QUICK START TITLE Acids and carbonates 08 DATE

QUICK START TITLE: Acids and carbonates 08 DATE: September 2021 Why do bath bombs fizz?

Check your answer When baking soda and citric acid are mixed and are then put in water, they undergo a chemical reaction. The reaction produces lots of bubbles, which you see as the bath bomb dissolves in the water. These bubbles that make the water become so fizzy are made of carbon dioxide gas

Reactions of ACID with CARBONATE produce a SALT plus WATER plus CARBON DIOXIDE Learning Objectives B to 1 : Can state that acid + carbonate is a neutralisation reaction 2: Can recall Acid + Carbonate --> Salt + Water + Carbon dioxide 3: Can describe the chemical test and outcome for Carbon dioxide 4 Can generate word equations from given acids and carbonates 5 5 -6 Can generate symbol equations from word equations and consider need for balancing equations

TASK 1 – The reaction You have just seen the reaction between vinegar and sodium hydrogen carbonate. When metal carbonates react with an acid a gas is produced. This is why we see fizzing. metal + carbonate acid salt + water + carbon dioxide The carbon and oxygen in the carbonate become carbon dioxide gas. This is another type of neutralisation reaction which forms a salt from an acid 08 September 2021

PROGRESS TARGETS IDENTIFY what gas is produced when acids react with metal carbonates. DESCRIBE what you would observe when acids react with carbonates. EXPLAIN the chemical test for carbon dioxide. APPLY data from experiments and be able to write some word/symbol equations for these reactions 08 September 2021

TASK 2 - Scenario Testing for Carbon Dioxide https: //www. youtube. com/watch? v=0 J 4 Hpjw. Kwsg Acid Carbonate Lime water turns from clear to cloudy with carbon dioxide
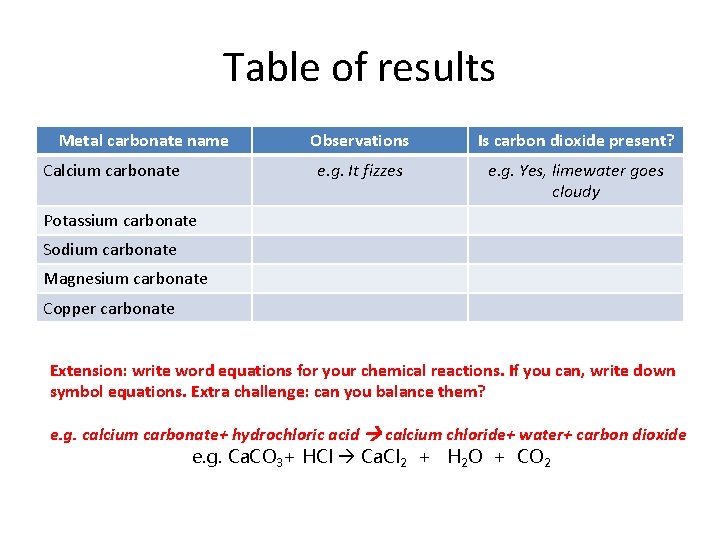
Table of results Metal carbonate name Calcium carbonate Observations Is carbon dioxide present? e. g. It fizzes e. g. Yes, limewater goes cloudy Potassium carbonate Sodium carbonate Magnesium carbonate Copper carbonate Extension: write word equations for your chemical reactions. If you can, write down symbol equations. Extra challenge: can you balance them? e. g. calcium carbonate+ hydrochloric acid calcium chloride+ water+ carbon dioxide e. g. Ca. CO 3+ HCl Ca. Cl 2 + H 2 O + CO 2

TASK 3 - Scenario A luxury soap manufacturer would like to produce a new, extremely fizzy, bath bomb. The fizzier it is, the quicker it will dissolve and release its fragrance. You have been asked to find out which metal carbonate would make the best bath bombs. You will be provided with different metal carbonates to investigate… 08 September 2021
YOU HAVE 5 MINUTES TO PLAN HOW YOU WILL CARRY OUT YOUR INVESTIGATI ON 08 September 2021 STA RT FINIS H MAKE SURE YOU KNOW WHAT DATA YOU SHOULD RECORD

TASK 4 – Things to consider Why do metal carbonates make carbon dioxide ? This is a symbol equation of the reaction between an acid (hydrochloric acid) and calcium carbonate. HCl + Ca. CO 3 CO 2 Ca. Cl 2 + H 2 O + 1. Use this symbol equation to write a word equation for this reaction 2. Can you write a word equations for all of the experiments you did in your investigation ? 3. Can you write symbol equations for all of the 08 September 2021 experiments you did in you investigation ?

PROGRESS TARGETS IDENTIFY what gas is produced when acids react with metal carbonates. DESCRIBE what you would observe when acids react with carbonates. EXPLAIN the chemical test for carbon dioxide. APPLY data from experiments and be able to write some word/symbol equations for these reactions 08 September 2021
- Slides: 11