Quiz Combustion Synthesis Reaction Decomposition reaction Single replacement

Quiz Combustion, Synthesis Reaction, Decomposition reaction, Single replacement, Double replacement 1. Identify Type: 2 H 2 O 2 (l) 2 H 2 O (l) + O 2 (g) 2. Identify Type: Si. O 2 (l) + 2 C (s) Si (l) + 2 CO (g) 3. Does this reaction take place? Al 2(SO 4)3 + 2 Fe 2(SO 4)3 + 2 Al 4. List all spectator ions in the following equation. Ba(NO 3)2(aq)+2 Na. SO 4(aq) Ba. SO 4(s)+2 Na. NO 3(aq)

Quiz Combustion, Synthesis Reaction, Decomposition reaction, Single replacement, Double replacement 1. Identify Type: 2 H 2 O 2 (l) 2 H 2 O (l) + O 2 (g) 2. Identify Type: Si. O 2 (l) + 2 C (s) Si (l) + 2 CO (g) 3. Does this reaction take place? Fe 2(SO 4)3 + 2 Al Al 2(SO 4)3 + 2 Fe 4. List all spectator ions in the following equation. Ba(NO 3)2(aq)+2 Na. SO 4(aq) Ba. SO 4(s)+2 Na. NO 3(aq)

5. 3 Redox reaction

• What is an ion? – Any charged atoms or molecules • How do you become ‘charged’? – By losing or gaining electrons • What is a cation? Did it lose or gain electrons? – Positive ion, it LOST electrons • What is an anion? Did it lose or gain electrons? – Negative ion, it GAINED electrons • The charge of an ion are also known as oxidation numbers

More Practice • If a metal iron, Fe, becomes an ion, Fe 3+, how many electrons did it lose or gain? • It lost 3 electrons • If a pure sulfur, S, becomes an ion, S 2 -, how many electrons did it lose or gain? • It gained 2 electrons

• Rules to find oxidation numbers 1. Neutral elements and diatomic molecules have an oxidation number of zero. Ex. Na, Fe, H 2, O 2, F 2 2. Monatomic ions have oxidation number equal to its charge. Ex, Na+ is 1, O 2 - is -2. Do you know how to use P. T to find oxidation #? 3. Oxidation numbers in polyatomic ions add up to the total charge of the ion. Ex: shown NS

Example • Sulfate is a polyatomic ion, with the formula of SO 42 • The oxidation number of sulfur and four oxygen must add up to -2 • Since oxygen is -2, the total negative charge of it is -8 (-2 * 4) • Sulfur must have an oxidation number of +6, to balance the oxygen, for the ion to have a charge of -2

(cations first, anions second) • What are the oxidation number of the elements in a nitrate ion? • NO 3 • Oxygen is -2 • Nitrogen is 5

Redox Reactions • We have covered 5 major types of chemical reactions • Redox reaction is a totally different way to classify reactions, which can encompass all of the 5 major reactions • As long as reactants and products are exchanging electrons, it is called a redox reaction
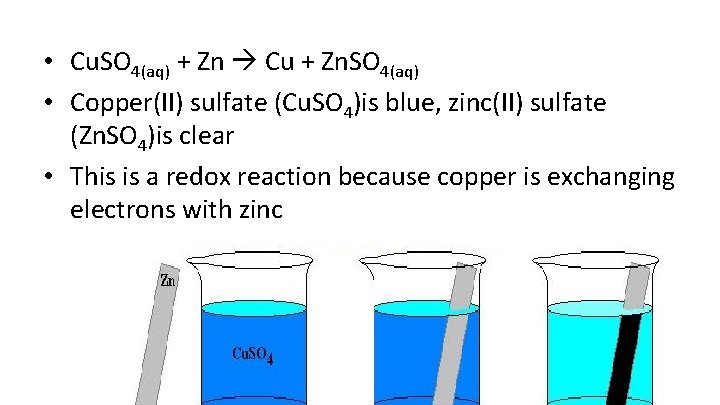
• Cu. SO 4(aq) + Zn Cu + Zn. SO 4(aq) • Copper(II) sulfate (Cu. SO 4)is blue, zinc(II) sulfate (Zn. SO 4)is clear • This is a redox reaction because copper is exchanging electrons with zinc

• Copper sulfate (Cu. SO 4) dissolves in water to form two ions, copper (II) ion and sulfate ion • Cu 2+ and SO 42 • How do we know copper is +2? • Because we memorized sulfate(SO 42 -) a charge of -2, and so copper (II) ion needs to be +2 to make Cu. SO 4 neutral • A roman numeral in parentheses represents the charge, and are needed for ALL transitional metal ions!

• The zinc metal gives the copper (II) ion (Cu 2+) two electrons • The zinc metal now becomes zinc (II) ion (Zn 2+) while copper (II) ion (Cu 2+) becomes copper metal (Cu) by gaining the two electrons • Reminder: electrons are negatively charged!

• When zinc lost the electrons (gave it away), the reaction, Zn Zn 2+ + 2 e-, is called oxidation reaction • When copper (II) ion gained the electrons, the reaction, Cu 2+ + 2 e- Cu, is called reduction reaction • Oxidation and reduction reaction happens at the same time, which we call redox reaction
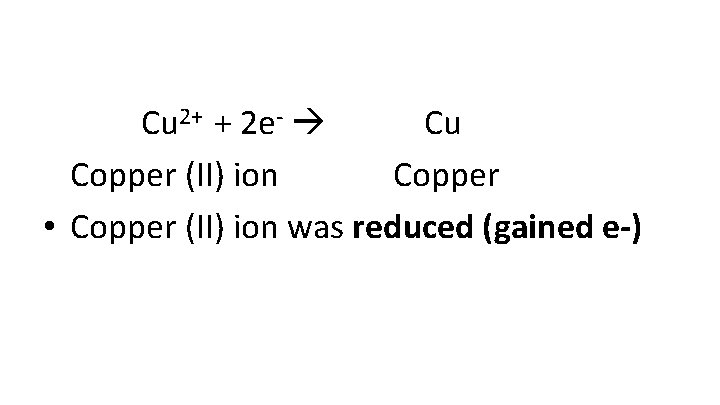
Cu 2+ + 2 e- Cu Copper (II) ion Copper • Copper (II) ion was reduced (gained e-)
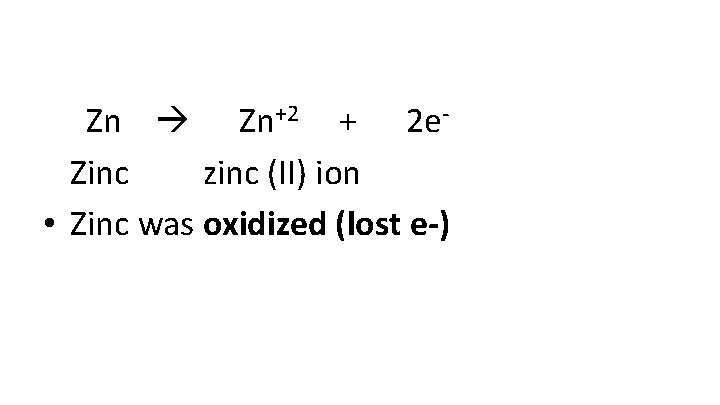
Zn Zn+2 + 2 e. Zinc zinc (II) ion • Zinc was oxidized (lost e-)

• • • LEO the lion says GER Loss of Electrons is Oxidation Gaining Electrons is Reduction OIL RIG m. Ore positive (Oxi) l. Ess positive (r. Educ)

• Aluminium LOST 3 electrons. Which one is the correct equation? A) Al + 3 e- Al 3+ B) Al 3+ + 3 e. C) Is this Oxidation or Reduction?
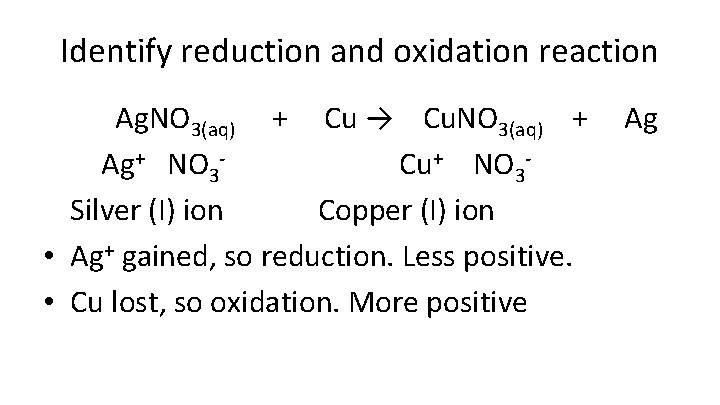
Identify reduction and oxidation reaction Ag. NO 3(aq) + Cu → Cu. NO 3(aq) + Ag Ag+ NO 3 - Cu+ NO 3 Silver (I) ion Copper (I) ion • Ag+ gained, so reduction. Less positive. • Cu lost, so oxidation. More positive

Identify reduction and oxidation reaction • 2 Na + Fe. Cl 2 → 2 Na. Cl + Fe Fe 2+ 2 Cl Na+ Cliron (II) ion sodium ion Reduction: Iron, gained e-, less positive Oxidation: Sodium, lost e-, more positive

- Slides: 20