QUIZ 1 What does UBIQUITOUS mean 2 How

QUIZ 1. What does UBIQUITOUS mean? 2. How does High Specific Heat relate to your swimming pool? 3. What is surface tension? 4. Draw the water molecule , add the charges. 5. What is adhesion, cohesion and how do they relate to capillary action? 6. What does UNIVERSAL SOLVENT mean?
p. H Notes Directions: Take notes Do Lab Review

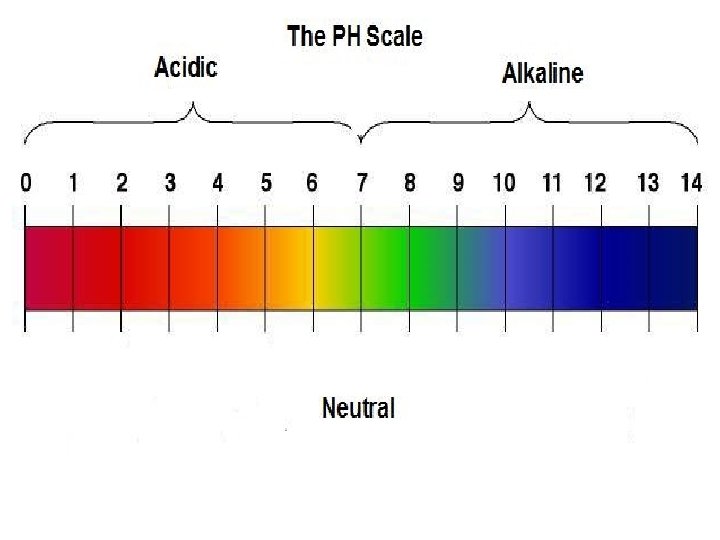
p. H PRE Quiz 1. 2. 3. 4. 5. 6. 7. What does p. H stand for? What is neutral on the scale? What liquid represents neutral? What is the highest rating on the scale? Is the highest rating an acid or base? What is the lowest rating? Is the lowest rating an acid or base?

p. H Defined • p. H is defined as the negative logarithm of the hydrogen ion concentration: p. H = - log [H+] • p. H stands for “potential for Hydrogen”

What is p. H? • The p. H scale measures how acidic or basic a substance is. – The p. H scale ranges from 0 to 14. • A p. H less than 7 is acidic. • A p. H of 7 is neutral. • A p. H greater than 7 is basic. http: //www. elmhurst. edu/~chm/vchembook/184 ph. html

? Logarithms ? • Logarithms of numbers that are multiples of ten are merely the exponents of the number including the sign. NUMBER EXPONENT LOG 1000 10³ 3 100 10² 2 10 10¹ 1 0 10⁰ 0 0. 1 10 -¹ -1 0. 01 10 -2 -2 0. 001 10 -3 -3

Soo… • The p. H scale is logarithmic and as a result, each whole p. H value ten times more or less concentrated. • For example, p. H 4 is ten times more acidic than p. H 5 and 100 times (10 times 10) more acidic than p. H 6. • For example, p. H 10 is ten times more alkaline than p. H 9 and 100 times (10 times 10) more alkaline than p. H 8.

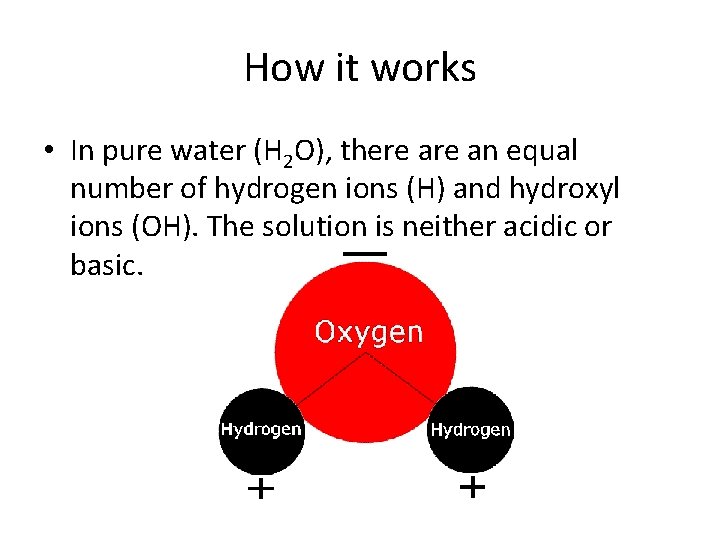
How it works • In pure water (H 2 O), there an equal number of hydrogen ions (H) and hydroxyl ions (OH). The solution is neither acidic or basic.
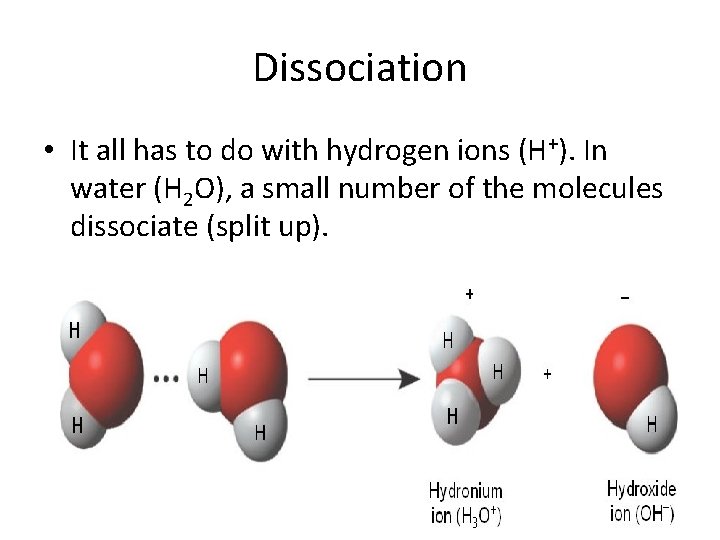
Dissociation • It all has to do with hydrogen ions (H+). In water (H 2 O), a small number of the molecules dissociate (split up).
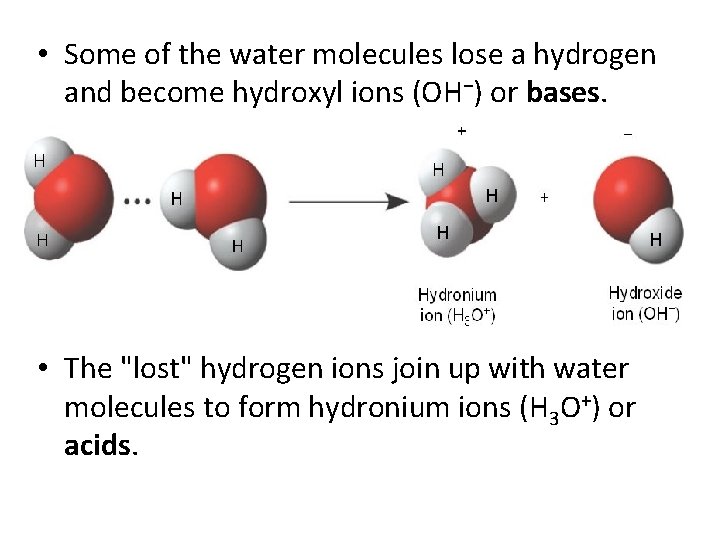
• Some of the water molecules lose a hydrogen and become hydroxyl ions (OH−) or bases. • The "lost" hydrogen ions join up with water molecules to form hydronium ions (H 3 O+) or acids.
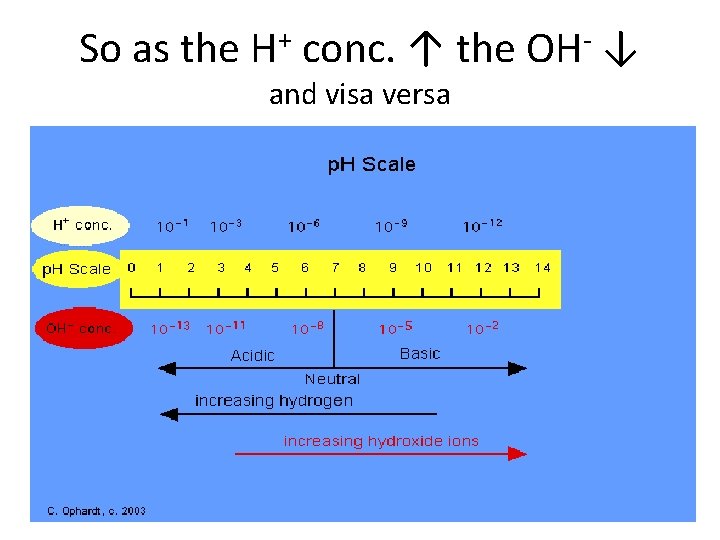
So as the H+ conc. ↑ the OH- ↓ and visa versa

1. 2. 3. 4. 5. RCC Draw and label a HYDRONIUM molecule. Draw and label HYDROXIDE molecule. Why is water neutral? What does dissociate mean? If you add acid to a p. H 3 does the p. H increase or decrease? 6. If you add acid to a p. H 10 does the p. H increase or decrease? 7. If you remove base from a p. H 9 does the p. H increase or decrease? 8. If you remove acid from a p. H 6 does the p. H increase or decrease?

Household items • p. H is measured with a p. H meter or with Litmus paper – which you used in your lab.

Properties of Acids (H+) • Sour taste (do not taste!) • Change indicator to red • React with active metals to form hydrogen gas and a metal compound • contains hydrogen • Proton donors

Properties of Bases (OH-) bitter taste (Do not taste!) slippery to the touch (DO not Touch!) change indicator to blue Emulsify (combine) fats and oils (thereby making them useful cleaning agents) • contain the OH- ion • proton acceptors. • •

When acids and bases mix… • A salt is any compound which can be derived from the neutralization of an acid and a base. • The word "neutralization" is used because the acid and base properties of H+ and OH- are destroyed or neutralized. • In the reaction, H+ and OH- combine to form HOH or H 2 O or water molecules. • See demo

Danger! • Acids and bases are examples of corrosive poisons which react locally on tissue cells. • Chemicals that are very basic or very acidic are reactive. • These chemicals can cause severe burns, causing ultimate disintegration of the tissue. • The eyes and lungs are particularly sensitive to corrosive poisons.

Life • Biological systems operate within a narrow p. H range and can be negatively impacted by a change of p. H. - Blood 7. 35 -7. 45 - Salt water fish 8. 0 and higher - Fresh water fish 5. 5 – 7. 5 - Swimming pool 7. 0 -7. 6

Conclusion…p. H is good to know • p. H AND YOU – REVIEW • CRASH COURSE • • Get HW sheet DO HW sheet Tomorrow we will CHECK HW sheet Monday we will have an assessment
- Slides: 21