Radioactive isotopes and radiation By Prof Ahmed Mostafa

Radioactive isotopes and radiation By Prof. Ahmed Mostafa Professor of anesthesia and ICU

Some basic facts about the structure of an atom are: - The nucleus consists of neutrons and protons and is orbited by electrons. - The mass number is equal to the number of neutrons plus the number of protons in the nucleus. It is the nearest integer to the atomic weight.

- The atomic number is equal to the number of protons (or electrons) in the nucleus, and determines which element is present. - An element may exist in different forms due to variations in its nuclear structure. These different forms are called isotopes of the element. Each isotope will differ in mass number but will possess the same atomic number.

- Notation used to identify isotopes is as follows: - Some isotopes are stable and retain their nuclear structure indefinitely, while other isotopes are unstable and decay spontaneously. Unstable isotopes are said to be radioactive, and they decay by emitting radiation.


Radioactive decay Decay of a radioactive isotope involves the emission of nuclear particles from the substance, resulting in the formation of another isotope or another element. An example is the uranium series, which occurs when uranium decays. This series of reactions finally results in the formation of lead, but involves a chain of intermediate decay reactions. Some of these are illustrated in the figure.


- Each of these reactions involves the emission of a specific type of particle with a characteristic mean energy level. - Each reaction also has a particular half-life (see figure).

Types of radiation (ionizing radiation) - α – particles: a combination of two protons and two neutrons (equivalent to a helium nucleus)

Types of radiation (ionizing radiation) - β – particles: an electron which is negatively charged, derived from a neutron splitting to give a proton and a high-energy electron. The creation of another proton in the nucleus will increase the atomic number of the atom and thus change the element present.

Types of radiation (ionizing radiation) Sometimes an electron may be emitted from the nucleus with a positive charge, in which case it is called a positron.

Types of radiation (ionizing radiation) - γ – radiation: electromagnetic radiation with wavelengths < 10− 12 m, which is emitted during most nuclear reactions, usually following the emission of an α or β particle. - X - rays: form of electromagnetic radiation with wavelengths in the range 10− 8 m to 10− 12 m. The main properties of x rays in medical applications are their ability to penetrate tissue and their ionising effect.
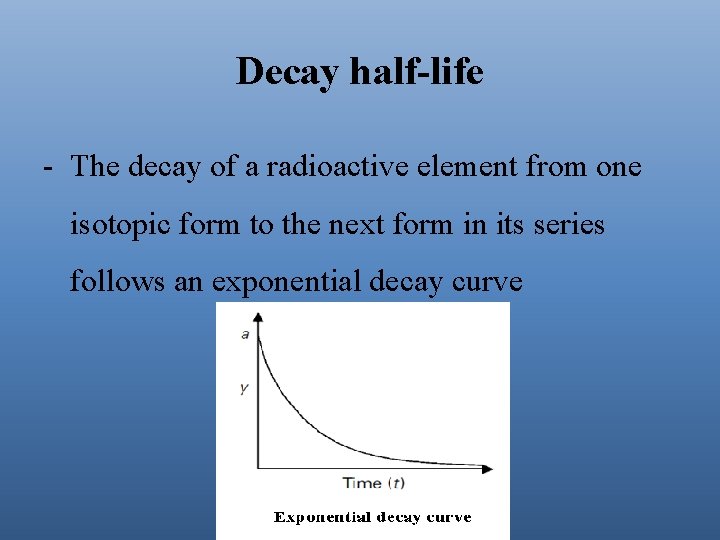
Decay half-life - The decay of a radioactive element from one isotopic form to the next form in its series follows an exponential decay curve

Decay half-life - The half-life for the element (T 1/2) is the time for the mass of element to decay to a half of its initial mass. - The half-life may have a value ranging from seconds to millions of years.

Applications of radioactive isotopes - Measurements with labelled substances: chromium-51 in measurement of red cell volume - Cancer therapy: use of yttrium-90 in pituitary tumours - Diagnostic uses: imaging techniques with technetium-99 as in assessment of cardiac function.

Hazards of exposure to ionizing radiation: - Short-term hazards (Acute radiation sickness): • These are dose dependent and include nausea, vomiting, anorexia, general lassitude and weakness, and can lead to death over a period of days or less. - Long-term hazards: • Include an increased incidence of cancers and genetic defects in the population, which occur after a period of years.

Thank you Dr. Ahmed Mostafa
- Slides: 17