Radioactivity inverse square law absorption and rates Inverse

Radioactivity – inverse square law, absorption, and rates

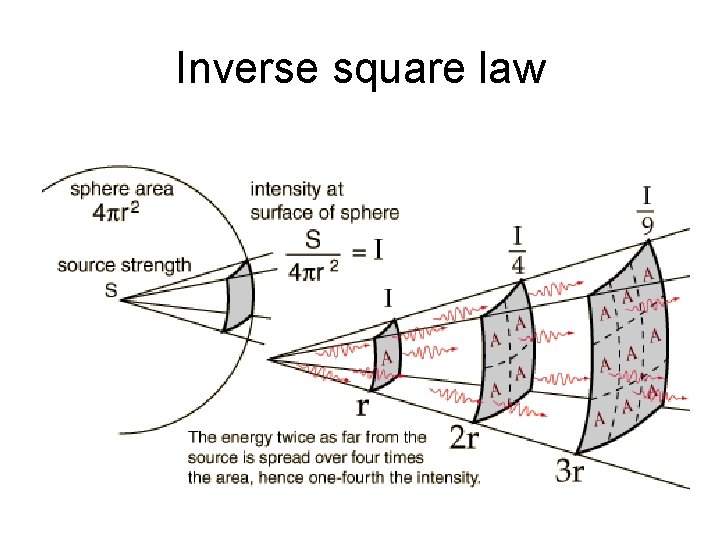
Inverse square law for radiation • Particles and photons emitted by radioactive nuclei continue moving until they are absorbed by some material. • The particles (or photons) that leave the sample will continue to move out in a radial direction. • The number that pass through an imaginary sphere of radius r in some interval of time is the same for different radii, but the area of the sphere depends on the radius. A = 4 pr 2 • Therefore, the number per unit area per unit time depends on the radius.
Inverse square law

Relation between source activity and intensity at various radii • Suppose that S is the number of particles emitted by the source per unit time. • This S might be due to an activity which is written as S decays per second. (in Bq) • At a distance r, the particles pass through a sphere of radius r and area A = 4 pr 2 • Intensity is I = S/A, the number of particles, per unit time, per unit area. • Then I = S/4 pr 2 • I decreases as the inverse square of the radius.

Intensity for r, 2 r, 3 r, etc. • If the intensity is I 1 at a radius r 1, then if we double the radius to 2 r 1, the intensity will be ¼ as much, because the area is now 2 x 2 = 4 times as much. • More generally, if we compare the intensity at radii r 1 and r 2, then we get a ratio of intensities that depends on the square of the radii: • I 2 / I 1 = (r 1/r 2)2 • For example, if we compare radii of r and 3 r, the intensities have a ratio of 1/9 = (1/3)2
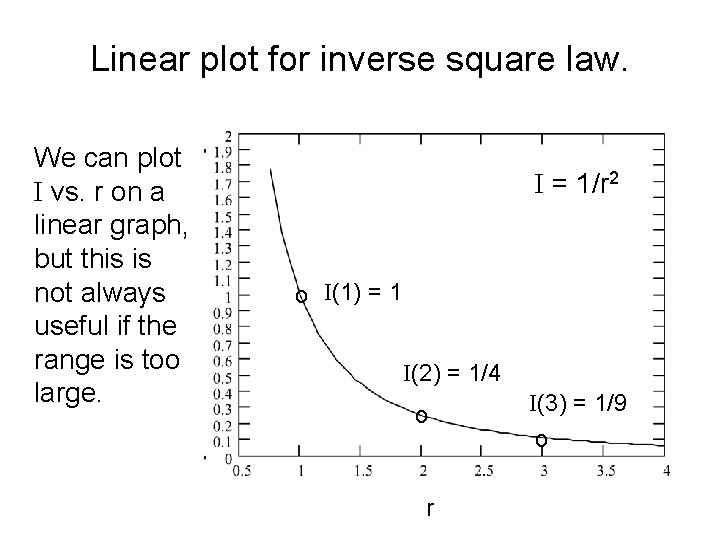
Linear plot for inverse square law. We can plot I vs. r on a linear graph, but this is not always useful if the range is too large. I = 1/r 2 o I(1) = 1 I(2) = 1/4 o r I(3) = 1/9 o
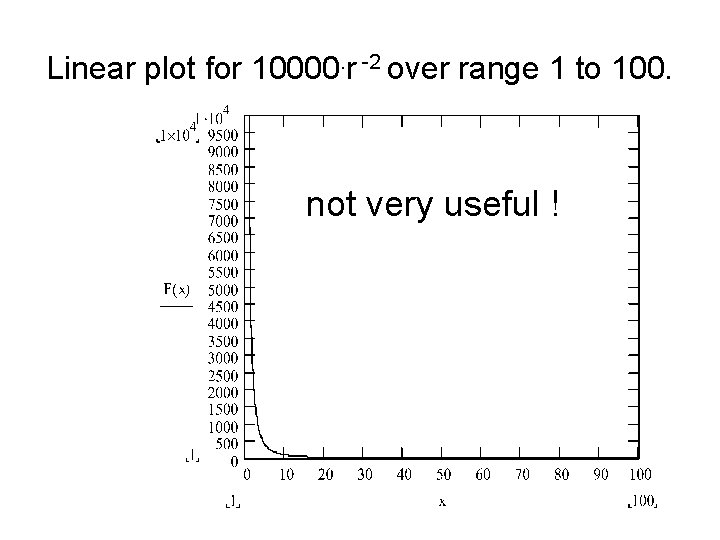
Linear plot for 10000. r -2 over range 1 to 100. not very useful !

Log-log plot for inverse square law. • If the intensity is a function of radius that is a power law rm (for example, inverse square is a power law, since I = a. rm where m = -2), • then, we can plot I vs. r on a log-log graph. • Applying the logarithm to both sides of the equation: and using log(ab) = log(a) + log(b) • log (I) = log(a. rm) = log(a) + m. log(r) • and if y = log(I), x = log(r), and log(a) = b, we have the eq. of a straight line y = m. x + b
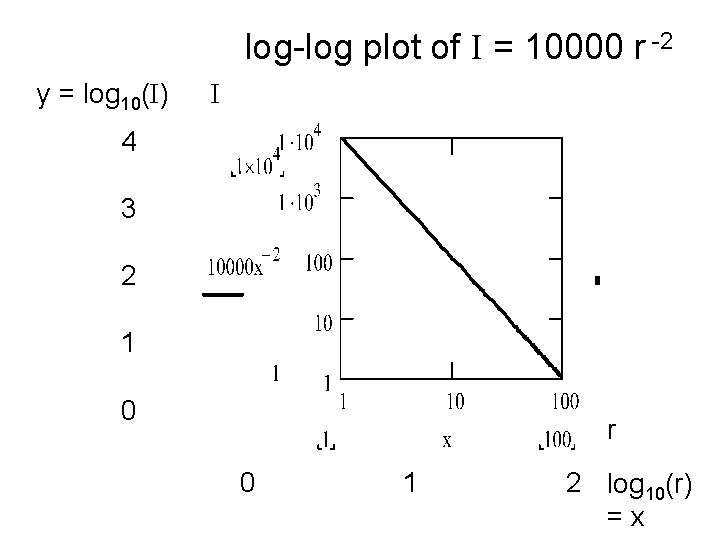
log-log plot of I = 10000 r -2 y = log 10(I) I 4 3 2 1 0 r 0 1 2 log 10(r) =x

Analysis of I = 10000 r -2 How does this equation produce a straight line on the log-log plot? log(I) = log(10000. r-2) = log(10000) + log(r-2) = log(104) + (-2). log(r) Now define y = log(I) and x = log(r) and then y = m. x + b with b = log(104) = 4 (the intercept) m = -2 is the slope

We study this in laboratory # 10. We use laboratory equipment to study the distance dependence of the radiation from a small source. This is examined experimentally using log-log graph paper. We also examine the use of shielding materials, which requires semi-log paper to plot the absorption of gamma rays.

Absorption of X-rays and gamma rays • X-rays and gamma rays can be very penetrating. • Scattering of photons is not very important. It is more probable for the photon to be absorbed by an atom in the photoelectric effect. • The photon is absorbed with some probability as it passes through a layer of material. This results in an exponential decrease in the intensity of the radiation (in addition to the inverse square law for distance dependence).
Exponential absorption of X-rays I = Io at detector, with no absorber
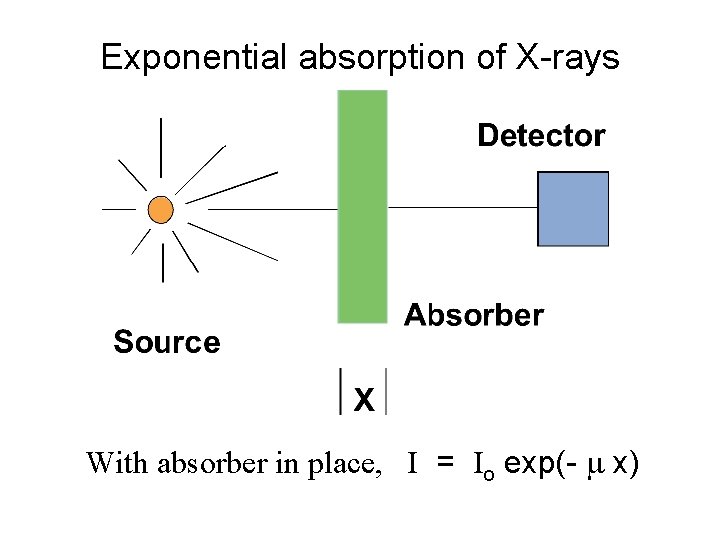
Exponential absorption of X-rays With absorber in place, I = Io exp(- m x)

Exponential absorption of X-rays The exponential decrease in the intensity of the radiation due to an absorber of thickness x has this form: I = Io exp(- m x) = Io e - m x where Io is the intensity without the absorber, I is the intensity with the absorber, and m is the linear absorption coefficient. m depends on material density and X-ray energy.
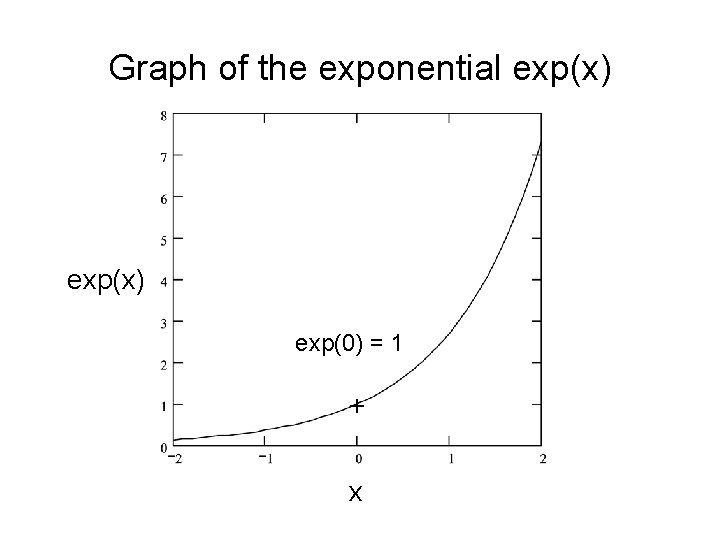
Graph of the exponential exp(x) exp(0) = 1 + x
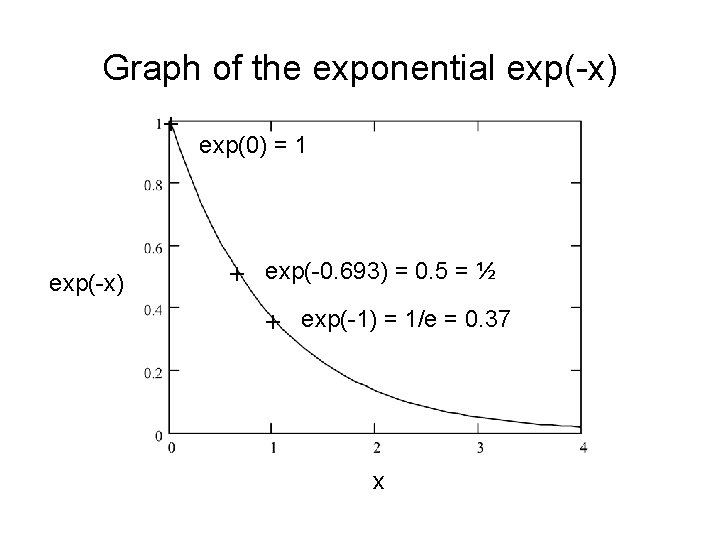
Graph of the exponential exp(-x) + exp(-x) exp(0) = 1 + exp(-0. 693) = 0. 5 = ½ + exp(-1) = 1/e = 0. 37 x

Half-thickness for absorption of X-rays For a particular thickness x ½ the intensity is decreased to ½ of its original magnitude. So if I(x½) = Io exp(- m x ½) = ½ Io we solve to find the half-thickness x ½. exp(- m x ½) = ½ and m x ½ = 0. 693 so x ½ = 0. 693 / m

Calculation of half-thickness To calculate x ½ (of lead, Pb) we need to know m. As an example, for X-rays of energy 50 ke. V, m = 88 cm-1 and x ½ = 0. 693/m so x ½ = 0. 693 / (88 cm-1) = 0. 0079 cm But for hard X-rays with energy 433 ke. V, m = 2. 2 cm-1 so x ½ = 0. 693 / (2. 2 cm-1) = 0. 31 cm
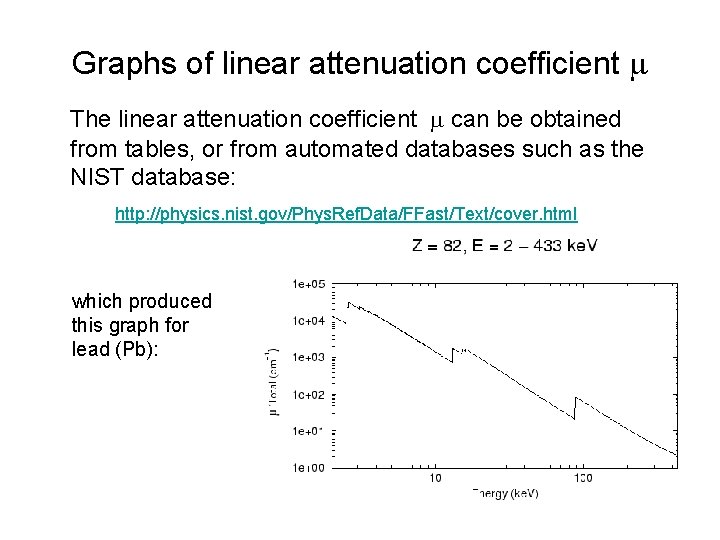
Graphs of linear attenuation coefficient m The linear attenuation coefficient m can be obtained from tables, or from automated databases such as the NIST database: http: //physics. nist. gov/Phys. Ref. Data/FFast/Text/cover. html which produced this graph for lead (Pb):
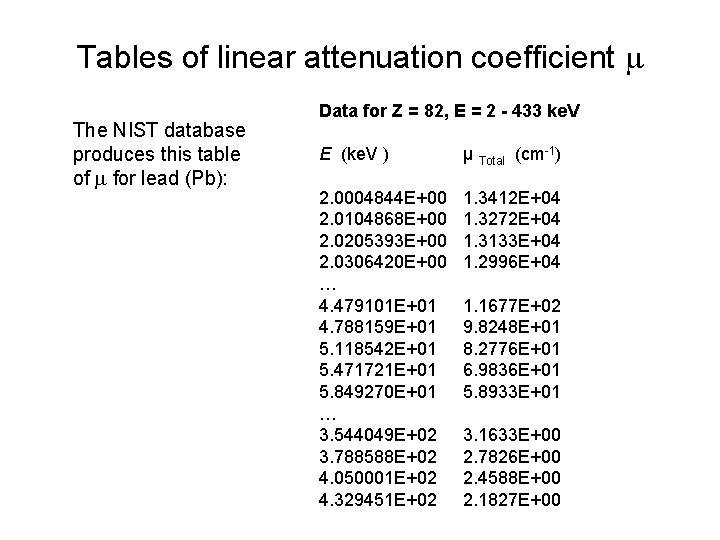
Tables of linear attenuation coefficient m The NIST database produces this table of m for lead (Pb): Data for Z = 82, E = 2 - 433 ke. V E (ke. V ) µ Total (cm-1) 2. 0004844 E+00 2. 0104868 E+00 2. 0205393 E+00 2. 0306420 E+00 … 4. 479101 E+01 4. 788159 E+01 5. 118542 E+01 5. 471721 E+01 5. 849270 E+01 … 3. 544049 E+02 3. 788588 E+02 4. 050001 E+02 4. 329451 E+02 1. 3412 E+04 1. 3272 E+04 1. 3133 E+04 1. 2996 E+04 1. 1677 E+02 9. 8248 E+01 8. 2776 E+01 6. 9836 E+01 5. 8933 E+01 3. 1633 E+00 2. 7826 E+00 2. 4588 E+00 2. 1827 E+00
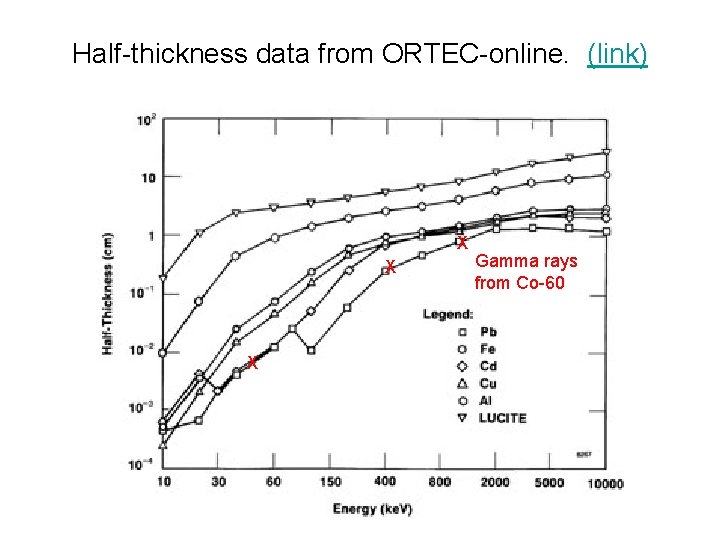
Half-thickness data from ORTEC-online. (link) X X X Gamma rays from Co-60

Shielding of X-rays and gamma rays • To reduce the intensity of radiation from a source, we can use an absorber in the path of the radiation. This is called shielding. • To minimize I = Io exp(- m x) we want to increase m or x. Then the exponential will be smaller, and I will be smaller for constant Io. • To increase the absorption coefficient m we need to increase the density of the shielding. • To increase the value of x we must use thicker shielding.

Shielding of charged particles (alpha and beta particles) • The absorption of charged particles is quite different from the absorption of X-rays (or g). • Charged particles lose kinetic energy continuously, instead of being absorbed in one single event like photons, and they also can scatter (change direction). • The result is a range, a distance that only a small number of particles reach. • Beyond the range, there is zero intensity.

Range of alpha and beta particles • The range of alpha particles is a few centimeters in air and much less in solids. • Alphas may be completely absorbed by a single sheet of paper or by your skin • Beta particles can travel a few meters in air or a few millimeters in organic materials, depending on their kinetic energy. One cm of polymer will usually stop beta particles. However, they can easily pass through skin or gloves.
- Slides: 25