Quiz 33005 Lab 30 tomorrow Please do prelab

Quiz 3/30/05 Lab 30 tomorrow! Please do pre-lab and summary In a group of 2 • Active metal oxides react with water to form hydroxides. ex: • 1. Na 2 O + H 2 O ____ • Halogens react with metals to form salts • 2. Mg + Cl 2 _____ • Metals react with oxygen to form oxides • 3. K + O 2 ______ • Substances may decompose when exposed to electricity: • 4. Na. Cl ____ + _____ • metal carbonates decompose to form metal oxides and carbon dioxide. • 5. Al 2(CO 3)3 ______+ _____ • metal hydroxides decompose to form metal oxides and water. • 6. Mg(OH)2 _____ + _____

Quiz 3/30/05 Answers • Active metal oxides react with water to form hydroxides. ex: • 1. Na 2 O + H 2 O 2 Na. OH_ • Halogens react with metals to form salts • 2. Mg + Cl 2 Mg. Cl 2 • Metals react with oxygen to form oxides • 3. 4 K + O 2 2 K 2 O • Substances may decompose when exposed to electricity: • 4. 2 Na. Cl 2 Na + Cl 2 • metal carbonates decompose to form metal oxides and carbon dioxide. • 5. Al 2(CO 3)3 Al 2 O 3 + 3 CO 2 • metal hydroxides decompose to form metal oxides and water. • 6. Mg(OH)2 Mg. O + H 2 O
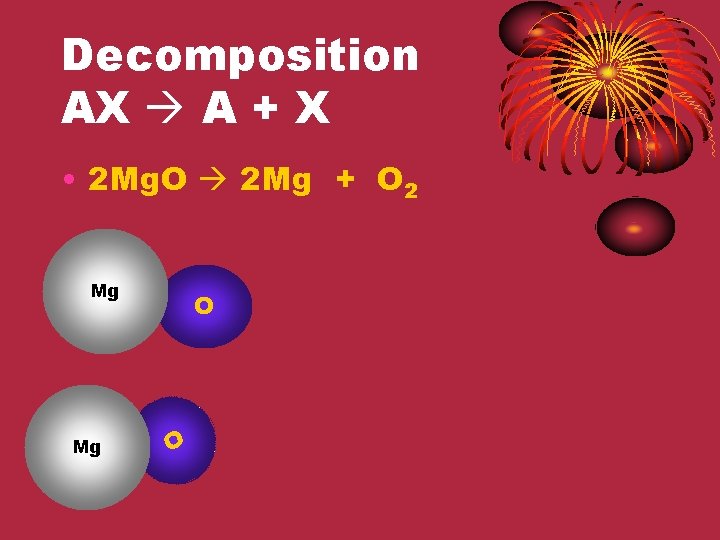
Decomposition AX A + X • 2 Mg. O 2 Mg + O 2 Mg Mg O O

Practice decomposing oxides: • Cu. O Cu + O 2 • Fe 2 O 3 Fe + O 2 • Mn. O 2 Mn + O 2 • Pb. O Pb + O 2 422 22 2 3

Decomposition of metal carbonates ACO 3 AO + CO 2 (g) • Sr. CO 3 (s) Sr. O (s) + CO 2 (g) O Sr C O O This reaction occurs when you heat strontium carbonate. strontium oxide and carbon dioxide form.

Practice decomposing carbonates: • Cu. CO 3 Cu. O + CO 2 • Na 2 CO 3 Na 2 O + CO 2 • Mg. CO 3 Mg. O + CO 2 • Ag 2 CO 3 Ag 2 O + CO 2
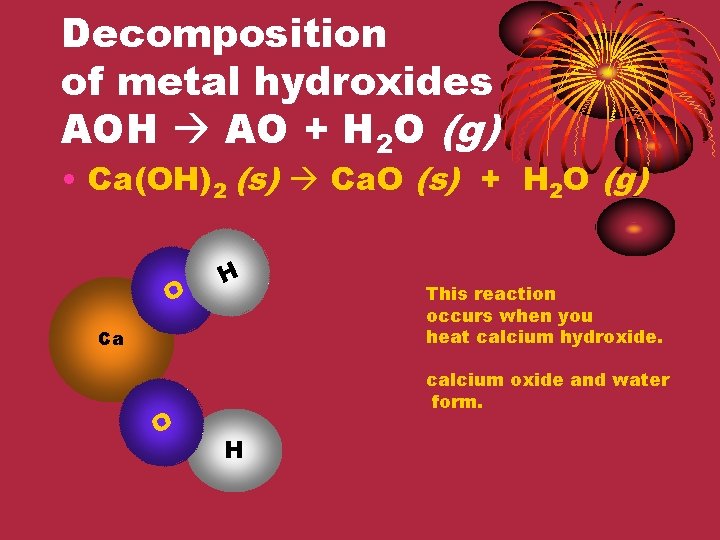
Decomposition of metal hydroxides AOH AO + H 2 O (g) • Ca(OH)2 (s) Ca. O (s) + H 2 O (g) O H Ca O This reaction occurs when you heat calcium hydroxide. calcium oxide and water form. H

Practice decomposing 23 2 hydroxides: • Na. OH Na 2 O + H 2 O • Al(OH)3 Al 2 O 3 + H 2 O • Cu(OH)2 Cu. O + H 2 O • Mg(OH)2 Mg. O + H 2 O
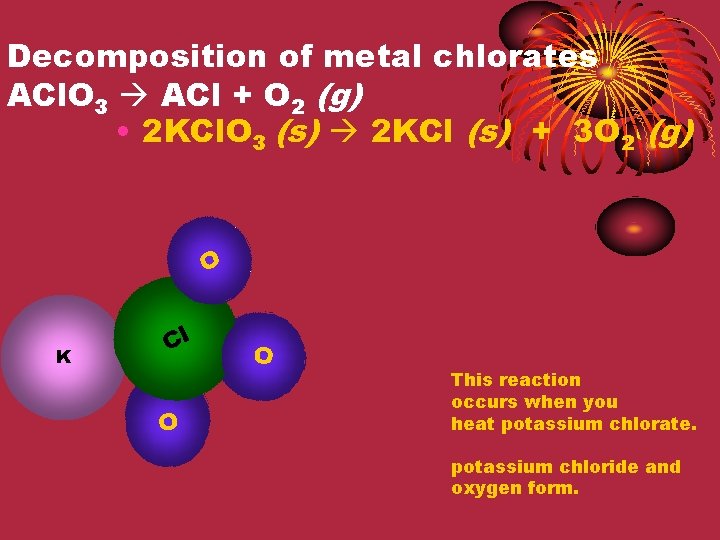
Decomposition of metal chlorates ACl. O 3 ACl + O 2 (g) • 2 KCl. O 3 (s) 2 KCl (s) + 3 O 2 (g) O Cl K Cl O O This reaction occurs when you heat potassium chlorate. potassium chloride and oxygen form.

Practice decomposing 32 2 chlorates: • HCl. O 3 HCl + • Na. Cl. O 3 Na. Cl + O 2 • Cu(Cl. O 3)2 Cu. Cl 2 + O 2 32 23
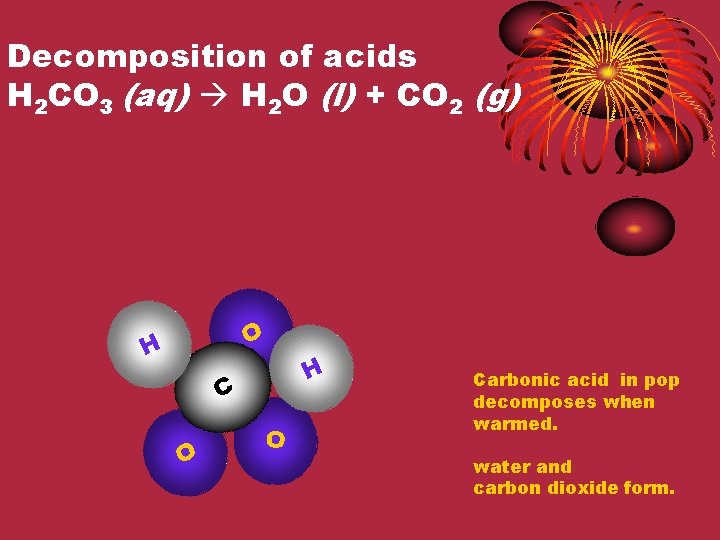
Decomposition of acids H 2 CO 3 (aq) H 2 O (l) + CO 2 (g) O H H C O O Carbonic acid in pop decomposes when warmed. water and carbon dioxide form.

Combined Practice: • 2 Ag 2 O 4 Ag + O 2 • 2 Cu. Cl. O 3 2 Cu. Cl 2 + 3 O 2 • Na 2 CO 3 Na 2 O + CO 2 • Ca(OH)2 Ca. O + H 2 O
- Slides: 12