Radiation Fundamental Concepts EGR 4345 Heat Transfer Thermal





























- Slides: 33

Radiation Fundamental Concepts EGR 4345 Heat Transfer

Thermal Radiation l l Occurs in solids, liquids, and gases Occurs at the speed of light Has no attenuation in a vacuum Can occur between two bodies with a colder medium in between

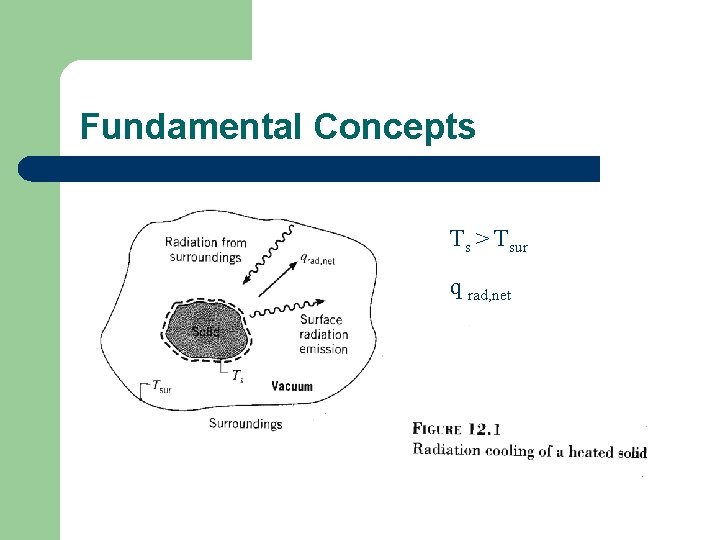
Fundamental Concepts Ts > Tsur q rad, net

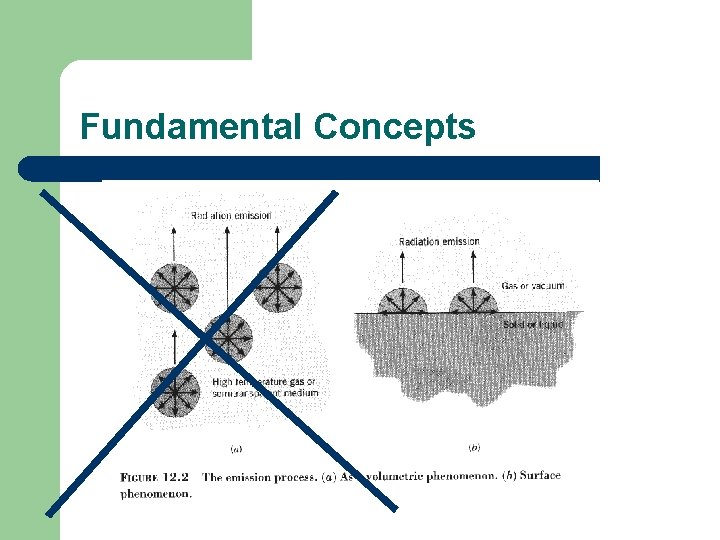
Types of Radiation l Two categories – – l l Volumetric phenomenon – gases, transparent solids Surface phenomenon – most solids and liquids Thermal radiation – emitted by all substances above absolute zero Includes visible & infrared radiation & some UV radiation.

Fundamental Concepts

Background l Electromagnetic radiation – energy emitted due to changes in electronic configurations of atoms or molecules l where =wavelength (usually in mm), n=frequency In a vacuum c=co=2. 998 x 108 m/s Other media: c= co /n where n=index of refraction l l

Background, cont. l l Radiation – photons or waves? Max Planck (1900): each photon has an energy of h=Planck’s constant=6. 625 x 10 -34 Js Shorter wavelengths have higher energy

Radiation Spectrum

Radiation Properties l l l Magnitude of radiation varies with wavelength – it’s spectral. Radiation is made up of a continuous, nonuniform distribution of monochromatic (single-wavelength) components. Magnitude & spectral distribution vary with temp & type of emitting surface.

Emission Variation with Wavelength

Radiation Properties l Directional distribution – a surface doesn’t emit the same in all directions.

Nomenclature

Nomenclature

Nomenclature

Spectral Intensity l Spectral Intensity of the Emitted Radiation, I , e – Rate at which radiant energy is emitted at the wavelength, , in the ( , ) direction, per unit solid angle about this direction, and per unit wavelength interval d about

Spectral Intensity

Spectral Intensity

Heat Flux

Emissive Power l Spectral, hemispherical emissive power – E – – – l Rate at which radiation of wavelength is emitted in all directions from a surface per unit wavelength d about and per unit surface area Flux based on actual surface area (not projected) Hemispherical often not used as emission is in all directions from surface Total emissive power, E

Emissive Power

Emissive Power l Diffuse emitter – intensity of the emitted radiation independent of direction

Spectral Irradiation l Spectral Irradiation, G – l Rate at which radiation of wavelength is incident on a surface per unit area of the surface and per unit wavelength d about Total Irradiation, G

Spectral Irradiation

Spectral Radiosity l Spectral Radiosity, J – l Rate at which radiation of wavelength is leaves a unit area of surface, per unit wavelength interval d about Total Radiosity, J

Spectral Radiosity

Blackbody Radiation l l Blackbody – a perfect emitter & absorber of radiation Emits radiation uniformly in all directions – no directional distribution – it’s diffuse Joseph Stefan (1879)– total radiation emission per unit time & area over all wavelengths and in all directions: s=Stefan-Boltzmann constant =5. 67 x 10 -8 W/m 2 K 4

Planck’s Distribution Law l l Sometimes we’re interested in radiation at a certain wavelength Spectral blackbody emissive power (Eb ) = “amount of radiation energy emitted by a blackbody at an absolute temperature T per unit time, per unit surface area, and per unit wavelength about the wavelength . ”

Planck’s Distribution Law l l Emitted radiation varies continuously with At any the magnitude of the emitted radiation increases with increasing temperature The spectral region in which the radiation is concentrated depends on temperature, with comparatively more radiation appearing at the shorter as the temperature increases Sun – approximated as a blackbody at 5800 K, radiation is mostly in the visible region

Planck’s Distribution Law l For a surface in a vacuum or gas l Other media: replace C 1 with C 1/n 2 Integrating this function over all gives us the equation for Eb. l

Radiation Distribution l l Radiation is a continuous function of wavelength Radiation increases with temp. At higher temps, more radiation is at shorter wavelengths. Solar radiation peak is in the visible range.

Wien’s Displacement Law l Peak can be found for different temps using Wien’s Displacement Law: l Note that color is a function of absorption & reflection, not emission

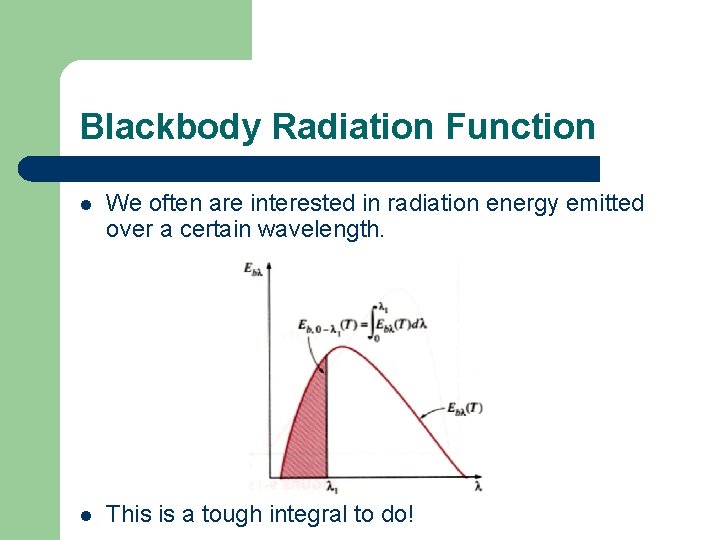
Blackbody Radiation Function l We often are interested in radiation energy emitted over a certain wavelength. l This is a tough integral to do!

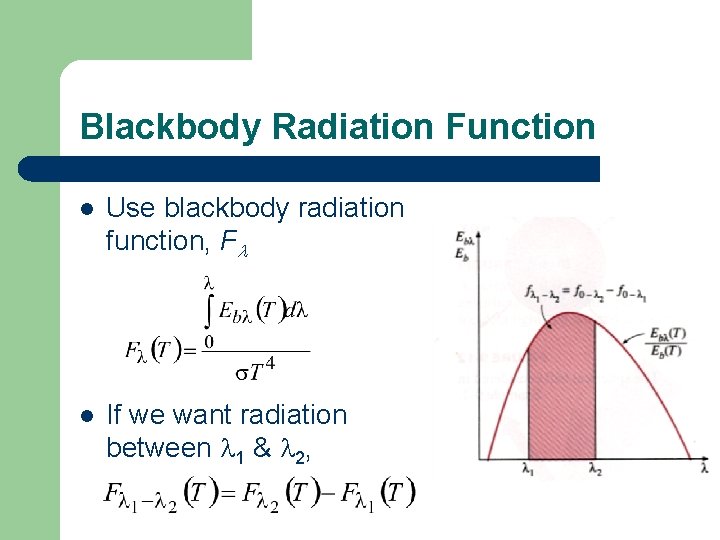
Blackbody Radiation Function l Use blackbody radiation function, Fl l If we want radiation between 1 & 2,