Radiation Therapy for SmallCell Lung Cancer ASCO Guideline































- Slides: 32

Radiation Therapy for Small-Cell Lung Cancer: ASCO Guideline Endorsement of an ASTRO Guideline Daly et al. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

2 Overview 1. Background & Methodology • • Introduction ASCO Endorsement Methodology Clinical Questions Target Population and Audience 2. Summary of Recommendations 3. Discussion • • • Role of SBRT in Stage I or II Node-Negative SCLC Prophylactic Cranial RT Thoracic Consolidation for ES-SCLC Endorsement Statement Additional Resources Expert Panel Members www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

1 Background & Methodology

4 Introduction • Radiation therapy (RT) is used in the management of both limited-stage small-cell lung cancer (LS-SCLC) and extensive-stage SCLC (ES-SCLC) For LS-SCLC, RT is used for the curative-intent treatment of thoracic disease, with consideration of prophylactic cranial irradiation (PCI) to reduce risk of brain metastases For ES-SCLC, radiation is considered as a consolidation therapy to the thorax following front-line systemic therapy. PCI is also considered • Areas of controversy surrounding the use of RT for SCLC include the appropriate dose and fractionation used for LS-SCLC, the appropriateness of PCI in both LS- and ESSCLC, and the role of consolidative thoracic RT for ES-SCLC • An ASCO Expert Panel critically appraised the American Society for Radiation Oncology’s (ASTRO) Radiation Therapy for Small-Cell Lung Cancer: An ASTRO Clinical Practice Guideline, 1 and added minor clarifications aimed at the ASCO audience • This endorsement reinforces the recommendations provided in the ASTRO guideline and acknowledges the effort put forth by ASTRO to produce this evidence-based guideline www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

5 ASCO Endorsement Methodology • The ASCO Clinical Practice Guidelines Committee (CPGC) endorsement review process includes: a methodological review by ASCO guidelines staff a content review by an Expert Panel final endorsement approval by ASCO CPGC • The full ASCO Guideline methodology manual can be found at: www. asco. org/guidelinemethodology • ASTRO Guideline methodology can be found at: https: //www. astro. org/Patient-Care-and. Research/Clinical-Practice-Statements/ASTRO-39; s-Guideline-Development-Process www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

6 Clinical Questions The ASTRO guideline addressed the following questions: 1. What are the indications, appropriate dose-fractionation schedules, techniques, and timing of thoracic RT for LS-SCLC? 2. What is the role of stereotactic body radiotherapy (SBRT) compared with conventional RT in stage I or II node-negative SCLC? 3. What are the indications, appropriate dose-fractionation schedules, and timing of prophylactic cranial RT for LS-SCLC and ES-SCLC? 4. What are the indications, appropriate dose-fractionation schedules, and timing of thoracic consolidation in patients with ES-SCLC? www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

7 Target Population and Audience Target Population • Patients with LS- and ES-SCLC Target Audience • Radiation oncologists, medical oncologists, surgeons, pulmonologists, primary care providers, nurse practitioners, physician assistants, nurses, and other providers. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

2 Summary of Recommendations

9 Summary of Recommendations Clinical Question 1 • What are the indications, appropriate dose-fractionation schedules, techniques, and timing of thoracic RT for LS-SCLC? Recommendation 1. 1 • For patients with LS-SCLC who can tolerate definitive therapy, thoracic RT is recommended. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org Quality of Evidence Strength of Recommendation High Strong

10 Summary of Recommendations Recommendation 1. 2 • For patients with LS-SCLC receiving chemotherapy and RT, thoracic RT should begin with cycle 1 or 2 of chemotherapy. • Implementation Remark: It is important to maintain the dosage and timing of chemotherapy with RT based on trial data. Timing is more critical for accelerated dose-intensive RT. • ASCO clarifying comment: including twice-daily, hyperfractionated regimens. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org Evidence Quality Strength of Recommendation Moderate Strong

11 Summary of Recommendations Recommendation 1. 3 • For postoperative patients with LS-SCLC and R 1 or R 2 resection, postoperative RT is conditionally recommended. Recommendation 1. 4 • For postoperative patients with LS-SCLC that is clinically nodenegative and pathologically N 2 -positive, mediastinal RT is conditionally recommended. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org Evidence Quality Strength of Recommendation Expert Opinion Conditional

12 Summary of Recommendations Recommendation 1. 5 • For patients with LS-SCLC, twice-daily RT in 150 -c. Gy fractions to 4, 500 c. Gy is recommended. Recommendation 1. 6 • For patients with LS-SCLC, daily RT in 200 -c. Gy fractions to 6, 000 -7, 000 c. Gy is conditionally recommended as an acceptable alternative to twice-daily RT. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org Evidence Quality Strength of Recommendation High Strong Evidence Quality Strength of Recommendation Moderate Conditional

13 Summary of Recommendations Recommendation 1. 7 • For patients with LS-SCLC, twice-daily RT in 150 -c. Gy fractions to 4, 500 c. Gy is recommended. Recommendation 1. 8 • For tumors that experience shrinkage with chemotherapy in patients with LS-SCLC, treating all involved nodal stations (at time of diagnosis) and postchemotherapy lung parenchymal tumor is recommended www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org Evidence Quality Strength of Recommendation High Strong Evidence Quality Strength of Recommendation Moderate Strong

14 Summary of Recommendations Recommendation 1. 9 • For patients with LS-SCLC, highly conformal techniques are recommended to minimize normal tissue dose. Evidence Quality Strength of Recommendation Low Strong Clinical Question 2 • What is the role of SBRT compared with conventional RT in stage I or II node-negative SCLC? www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

15 Summary of Recommendations Recommendation 2. 1 • For patients with stage I or II node-negative LS-SCLC who are medically inoperable, either SBRT or conventional fractionation is recommended. • Implementation Remarks: Ideally, the node-negative status should be confirmed by invasive nodal staging. Ultracentral tumors (ASCO clarifying comment: meaning those with the planning target volume touching or overlapping the proximal bronchial tree, esophagus, or trachea) may be more appropriately treated with conventional fractionation schema. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org Evidence Quality Strength of Recommendation Moderate Strong

16 Summary of Recommendations Recommendation 2. 2 • For patients with stage I or II node-negative LS-SCLC receiving SBRT, chemotherapy should be delivered to patients in whom it is medically tolerated. Evidence Quality Strength of Recommendation Moderate Strong Clinical Question 3 • What are the indications, appropriate dose-fractionation schedules, and timing of prophylactic cranial RT for LS-SCLC and ES-SCLC? www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

17 Summary of Recommendations Recommendation 3. 1 • For patients with SCLC who respond to initial therapy, restaging with brain MRI to guide decision making regarding PCI is recommended. Recommendation 3. 2 • For patients with stage I SCLC, PCI is conditionally not recommended • Implementation Remark: In lieu of PCI, surveillance using brain MRI with contrast can serve as an alternative. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org Evidence Quality Strength of Recommendation Low Strong Evidence Quality Strength of Recommendation Low Conditional

18 Summary of Recommendations Recommendation 3. 3 • For patients with stage II-III LS-SCLC who are < 70 years of age with good performance status (ECOG 0 -2) and respond to thoracic chemoradiation, PCI is recommended. Recommendation 3. 4 • For patients with LS-SCLC who have limited performance status, older age, and/or significant comorbidities, shared decision making on PCI (considering patient- and disease-specific characteristics) is recommended. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org Evidence Quality Strength of Recommendation High Strong Evidence Quality Strength of Recommendation Low Strong

19 Summary of Recommendations Recommendation 3. 5 • For patients with LS-SCLC receiving PCI, 2, 500 c. Gy in 10 fractions is recommended Recommendation 3. 6 • For patients with ES-SCLC who respond to chemotherapy, consultation with a radiation oncologist to enhance shared decision making on PCI versus MRI surveillance (considering patient- and disease-specific characteristics) is recommended. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org Evidence Quality Strength of Recommendation Moderate Strong

20 Summary of Recommendations Recommendation 3. 7 • For patients with ES-SCLC who elect PCI, 2, 500 c. Gy in 10 fractions or 2, 000 c. Gy in 5 fractions is recommended. Evidence Quality Strength of Recommendation Moderate Strong Clinical Question 4 • What are the indications, appropriate dose-fractionation schedules, and timing of thoracic consolidation in patients with ES-SCLC? www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

21 Summary of Recommendations Recommendation 4. 1 • For patients with ES-SCLC with a response to chemotherapy alone but residual tumor in the thorax, thoracic RT is recommended. Recommendation 4. 2 Evidence Quality Strength of Recommendation High Strong Evidence Quality Strength of Recommendation Moderate Conditional • For patients with ES-SCLC with a response to chemotherapy alone, thoracic RT to a dose of 3, 000 c. Gy in 10 fractions is conditionally recommended • Implementation Remark: In patients expected to have a prolonged survival, higher doses may be appropriate www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

22 Summary of Recommendations Recommendation 4. 3 • For patients with ES-SCLC who will receive thoracic RT, the treatment should be given after completion of chemotherapy alone. Recommendation 4. 4 • For patients with ES-SCLC with a response to chemotherapy and immunotherapy and residual disease in the thorax, thoracic RT to 3, 000 c. Gy in 10 fractions within 6 -8 weeks (ASCO clarifying comment: of completion of the chemotherapy prior to maintenance immunotherapy) is conditionally recommended. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org Evidence Quality Strength of Recommendation High Strong Evidence Quality Strength of Recommendation Expert Opinion Conditional

3 Discussion

24 Role of SBRT in Stage I or II Node-Negative SCLC • Although randomized data demonstrate that SBRT is more effective than fractionated RT in patients with early-stage NSCLC who are ineligible for surgery, 2 no such data exist for SCLC. • Nonetheless, the use of SBRT is increasing in patients with SCLC who either are not candidates for resection or elect to forgo surgery. 3 • There is now limited retrospective data to support that this is a safe and effective approach. • In selected patients with good performance status who do receive SBRT, the addition of chemotherapy is likely to decrease the risk of metastatic failure and may improve survival. • Because SBRT is delivered using a short schedule, it can typically be given as initial therapy with chemotherapy sequentially afterward, without significant delay. If the chemotherapy is delivered first, SBRT should be integrated as early as feasible between cycles, due to concern that early tumor response may make localization more difficult. • There is very limited data on the safety of delivering SBRT and chemotherapy concurrently, and it is not currently recommended. 4 www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

25 Prophylactic Cranial RT • The ASCO Expert Panel agrees with ASTRO recommendation for shared decision making around PCI for ES-SCLC, given an alternative to PCI exists in MRI surveillance, 5 and this approach has been shown to be effective in a randomized trial. • However, PCI should remain the standard of care in LS-SCLC, until further data are available that show MRI surveillance in lieu of PCI does not compromise overall survival (OS), given that cure is possible in limited-stage disease. • The panel agrees that there are individual cases of LS-SCLC where the risks of neurocognitive toxicity may outweigh the potential benefit to PCI, particularly in patients with marginal performance status or baseline neurologic deficits. • These cases should be considered on a case-by-case basis, with shared decision making employed. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

26 Thoracic Consolidation for ES-SCLC • There have been three randomized trials that have addressed whether the addition of consolidative thoracic radiation (c. TRT) is beneficial in the setting of ES-SCLC. 6 -8 There have since been two meta-analyses evaluating the role of c. TRT in ES-SCLC, one that analyzed two trials, supporting a PFS and OS benefit. Another analyzed all three of these trials concluded that there was a PFS but no OS benefit. 9, 10 • As the two largest trials support an OS benefit and low toxicity, ASCO endorses the recommendation for radiation to the thorax in patients with a response to chemotherapy in ESSCLC. c. TRT should be delivered after completion of induction chemotherapy. The dose of 30 Gy/10 fractions is a conditional recommendation, and ASCO supports consideration of a higher dose (45 -54 Gy) if the patient is expected to have prolonged survival. • Until ongoing studies report outcomes, the use of c. TRT remains a conditional recommendation when immune checkpoint therapy is included in the initial systemic therapy regimen. The dose of radiation is unknown in this setting, and the ASTRO guidelines suggest 30 Gy/10 fractions given its low toxicity but ASCO acknowledges that this is purely an expert consensus. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

27 Endorsement Statement ASCO endorses the Radiation Therapy for Small-Cell Lung Cancer: An ASTRO Clinical Practice Guideline, with a few clarifying comments. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

28 Reprint Permission This is an endorsement of Simone CB et al: Radiation Therapy for Small Cell Lung Cancer: An ASTRO Clinical Practice Guideline, Practical Radiation Oncology, 2020, 10(3): 158 -173 by permission of Elsevier on behalf of the American Society for Radiation Oncology www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

29 Additional Resources • More information, including a supplement and clinical tools and resources, is available at www. asco. org/thoracic-cancerguidelines • The original ASTRO guideline is available at www. astro. org/Patient-Care-and-Research/Clinical-Practice. Statements/Small-Cell-Lung-Cancer-Guideline • Patient information is available at www. cancer. net www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

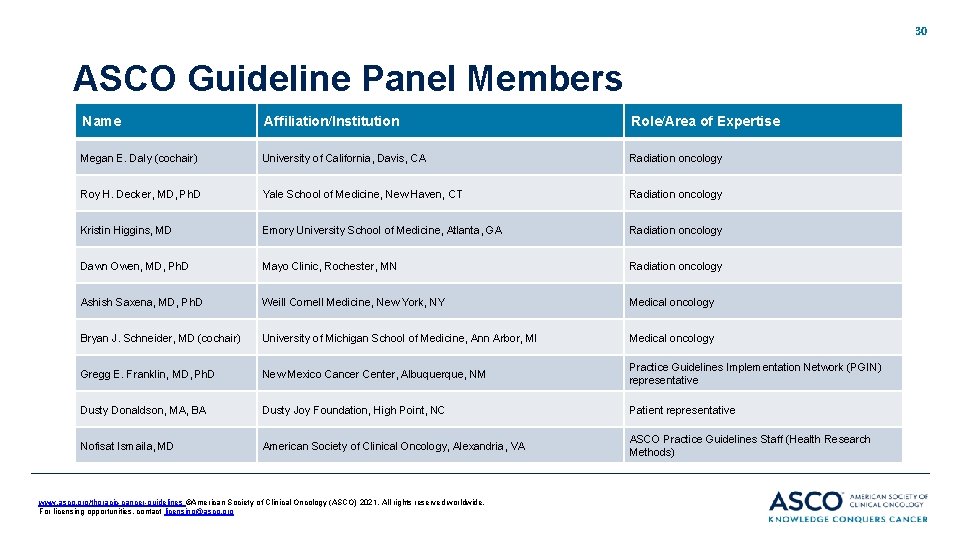
30 ASCO Guideline Panel Members Name Affiliation/Institution Role/Area of Expertise Megan E. Daly (cochair) University of California, Davis, CA Radiation oncology Roy H. Decker, MD, Ph. D Yale School of Medicine, New Haven, CT Radiation oncology Kristin Higgins, MD Emory University School of Medicine, Atlanta, GA Radiation oncology Dawn Owen, MD, Ph. D Mayo Clinic, Rochester, MN Radiation oncology Ashish Saxena, MD, Ph. D Weill Cornell Medicine, New York, NY Medical oncology Bryan J. Schneider, MD (cochair) University of Michigan School of Medicine, Ann Arbor, MI Medical oncology Gregg E. Franklin, MD, Ph. D New Mexico Cancer Center, Albuquerque, NM Practice Guidelines Implementation Network (PGIN) representative Dusty Donaldson, MA, BA Dusty Joy Foundation, High Point, NC Patient representative Nofisat Ismaila, MD American Society of Clinical Oncology, Alexandria, VA ASCO Practice Guidelines Staff (Health Research Methods) www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

31 References 1. Simone CB II, Bogart JA, Cabrera AR, et al: Radiation therapy for small cell lung cancer: An ASTRO clinical practice guideline. Pract Radiat Oncol 10: 158 -173, 2020 2. Ball D, Mai GT, Vinod S, et al: Stereotactic ablative radiotherapy versus standard radiotherapy in stage 1 non-small-cell lung cancer (TROG 09. 02 CHISEL): A phase 3, open-label, randomised controlled trial. Lancet Oncol 20: 494 -503, 2019 3. Stahl JM, Corso CD, Verma V, et al: Trends in stereotactic body radiation therapy for stage I small cell lung cancer. Lung Cancer 103: 11 -16, 2017 4. Li C, Xiong Y, Zhou Z, et al: Stereotactic body radiotherapy with concurrent chemotherapy extends survival of patients with limited stage small cell lung cancer: A single-center prospective phase II study. Med Oncol 31: 369, 2014 5. Schneider BJ, Ismaila N, Aerts J, et al: Lung cancer surveillance after definitive curative-intent therapy: ASCO guideline. J Clin Oncol 38: 753 -766, 2020 6. Jeremic B, Shibamoto Y, Nikolic N, et al: Role of radiation therapy in the combined-modality treatment of patients with extensive disease small-cell lung cancer: A randomized study. J Clin Oncol 17: 2092 -2099, 1999 7. Slotman BJ, van Tinteren H, Praag JO, et al: Use of thoracic radiotherapy for extensive stage small-cell lung cancer: A phase 3 randomised controlled trial. Lancet 385: 36 -42, 2015 8. Gore EM, Hu C, Sun AY, et al: Randomized phase II study comparing prophylactic cranial irradiation alone to prophylactic cranial irradiation and consolidative extracranial irradiation for extensive-disease small cell lung cancer (ED SCLC): NRG Oncology RTOG 0937. J Thorac Oncol 12: 15611570, 2017 9. Rathod S, Jeremic B, Dubey A, et al: Role of thoracic consolidation radiation in extensive stage small cell lung cancer: A systematic review and metaanalysis of randomised controlled trials. Eur J Cancer 110: 110 -119, 2019 10. Palma DA, Warner A, Louie AV, et al: Thoracic radiotherapy for extensive stage small-cell lung cancer: A meta-analysis. Clin Lung Cancer 17: 239 -244, 2016 www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org

32 Disclaimer The Clinical Practice Guidelines and other guidance published herein are provided by the American Society of Clinical Oncology, Inc. (ASCO) to assist providers in clinical decision making. The information herein should not be relied upon as being complete or accurate, nor should it be considered as inclusive of all proper treatments or methods of care or as a statement of the standard of care. With the rapid development of scientific knowledge, new evidence may emerge between the time information is developed and when it is published or read. The information is not continually updated and may not reflect the most recent evidence. The information addresses only the topics specifically identified therein and is not applicable to other interventions, diseases, or stages of diseases. This information does not mandate any particular course of medical care. Further, the information is not intended to substitute for the independent professional judgment of the treating provider, as the information does not account for individual variation among patients. Recommendations reflect high, moderate, or low confidence that the recommendation reflects the net effect of a given course of action. The use of words like “must, ” “must not, ” “should, ” and “should not” indicates that a course of action is recommended or not recommended for either most or many patients, but there is latitude for the treating physician to select other courses of action in individual cases. In all cases, the selected course of action should be considered by the treating provider in the context of treating the individual patient. Use of the information is voluntary. ASCO provides this information on an “as is” basis and makes no warranty, express or implied, regarding the information. ASCO specifically disclaims any warranties of merchantability or fitness for a particular use or purpose. ASCO assumes no responsibility for any injury or damage to persons or property arising out of or related to any use of this information, or for any errors or omissions. www. asco. org/thoracic-cancer-guidelines ©American Society of Clinical Oncology (ASCO) 2021. All rights reserved worldwide. For licensing opportunities, contact licensing@asco. org