Quieting Down Opioid Prescribing Maria Foy Pharm D

Quieting Down Opioid Prescribing Maria Foy, Pharm. D, BCPS, CPE Tanya J. Uritsky, Pharm. D, BCPS, CPE Pharmacy Care Coordinator/Palliative Care Clinical Pharmacy Specialist, Pain Management and Palliative Care Abington Hospital Jefferson Health Hospital of the University of Pennsylvania
Disclosures • Tanya Uritsky has nothing to disclose • Maria Foy is on the speaker’s bureau for Astra Zeneca

Goals and Objectives At the completion of this activity, the participant should be able to: § Recommend an opioid de-escalation protocol based on current opioid dosage and length of therapy § Provide dosing for inpatient ketamine during the deescalation process § Utilize co-analgesics for the treatment of chronic pain

Question Symptoms of central sensitization may include (select all that apply): 1. Allodynia 2. Hyperalgesia 3. Swelling 4. Bradycardia

Pain System Changes and Opioids Tanya J. Uritsky, Pharm. D, BCPS, CPE
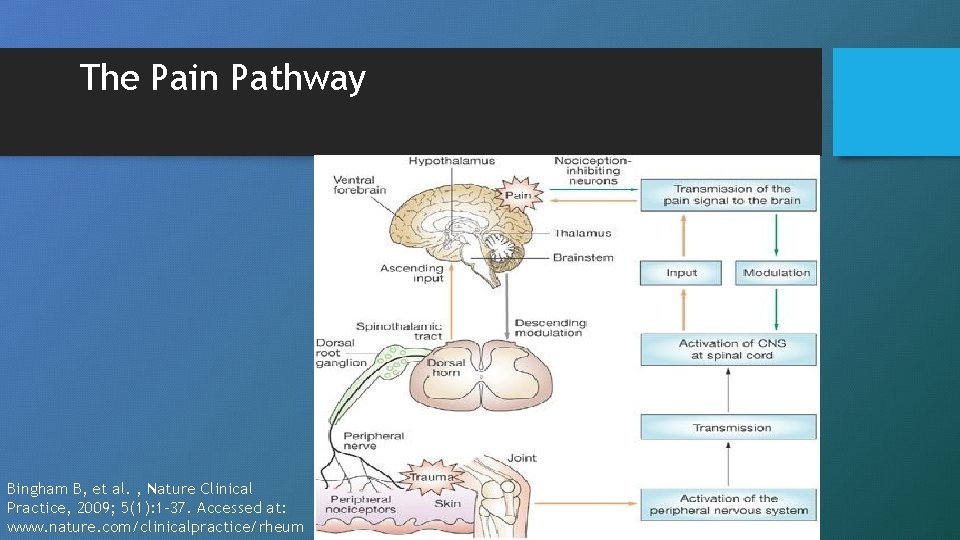
The Pain Pathway Bingham B, et al. , Nature Clinical Practice, 2009; 5(1): 1 -37. Accessed at: www. nature. com/clinicalpractice/rheum
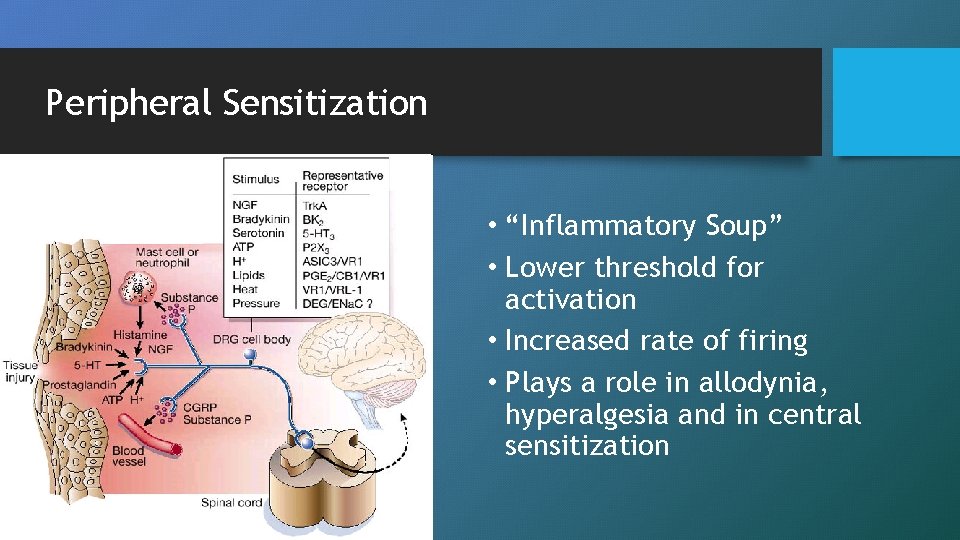
Peripheral Sensitization • “Inflammatory Soup” • Lower threshold for activation • Increased rate of firing • Plays a role in allodynia, hyperalgesia and in central sensitization

Central Sensitization • Amplification of neural signaling that elicits pain hypersensitivity • An uncoupling of the clear stimulus-response relationship that defines nociceptive pain • Manifests as: • Hyperalgesia/secondary hyperalgesia • Allodynia • Prolonged pain after transient stimulus • Can persist long after healing of the injury Woolf CJ. Pain, 2011; 152(3 Suppl): S 2 -S 15.
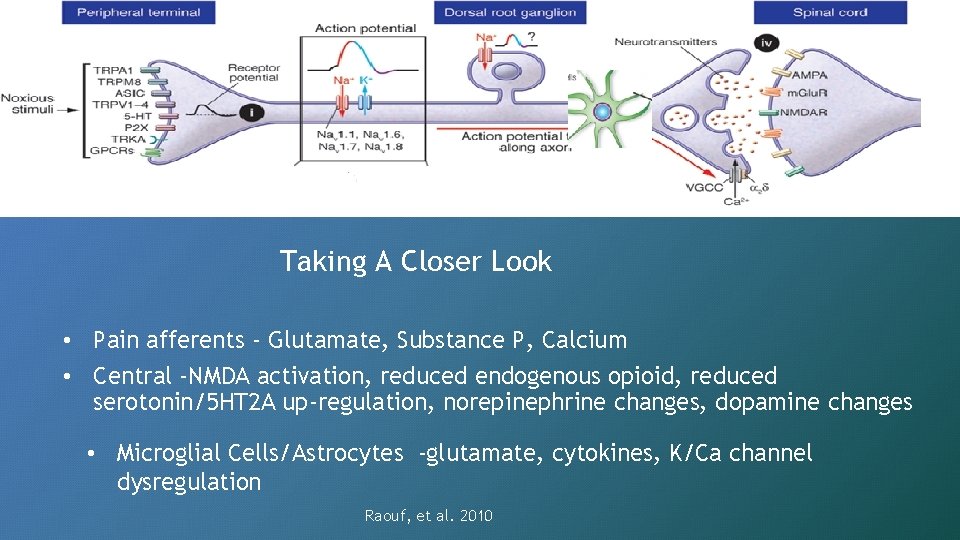
Taking A Closer Look • Pain afferents - Glutamate, Substance P, Calcium • Central -NMDA activation, reduced endogenous opioid, reduced serotonin/5 HT 2 A up-regulation, norepinephrine changes, dopamine changes • Microglial Cells/Astrocytes -glutamate, cytokines, K/Ca channel dysregulation Raouf, et al. 2010
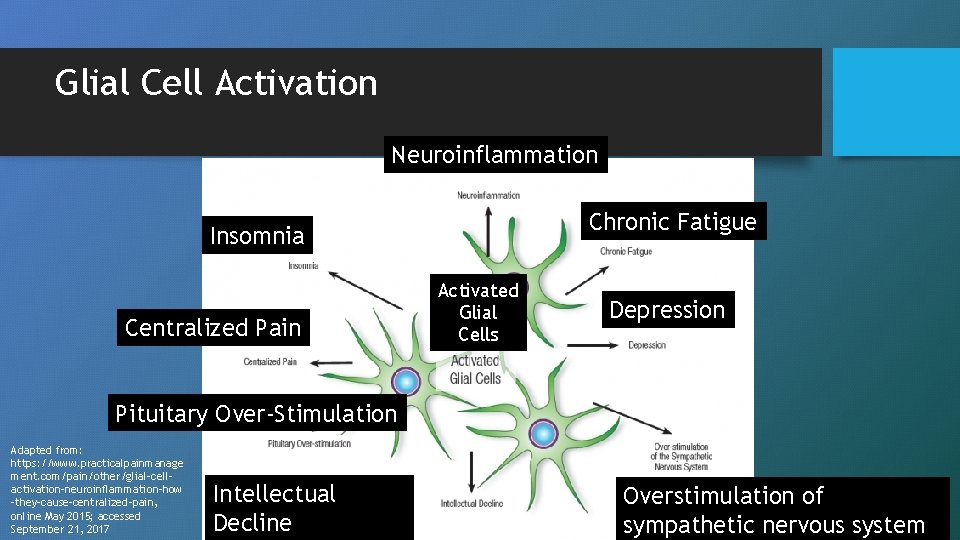
Glial Cell Activation Neuroinflammation Chronic Fatigue Insomnia Centralized Pain Activated Glial Cells Depression Pituitary Over-Stimulation Adapted from: https: //www. practicalpainmanage ment. com/pain/other/glial-cellactivation-neuroinflammation-how -they-cause-centralized-pain, online May 2015; accessed September 21, 2017 Intellectual Decline Overstimulation of sympathetic nervous system
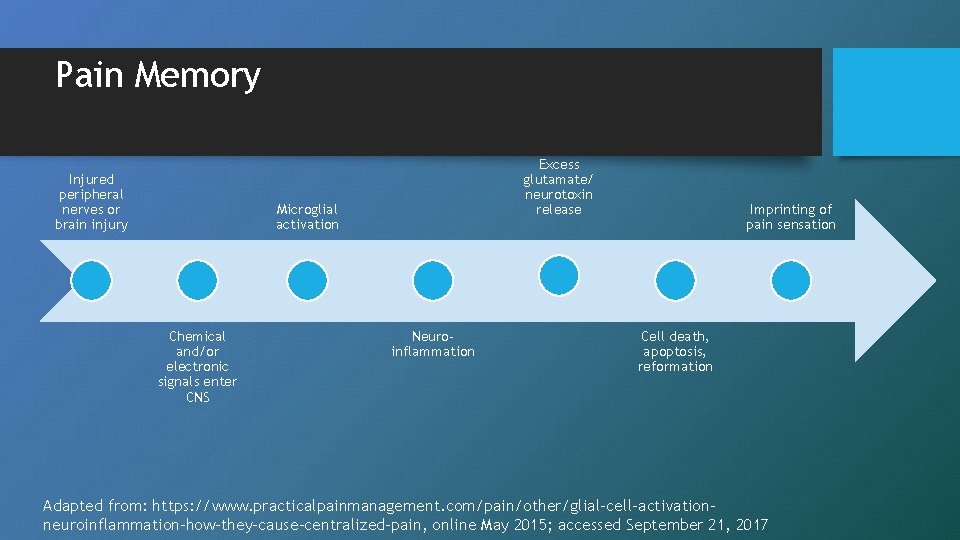
Pain Memory Injured peripheral nerves or brain injury Excess glutamate/ neurotoxin release Microglial activation Chemical and/or electronic signals enter CNS Neuroinflammation Imprinting of pain sensation Cell death, apoptosis, reformation Adapted from: https: //www. practicalpainmanagement. com/pain/other/glial-cell-activationneuroinflammation-how-they-cause-centralized-pain, online May 2015; accessed September 21, 2017

What You See • Major Symptoms • Constant pain • Insomnia, depression, fatigue • Secondary Symptoms • Anxiety, anorexia, hopelessness, allodynia • Behavior • Reclusiveness, immobility • Becoming house/bed bound • Sympathetic Nervous System Excitation • Hypertension, tachycardia, hyperthermia, hyperhidrosis, mydriasis, etc. https: //www. practicalpainmanagement. com/pain/other/glial-cell-activation-neuroinflammationhow-they-cause-centralized-pain, online May 2015; accessed September 21, 2017

But Why? Lowers pain threshold • Stress • Poor sleep • Operant learning • Individual beliefs and expectations Predictors of chronic pain • Anxiety disorder • Physical, psychological, emotional trauma or abuse • Depression

The Common Denominator Fibromyalgia PTSD Interstitial Cystitis Primary Dysmenorrhea Central Sensitization Tension Headache Migraines Multiple Chemical Sensitivities Restless Legs Syndrome Chronic Fatigue Syndrome Functional GI disorders Temporomandibular Disorders Myofascial Pain Syndromes

Treatments of Central Sensitization • Ketamine • Gabapentin • Pregabalin • Duloxetine, milnacipran, lamotrigine • Cox-2 inhibitors • Cognitive Behavioral Therapy • Aerobic exercise/physical therapy • Chronic pain rehabilitation • Massage therapy • Relaxation • Acupuncture

Question Common attributes of both central sensitization and addiction are: A. Confusion B. Allodynia C. Hyperalgesia D. Environmental Factors

When should opioid tapering be considered? • Lack of functional improvement • Pain not effectively treated • Intolerable side effects • Patient preference Benefits • Unacceptable risk • • • Nonadherence with treatment plan Concern for OUD or SUD Aberrant behaviors Concomitant medications High-dose opioid therapy Psychologic comorbidities Risks Mayo Clin Proc. 2015; 90(6): 828 -842; VA/Do. D 2017

General Principles • Recommendations vary in guidelines and primary literature • Limited evidence exists comparing tapering strategies • Must be patient-centered – no single strategy will apply to all patients Mayo Clin Proc. 2015; 90(6): 828 -842; VA/Do. D 2017

Opioid De-escalation • Should only be attempted after a discussion with patient and shared decision-making regarding a treatment plan: • Consider non-opioid and non-pharmacologic therapeutic options for pain control • Discuss options for treatment for concomitant substance use disorder, as necessary • Recommend options for mental and behavioral health treatment, as appropriate Mayo Clin Proc. 2015; 90(6): 828 -842; VA/Do. D 2017; Ment Health Clin. 2015; 5(3): 102 -108; VA PBM Academic Detailing Service 2016

Opioid De-escalation • Initial rate of reduction can be variable and is related to risk stratification • Patient risk (OUD, mental health conditions, other comorbidities, illicit behaviors) • Duration of opioid therapy • Opioid formulation • Reduction of 5 -20% every 4 weeks is common • Often first reduce the dose of the medication to the smallest commonly available unit dosage and then increase the amount of time between doses • PRN use of short-acting therapy generally does not need to be tapered

Factors Determining Taper Duration • • Faster taper recommended Higher risk patient Shorter opioid treatment duration Short-acting opioid therapy Consider > 25% initial dose reduction • • Slower taper required Lower risk patient Longer opioid treatment duration Long-acting opioid therapy Consider 10 -25% reduction every 4 weeks Mayo Clin Proc. 2015; 90(6): 828 -842; VA/Do. D 2017; Ment Health Clin. 2015; 5(3): 102 -108; VA PBM Academic Detailing Service 2016

Taper Examples Slowest taper Slow taper Fast Taper Duration Years Months to years Weeks Method Reduce by 210% every 1 -2 months Reduce by 520% every 4 weeks Rapid Taper Days Reduce by 10 Reduce by 2020% every week 50% of first dose if needed, then reduce by 10 -20% daily Adapted from: VA PBM Academic Detailing Service 2016

Patient Case • RA is a 59 yo WM with a history of chronic lumbar pain from a MVA, for which he has been prescribed opioid therapy for the last nine years. Pain is thought to be primarily neuropathic in origin. Patient and provider are concerned about worsened pain and function, despite recent opioid dose increases. Patient’s provider would like to taper RA’s opioid therapy in favor of alternatives for pain and requests your assistance.

Patient Case • Active problems • • • Chronic low back pain Depression Hypertension Hyperlipidemia Pertinent labs are WNL • UDS, PDMP, refill history have been appropriate • Current medications: • • • Morphine SR 30 mg TID Sertraline 50 mg daily Lisinopril 10 mg daily Atorvastatin 40 mg daily Aspirin 81 mg daily
Patient Case How quickly should RA’s therapy be tapered? a) Rapidly over the next two weeks, due to his high risk b) Quickly over the next month, due to his concern for adverse effects c) Slowly over the next several months, due to his duration of therapy d) Very slowly over the next year, due to his high opioid dose

Patient Case • How quickly should RA’s therapy be tapered? a) Rapidly over the next two weeks, due to his high risk b) Quickly over the next month, due to his concern for adverse effects c) Slowly over the next several months, due to his duration of therapy d) Very slowly over the next year, due to his high opioid dose

Possible Taper Schedule Current regimen: morphine SA 30 mg TID • Month 1: 30 mg QAM, 15 mg at noon, 30 mg QPM • Month 2: 30 mg QAM, 15 mg at noon, 15 mg QPM • Month 3: 15 mg TID • Month 4: 15 mg Q 12 H • Month 5: 15 mg once daily, then stop Mayo Clin Proc. 2015; 90(6): 828 -842; VA/Do. D 2017; Ment Health Clin. 2015; 5(3): 102 -108; VA PBM Academic Detailing Service 2016

How might this tapering plan differ if a fast taper were required due to patient risk?

What additional pharmacotherapy could you recommend for RA?

Withdrawal Symptom Management • Withdrawal symptoms are not life-threatening, but may occur even with a gradual taper • Short-term medications may be considered to limit withdrawal symptoms • Do NOT use benzodiazepines or opioids to manage withdrawal symptoms
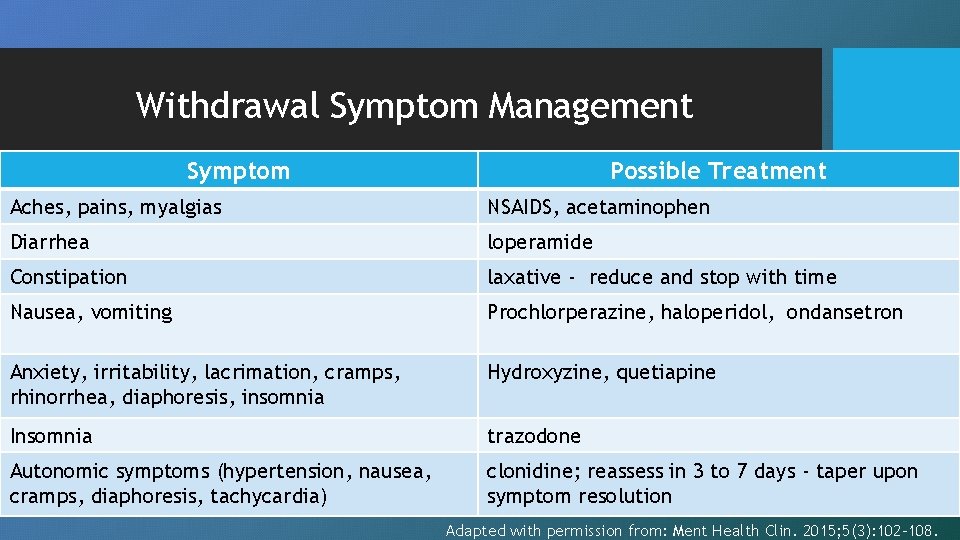
Withdrawal Symptom Management Symptom Possible Treatment Aches, pains, myalgias NSAIDS, acetaminophen Diarrhea loperamide Constipation laxative - reduce and stop with time Nausea, vomiting Prochlorperazine, haloperidol, ondansetron Anxiety, irritability, lacrimation, cramps, rhinorrhea, diaphoresis, insomnia Hydroxyzine, quetiapine Insomnia trazodone Autonomic symptoms (hypertension, nausea, cramps, diaphoresis, tachycardia) clonidine; reassess in 3 to 7 days - taper upon symptom resolution Adapted with permission from: Ment Health Clin. 2015; 5(3): 102 -108.

Co-analgesic Therapy Maria Foy, Pharm. D, BCPS, CPE

Patient-Centered Non-Opioid Therapies • Based on focused history, diagnoses, pain assessment, pain type • Nociceptive (arthritis, joint/muscle pain) acetaminophen, NSAIDs • Neuropathic (diabetic neuropathy, PHN) anticonvulsants, antidepressants • Patient-specific risk for adverse events • History of GI bleed, renal or hepatic dysfunction, age, uncontrolled hypertension • Patient-specific indications for dual benefit of a particular medication • Insomnia, obesity, depression, anxiety • Start one new agent at a time • May trial multiple agents in a therapeutic category

Options for Alternative Therapy • N-methyl-D-aspartate (NMDA) antagonists • Acetaminophen • Non-steroidal anti-inflammatory agents (NSAIDs) • Tricyclic antidepressants (TCAs) • Serotonin-norepinephrine reuptake inhibitors (SNRIs) • Anticonvulsants • Topicals

NMDA Antagonists • Activation of N-methyl-D-aspartate (NMDA) receptors associated with hyperalgesia, neuropathic pain, and reduced functionality of opioid receptors • Antagonists may play a role in improvement • Limiting factors for use • Side effects (CNS effects, cardiovascular, GI) • Provider expertise and training • Ketamine and methadone most commonly used US Pharm. 2011; 36(5): HS 4 -HS 8; Comprehensive Treatment of Chronic Pain by Medical, Interventional, and Integrative Approaches 2013

Ketamine for Chronic Pain FDA approved indications • Anesthetic agent for diagnostic and surgical procedures not requiring muscle relaxation • Induction of anesthesia • A supplement to low potency anesthetics • Nitrous oxide Off Label Uses • Prevention/reversal of central sensitization • Treatment of pain not responding to opioids • Treatment for neuropathic pain • Treatment for acute pain in opioid tolerant patients
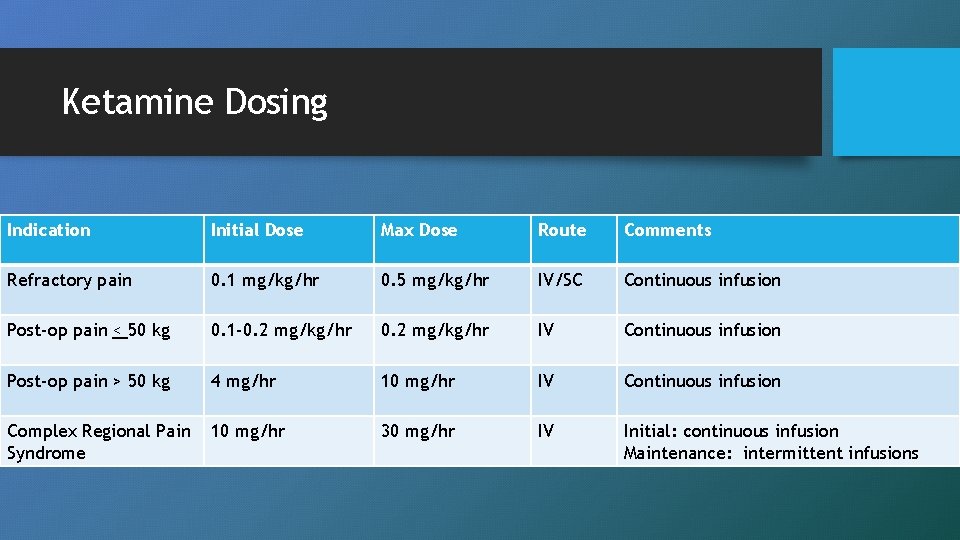
Ketamine Dosing Indication Initial Dose Max Dose Route Comments Refractory pain 0. 1 mg/kg/hr 0. 5 mg/kg/hr IV/SC Continuous infusion Post-op pain < 50 kg 0. 1 -0. 2 mg/kg/hr IV Continuous infusion Post-op pain > 50 kg 4 mg/hr 10 mg/hr IV Continuous infusion Complex Regional Pain Syndrome 10 mg/hr 30 mg/hr IV Initial: continuous infusion Maintenance: intermittent infusions

Ketamine Adverse Effects • • Hypotension , hypertension, tachycardia Psychomimetic effects (hallucinations, dreamlike state) Increased intracranial pressure Excessive sedation Excessive salivation Nausea, vomiting Tonic clonic movement, tremors, diploplia

Ketamine Clinical Pearls • Ketamine produces opioid sparing effects • Consider decreasing opioid dosing by 25 -50% at initiation of ketamine therapy, followed by 25% every 12 -24 hours as tolerated • Signs of withdrawal indicate too rapid of a opioid dose reduction. Treat with a slower taper or bolus dose of ketamine • Co-administration of a benzodiazepine or haloperidol is recommended to prevent or minimize psychomimetic effects • Consider lorazepam 0. 5 -1 mg IV/po q 6 -8 h • Give dose prior to initiation of infusion • If significant adverse reactions occur, stop the infusion. Side effects should subside in 15 -30 minutes

Methadone • 3 mechanisms of analgesia • Mu receptor agonist, NMDA receptor antagonist, norepinephrine reuptake inhibitor • Variable half life • Average of 24 hours, range of 8 -56 hours • Multiple cytochrome P 450 drug-drug interactions • Can prolong the QTC interval • Higher dosing • Congenital predisposition to pro-longed QTC • Dose increases should not occur before a minimum of 5 -7 days • Pain control at day 3 can identify risk of overdose at day 5

Acetaminophen • Dose: 650 -1000 mg Q 4 -6 hours • Recommend scheduled use, not PRN • Limit to 3 -4 g/day in healthy patients, 2 g/day in hepatic impairment • Be aware of duplication of therapy • No antiplatelet effect (< 2 g/day)

Acetaminophen Toxicity • Acute hepatotoxicity • Seen with ingestion of 7. 5 -15 grams • Fatal at 20 -25 g • Heavy alcohol consumption, fasting, or malnutrition may increase metabolism to toxic metabolite • Chronic toxicity • Ingestion of >4 g/day chronically • Arguments to lower maximum dose ongoing • Consuming >2 alcoholic drinks/day with doses greater than 2 g/day • Alcoholic liver disease

NSAIDs • Naproxen, ibuprofen, meloxicam, diclofenac, sulindac, etodolac, indomethacin • Caution in renal disease, cardiovascular disease, heart failure, history of GI bleed • Consider celecoxib in patients with history of GI intolerance

NSAID Adverse Effects • NSAID therapy adverse effects (AE) associated with various body systems • Gastrointestinal, Cardiovascular • Hepatic, Renal, • Central Nervous, Respiratory • Special considerations for children and pregnant or lactating women • Chronic therapy and higher doses associated with increased AE risk

Tricyclic Antidepressants • Tertiary amines • Amitriptyline, imipramine • Slightly more efficacious for pain, but less well tolerated • Secondary amines • Nortriptyline, desipramine • More well tolerated • Dose at bedtime, due to likelihood of sedation (antihistaminergic properties) • Consider in: • Younger patients for neuropathic pain • Patients with concomitant insomnia or depression • Caution in: • Elderly because of anticholinergic effects • Cardiovascular disease

TCA Adverse Effects • • Sedation Dry mouth, Blurred vision Confusion, delirium Constipation Orthostatic hypotension Weight gain Cardiovascular effects • Conduction blocks • Tachycardia • Ventricular arrhythmias

Selective Norepinephrine Reuptake Inhibitors • Duloxetine, venlafaxine, desvenlafaxine, milnacipran, levomilnacipran • Consider for neuropathic pain in patients with concomitant depression/anxiety • May increase blood pressure • Nausea and vomiting common • Duloxetine • Also indicated for musculoskeletal pain and fibromyalgia • Avoid in hepatic insufficiency, Cr. Cl <30 m. L/min • Venlafaxine • IR product dosed BID or TID • Adjust dose based on renal function • Abrupt withdrawal associated with serious abstinence syndrome

Anticonvulsants • Gabapentin, pregabalin, topiramate, oxcarbazepine, carbamazepine, lamotrigine, valproic acid • Gabapentin generally first line • Renal dose adjustment required • Titrate to a target dose of 1800 mg/day in divided doses • Max dose 3600 mg/day • Pregabalin may be preferred over gabapentin • Improved tolerability, faster onset, and ease of titration • Renal dose adjustment required • Monitor for sedation, fluid retention

Anticonvulsants • Monitor for cognitive effects, especially with topiramate • Consider topiramate • Obese patients due to appetite suppression • Concomitant substance use disorders (smoking cessation, alcohol use disorder, others) • Migraines • Caution with carbamazepine due to drug-drug interactions

Topicals • Diclofenac gel or patch, menthol/methyl salicylate cream, trolamine salicylate cream, capsaicin cream or patch, lidocaine gel, cream, or patch • Good options in patients at high risk for side effects from other agents • Diclofenac gel, capsaicin cream must be reapplied 3 -4 times daily for optimal benefit • Diclofenac gel ideal in patients who cannot take oral NSAIDs (minimal systemic absorption) • Capsaicin cream onset of action: 2 – 4 weeks • Avoid use with irritated or broken skin

Back to RA • Active problems • Chronic low back pain • Depression • Hypertension • Hyperlipidemia • Pertinent labs are WNL • UDS, PDMP, refill history have been appropriate • Current medications: • Morphine SR 30 mg TID tapering plan in place • Sertraline 50 mg daily • Lisinopril 10 mg daily • Atorvastatin 40 mg daily • Aspirin 81 mg daily

What additional pharmacotherapy could you recommend for RA? a) Stop sertraline, start duloxetine 30 mg daily x 1 week, then increase to 60 mg daily b) Consider cross taper of morphine to methadone, following baseline EKG c) Start meloxicam 30 mg daily with food d) Both A and B

What additional pharmacotherapy could you recommend for RA? a) Stop sertraline, start duloxetine 30 mg daily x 1 week, then increase to 60 mg daily b) Consider cross taper of morphine to methadone, following baseline EKG c) Start meloxicam 30 mg daily with food d) Both A and B

Self Assessment • Which of the following should be considered when determining the speed of an opioid taper? a) Patient gender b) Duration of opioid treatment c) Opioid dose
Self Assessment • Which of the following should be considered when determining the speed of an opioid taper? a) Patient gender b) Duration of opioid treatment c) Opioid dose

Key Takeaways • Opioid tapers must be individualized for patient needs • Recommend slower tapers for those with a longer duration of opioid therapy and with lower risk of use • Consider rapid tapers for higher risk patients • Optimization of non-opioid therapy can assist in tapering efforts • Ketamine use is becoming more common for treatment of refractory pain and for opioid sparing effects

References • Berna C, Kulich RJ, Rathmell JP. Tapering long-term opioid therapy in chronic noncancer pain: evidence and recommendations for everyday practice. Mayo Clin Proc. 2015; 90(6): 828 -842. • Kral LA, Jackson K, Uritsky T. A practical guide to tapering opioids. Ment Health Clin. 2015; 5(3): 102 -108. • VA/Do. D clinical practice guideline for management of opioid therapy for chronic pain. Washington, DC: Veterans Health Administration; 2017. • Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain United States, 2016. MMWR Recomm Rep 2016; 65(1); 1– 49. • VA PBM Academic Detailing Service. Pain management opioid taper decision tool – A VA clinician’s guide. Washington, DC; Veterans Health Administration; 2016. • Vorobeychik Y, Willoughby CD, Mao J. NMDA Receptor Antagonists in the Treatment of Pain. In: Deer T. et al. (eds) Comprehensive Treatment of Chronic Pain by Medical, Interventional, and Integrative Approaches. New York, NY. Springer. 2013. • Jamero D, Borghol A, Vo N, Hawawini F. The emerging role of NMDA antagonists in pain management. US Pharm. 2011; 36(5): HS 4 -HS 8.
- Slides: 57