RADIATION AWARENESS in Nevada Jon Bakkedahl N Nevada

RADIATION AWARENESS in Nevada Jon Bakkedahl N Nevada Radiation Control Program 775. 687 -7550 (main) 1 -877 -GETRAD 1 jbakkedahl@health. nv. gov

Radiation Control Program Nevada Division of Public and Behavioral Health n THE State Agency for Radiation Protection ¨ (NRS 459) n n n Public Health Public Safety National Security

In 1895, X-Rays discovered by Wilhelm Roentgen n Stage set for the use of radiation n Studies provide a detailed understanding of hazards and benefits of radiation n

In 1896, Henri Becquerel discovered radiation from uranium ore n Becquerel was studying a body's exposure to light of another color. This new found light was given the name “x-ray” for being unknown n

In the same year, Marie and Pierre Curie discovered radium and polonium were radioactive elements in the same ore n Credited with the world's first studies conducted into the treatment of cancer using radioactive isotopes. n

Radiation Awareness Review Identify types of radiation n Identify terminology and risks associated with radiation n Identify self aid procedures for protection against radiation exposure n understand the need for detection equipment n

Radiation Control Program n Ionizing Radiation ¨ unstable ¨ Physical change in atoms by making them electrically charged n Non-Ionizing Radiation ¨ Visible light/heat/radio waves/microwaves ¨ Does not have sufficient energy to cause ionization

Ionizing Radiation? n Ionizing radiation is electromagnetic energy or energetic particles emitted from a source ¨ Electromagnetic energy: radio waves, light, x- rays, etc. ¨ Source: unstable atoms of any material ¨ Ionize: To strip electrons from other atoms causing chemical changes in molecules

Four Basic Types of Ionizing Radiation n Alpha n Beta n Gamma/X-ray n Neutron

Alpha Radiation Heavy charged particles n Not penetrating n ¨ Travel centimeters in air or a few microns in tissue ¨ Stopped by paper or clothing n Internal hazard

Beta Radiation High energy small particle n Moderately penetrating n ¨ Up to a few meters in air ¨ Several millimeters in tissue n Primarily internal hazard, some external

Gamma Radiation & X-rays High and Low energy rays (photons) n Very penetrating n Difficult to shield n Protective clothing will not protect against photon radiation n External and internal hazard n

Neutron Radiation Uncharged high speed particle n Can be very penetrating n Requires special consideration for shielding n External and internal hazard not likely to encounter dangerous levels of neutron radiation n

Examples of Radioactive Materials Material Emits Use Americium 241 a, g Smoke detectors, density gauges Cobalt 60 g, b Medical therapy, Industrial Radiography Cesium 137 g, b Many industrial uses Radium 226 g, b, a Medical therapy, Dials Uranium 238 g, b, a Reactors and weapons Iridium 192 g, b Industrial radiography Technetium 99 m g Worldwide Medical

Radioactive Material and Radioactivity n Radioactive decay is measured in halflives Half-life is unique to each radioactive isotope and can vary greatly n Radioactive pharmaceutical products (called radiopharmaceuticals) typically have half-lives of a few hours or days n

Radiation Half-life n Time required for a radioactive substance to lose half of its radioactivity ¨ Each radionuclide has a unique half-life ¨ Fraction of a second to millions of years Examples: N-13 Tc-99 m I-131 Ir-192 Co-60 Cs-137 Am-241 Pu-239 U-238 ~10 mins ~6 hrs ~8 days Radioactive ~73 days 10 half-lives 198765432 half-life ~5 years Material ~30 years ~432 years ~24, 400 years ~4, 150, 000 years

Progeny Each radioactive isotope decays to something else n Progeny (daughters) might be more dangerous than parent n You might be detecting progeny n Example: Cs-137 b = Ba-137 m (2. 55 min ½ life) g


Units of Measure n Traditional units of measure and International System of Units (SI) are used in measuring radiation and radioactivity n For radiation measurement: Exposure Absorbed Dose Equivalent Common Units roentgen (R) rad rem SI Units coulomb/kilogram (C/kg) gray (Gy) sievert (Sv)

Units of Measure (Cont. ) -Traditional units of curies (Ci) 1 Ci = that quantity of radioactive material in which 37 billion atoms disintegrate per second -SI units of Becquerel (Bq) 1 Bq = that quantity of radioactive material in which 1 atom disintegrates per second
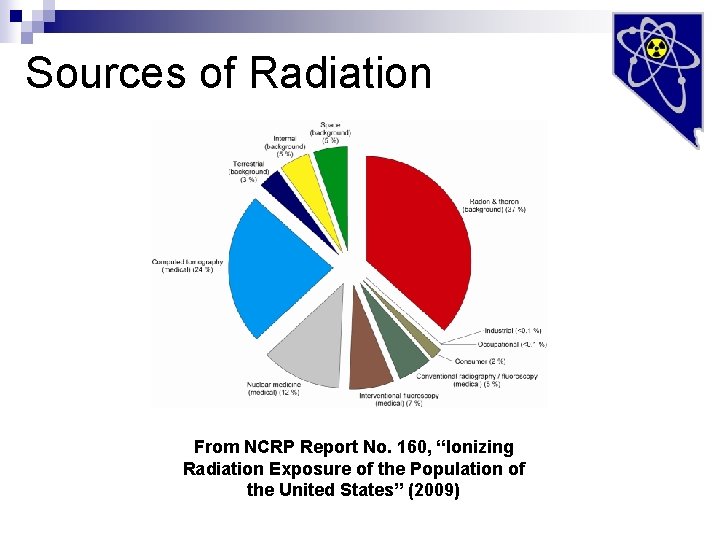
Sources of Radiation From NCRP Report No. 160, “Ionizing Radiation Exposure of the Population of the United States” (2009)

Radiation Doses In Perspective n Natural background and manmade radiation 360 -500 mrem/yr n (~20 +/- μR/h is background) n Diagnostic chest x-ray 10 mrem n Flight from LA to Paris 4. 8 mrem n Barium enema 800 mrem n Smoking 1. 5 packs per day 16, 000 mrem/yr n Heart catheterization 45, 000 mrem n Mild acute radiation sickness 200, 000 mrem n LD 50/30 for radiation 450, 000 mrem

Acute vs Chronic Dose n Acute - Large dose/short time (300 rem/hr) ¨ May cause early effects ¨ Bomb victims; radiography accidents n Chronic - Small doses over time ¨ May cause delayed effects ¨ Possible for workers ¨ Uranium Miners
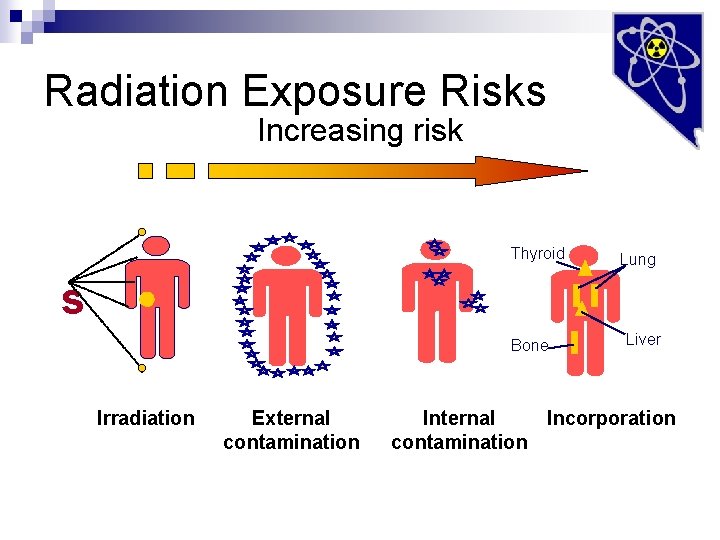
Radiation Exposure Risks Increasing risk Thyroid s Lung · Bone Irradiation External contamination Liver Internal Incorporation contamination

Radiation Control Program n Health Physics ¨ The effects of radiation on the body n Radiation Protection ¨ Minimizing unwanted radiation exposure n ALARA time; distance; shielding ¨ PPE ¨

Time Source Result Dose 25 mrem 100 mrem per hour x 15 minutes (. 25 hour) = 25 mrem
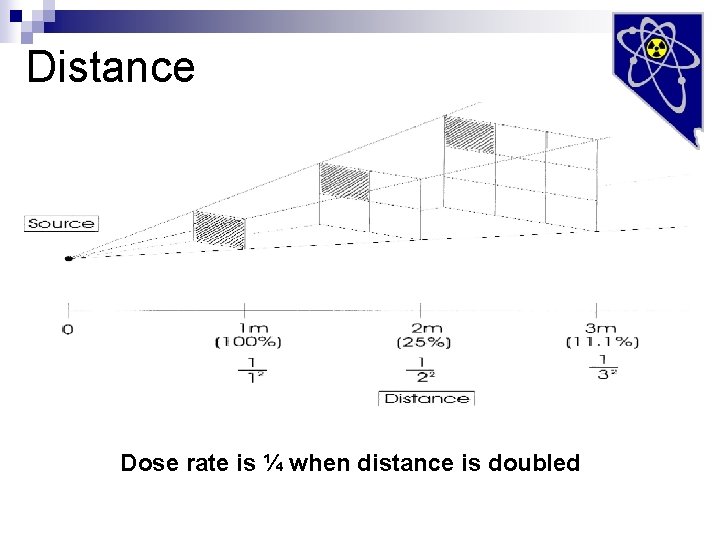
Distance Dose rate is ¼ when distance is doubled
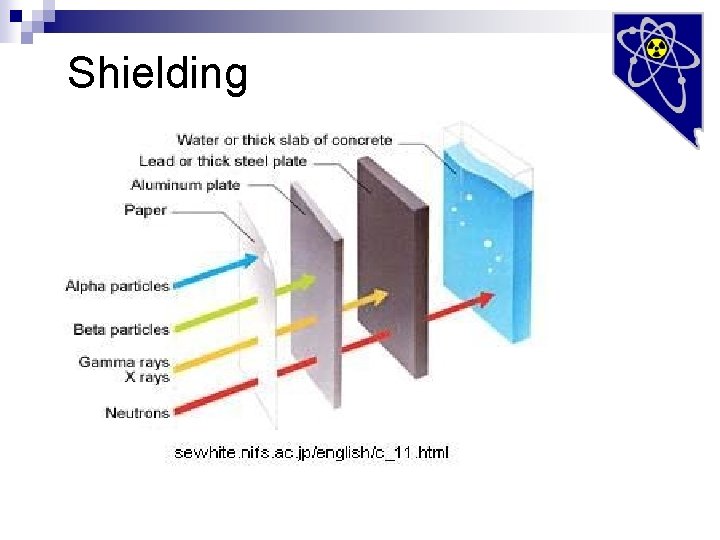
Shielding

Basic Radiation PPE n Protect your respiratory tract ¨ Respirator, surgical mask, etc. n Protect your skin ¨ Gloves! ¨ Outer clothing ¨ Chemical suit is not always needed n Downgrade every chance you get! ¨ If radiation is the only hazard, longer time downrange caused by bulky PPE could be a greater hazard than contamination (ALARA)

Radioactivity –Hazardous? 3 Things you should know to find out what kind of hazards are present: n What type of radiation is emitted? ¨ Alpha, beta, or gamma Half-Life – long or short? n Solid or Dispersible? n

Detecting Ionizing Radiation • Not detected by human senses • Requires use of detection instruments • No single instrument can detect or measure all types of radiation

Detection Instruments n Dosimeter Track total personal Dose (exposure) ¨ Self Reading (pocket ion chamber); Lab (TLD, OSLD) ¨ n Pager ¨ ¨ n Personal Radiation Detector (PRD, PRM) ¨ ¨ n Detects only (yes/no), poor measurement ability Very Sensitive Real time dose and dose rate pocket, belt clip size Survey instrument Detects and measures with a variety of probes Find source of radiation ¨ Find surface contamination ¨ ¨ n RIID (Radio-Isotope Identifier) ¨ Detects and Identifies Gamma radiation sources

Geiger-Mueller (GM) n Pancake n Side/end Window n Energy Compensated

Scintillators n Crystal ¨ Gamma n Plastic ¨ Gamma/Beta n Zinc-Sulfide ¨ Alpha

Ion Chambers n Pressurized n Non Pressurized n Beta Window

Remote Handled

Waste Isolation Pilot Plant – WIPP US Department of Energy Transuranic Waste (TRU) Isotopes Heavier Than Uranium Generated by Weapons Labs High Alpha Component Inhalation Hazard 40 shipments/year across I-80 Beginning 2010

Industrial Radiography

Portable Density Gauges

Radiopharmaceutical packages

Radon potential based on actual data since 1989 26% radon potential All homes should be tested Elevated radon concentrations are found everywhere

National Research Council conclusions: n Serious public health problem n 2 nd leading cause of lung cancer n No evidence of a threshold n Effects of radon & smoking is sub- multiplicative in combination
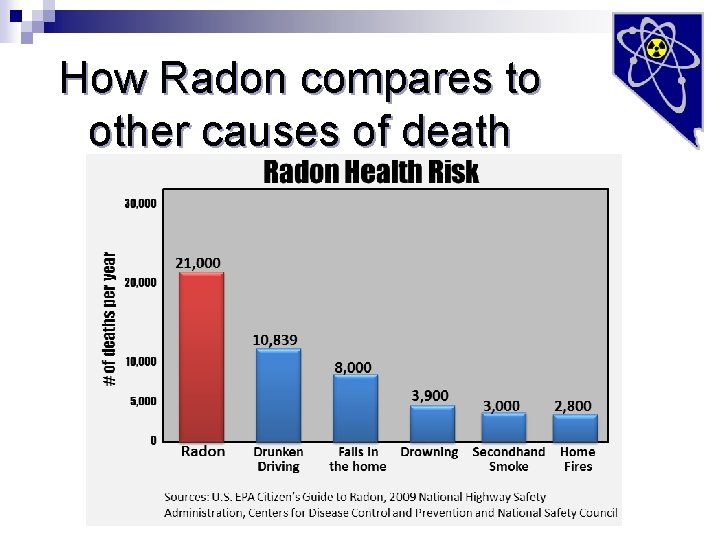
How Radon compares to other causes of death

Radon is a global public health concern n Radon is a problem in Nevada n Radon is the leading environmental cause of cancer mortality in the U. S. and 8 th leading cause of cancer mortality overall. n Radon is the leading cause of lung cancer for nonsmokers n Your help is critical in getting the word out n ¨ http: //Breathing. Easier. info ¨ https: //www. youtube. com/watch? v=AQLSStm_ jwk

CONTROL THE SCENE AND ESTABLISH “SAFE” AREAS • Recommended values to be used when Radiation Control staff are not yet at the site and responders have limited instrumentation THESE ARE RECOMMENDATIONS ! • Responders should follow their department’s SOP’s and consult with state Radiation Control program.
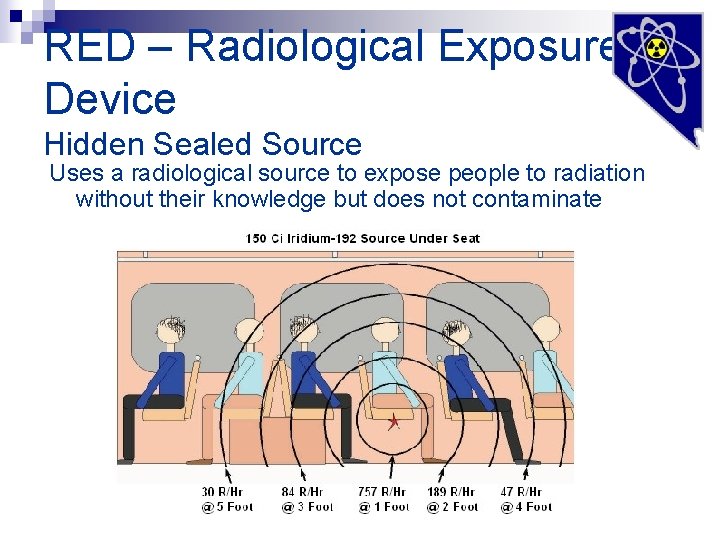
RED – Radiological Exposure Device Hidden Sealed Source Uses a radiological source to expose people to radiation without their knowledge but does not contaminate

RDD - Radiological Dispersal Device (Dirty Bomb) Uses explosive or mechanical means to disburse radioactive material Weapon of Mass Disruption Trauma from explosion could kill Exposure to radiation could result in cancer RDDs planted by Chechen rebels were found and disarmed in 1995 and 1998

RDD Not Always Explosive Just Has To Disburse
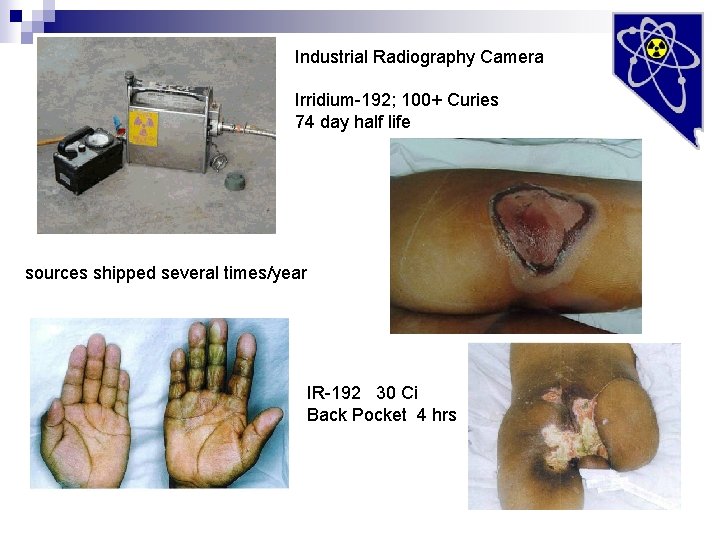
Industrial Radiography Camera Irridium-192; 100+ Curies 74 day half life sources shipped several times/year IR-192 30 Ci Back Pocket 4 hrs
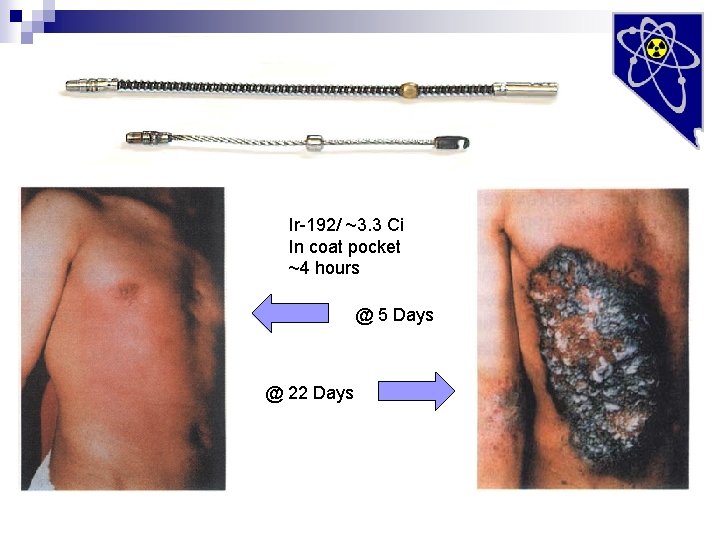
Ir-192/ ~3. 3 Ci In coat pocket ~4 hours @ 5 Days @ 22 Days
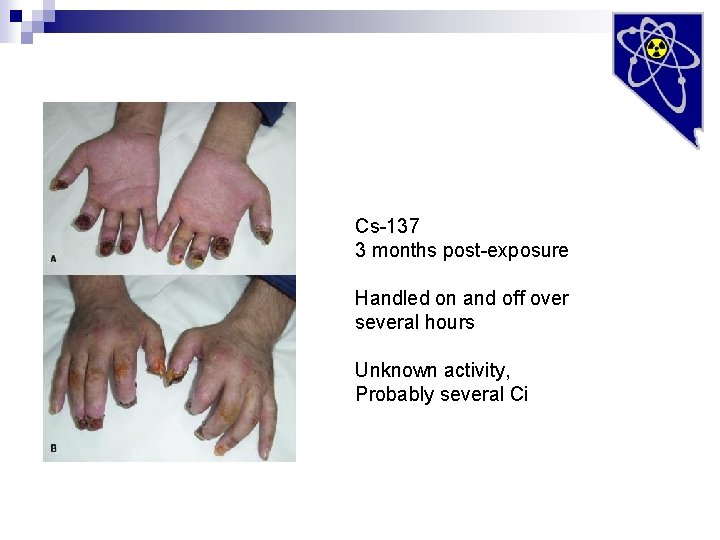
Cs-137 3 months post-exposure Handled on and off over several hours Unknown activity, Probably several Ci
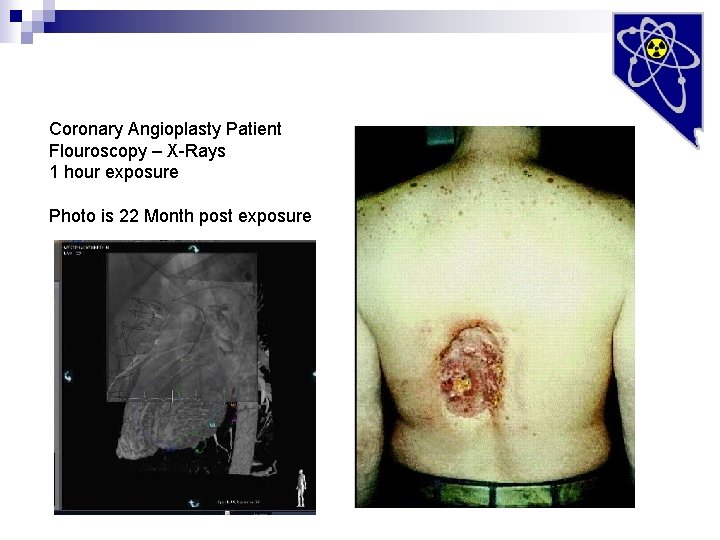
Coronary Angioplasty Patient Flouroscopy – X-Rays 1 hour exposure Photo is 22 Month post exposure

Poison (exposure or disbursal) Alexander Litvinenko Poisoned with Polonium-210 Died 11/23/2006 1 gram of Po-210 = 4500 Ci 3. 5 micrograms is fatal 166 of 596 people tested in Britain had probable contact with Po-210 with 13 requiring further monitoring

? Nevada Radiological Incident Response 24 -hour duty officer 1 -877 -GET RAD 1 (438 -7231) DPS Dispatch. . . 775 -687 -0400
- Slides: 54