear Power Plants • Today’s power plants produce energy through fission chain reactions. • Fusion reactions are cleaner and safer but, with our present technology, it takes more power to do fusion then it produces.

Nuclear Power Plants

Nuclear incidents • 3 -mile Island • 1979 • Partial nuclear meltdown in Pennsylvania

Chernobyl- April 26, 1986

Time Line • 26 April 1986, 1: 23: 00: A test of the cooling system begins in unit no. 4 of the Chernobyl power plant • 26 April 1986, 1: 23: 40: The emergency shutdown fails. • 26 April 1986, 1: 23: 44: The reactor in unit no. 4 runs out of control and explodes. • 26 April 1986: In the town of Pripyat, 3 km away, there are 45 000 people, including 16 000 children enjoying the first warm Sunday of the spring.

• 27 April 1986: The inhabitants of the Pripyat power plant settlement are evacuated. • 28 April 1986, 21: 00: The Soviet news agency Tass announces that there has been an accident at the Chernobyl nuclear power station and that there have been casualties. An investigative committee is established. • 29 April 1986: The accident at Chernobyl is first reported in German newscasts.

• 26 April to 4 May 1986: Most of the radiation is released in the first ten days. • 27 April to 5 May 1986: About 1800 helicopter flights deposit around 5000 tonnes of extinguishing materials such as sand lead onto the burning reactor.

Radiation Spread • The U. S. S. R. was silent about the accident until after it had been detected by its neighbors to the north. • http: //hyperphysics. phyastr. gsu. edu/hbase/nucene/imgnuk/c hercloud. gif

Fukushima • 2011 • Japan • Largest disaster since Chernobyl • 3 reactors experienced meltdowns before being brought under control.


Radioisotopes • Elements have one or more isotopes that are unstable and decay to produce other elements
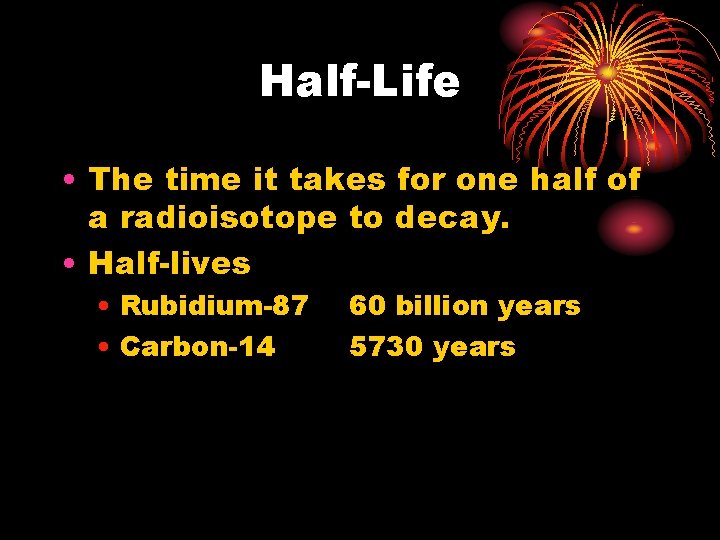
Half-Life • The time it takes for one half of a radioisotope to decay. • Half-lives • Rubidium-87 • Carbon-14 60 billion years 5730 years

Half-life • Ex. The half-life of mercury-195 is 31 hours. If you start with a 5. 00 g of mercury-195, how much of it will still be present after 93 hours?

Half-life • Try: Gold-191 has a half-life of 12. 4 hours. What mass of this isotope would remain after 49. 6 hours if you started with a 7. 50 g sample of Au-191?

Carbon dating • Process of determining the age of an object by measuring the amount of carbon-14 remaining in the object. • http: //www. youtube. com/watch? v=31 -P 9 pc. PStg

Nuclear Use • Medical: PET Scan • http: //en. wikipedia. org/wiki/File: PE T-MIPS-anim. gif • Smoke Detector • contains an Radioisotope: americium-241.