Question 1 Chap 14 What is the direction














- Slides: 20

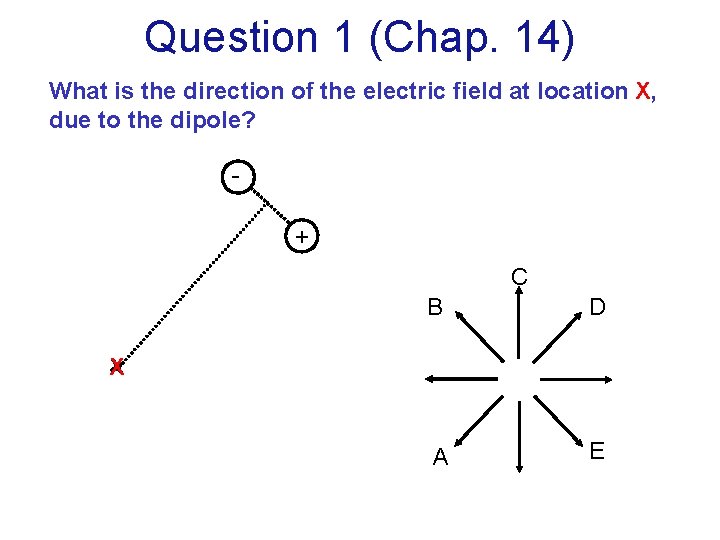
Question 1 (Chap. 14) What is the direction of the electric field at location X, due to the dipole? + C B D A E X

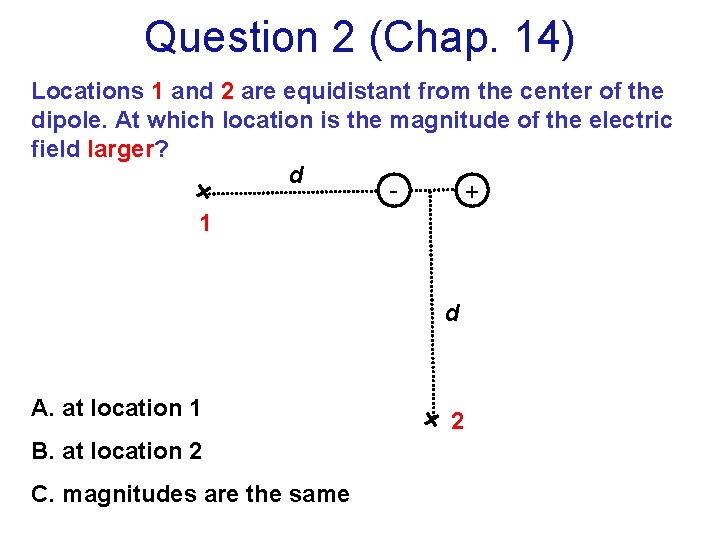
Question 2 (Chap. 14) Locations 1 and 2 are equidistant from the center of the dipole. At which location is the magnitude of the electric field larger? d + 1 d A. at location 1 B. at location 2 C. magnitudes are the same 2

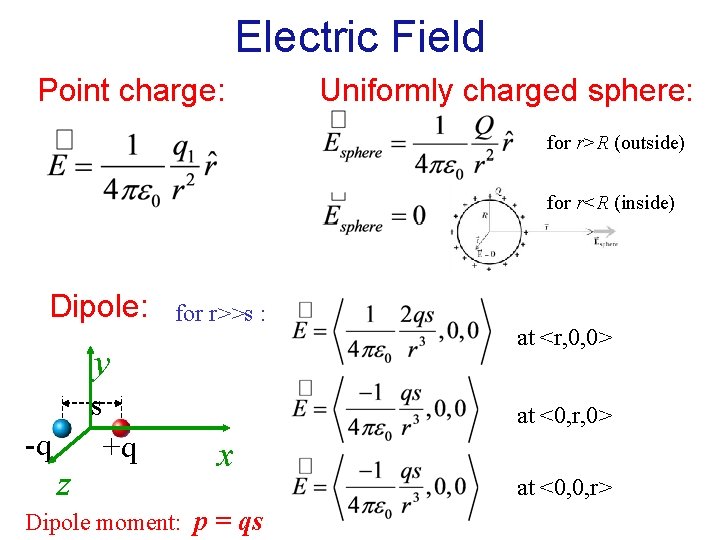
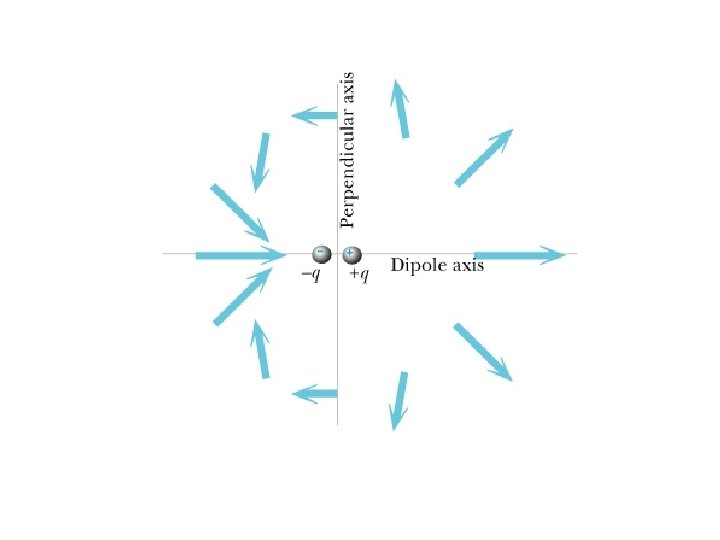
Electric Field Point charge: Uniformly charged sphere: for r>R (outside) for r<R (inside) Dipole: for r>>s : y s -q z at <r, 0, 0> at <0, r, 0> +q x Dipole moment: p = qs at <0, 0, r>


Chapter 15 Matter and Electric Fields

Net Charge Matter is made out of atoms. Atom contains charged particles: electrons (-e), protons (+e) Neutral atom: number of electrons and protons is equal: Example: Hydrogen atom: 1 proton, 1 electron net charge = (+e) + (-e)=0 Sodium atom: 11 protons, 11 electrons Sodium atom (Na) can lose an electron: Sodium ion (Na+): (+11 e) + (-10 e) = +e Ordinary matter is electrically neutral. However, can be charged by adding/removing charged particles Can we create an excess charge inside a sample?

Conservation of Charge The net charge of a system and its surroundings cannot change If one object gets charged positively, there must be an object which gets charged negatively. The net electric charge is conserved in any physical process. Charge can be transferred from one object to another.

Neutral Matter in the Electric Field A wooden dowel rod is balanced on a sharp needle and placed between a pair of parallel plates connected to an electrostatic generator. When the plates are charged, the dowel rod: A) Could not care less B) Will orient perpendicular to the direction of the E – field C) Will orient parallel to the direction of the E – field D) Will jump out of the area with E-field

The Structure of an Atom Hydrogen 10 -10 m (1 Å) Nucleus (one proton), ~10 -15 m Charge of electron cloud equals that of nucleus neutral atom. If the electron cloud is centered on the nucleus electric field produced by electrons exactly cancels the field produced by nucleus.

Polarization of Atoms + E - + Force due to E created by positive charge shifts electron cloud and nucleus in opposite directions: electric dipole. An atom is said to be polarized when its electron cloud has been shifted by the influence of an external charge so that the electron cloud is not centered on the nucleus.

Induced Dipole An applied electric field creates induced dipoles! E • it is not a permanent dipole • an induced dipole is created when a neutral object is polarized by an applied electric field

Polarization Amount of polarization p in most materials is proportional to the magnitude of the applied electric field: - “polarizability” of a material In an induced dipole, is the distance between the charges fixed? The distance is proportional to the strength of the applied field.

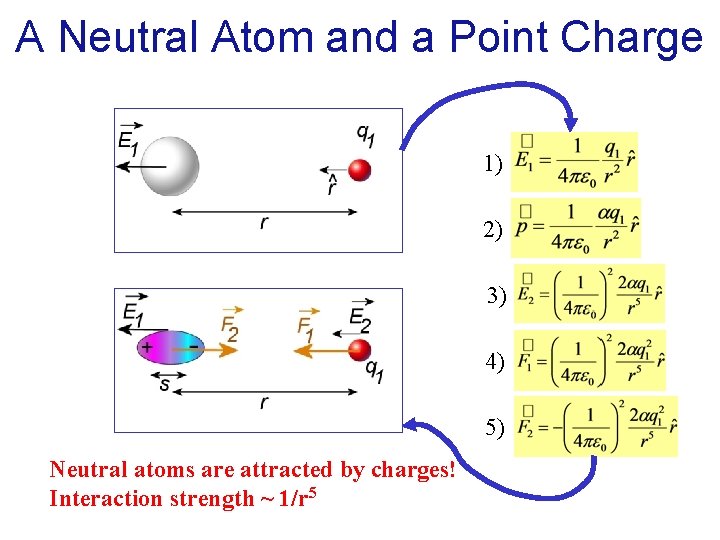
A Neutral Atom and a Point Charge 1. Charge q 1 creates field E 1 at the location of the atom

A Neutral Atom and a Point Charge 1) 2. Field E 1 polarizes the atom creating a dipole

A Neutral Atom and a Point Charge 1) 2) 3. Dipole creates field E 2 at the location of q 1

A Neutral Atom and a Point Charge 1) 3) 2) 4. Induced dipole exerts force F 1 on the charge:

A Neutral Atom and a Point Charge 1) 3) 2) 4) 5. The charge q 1 exerts force F 2 on the dipole (reciprocity):

A Neutral Atom and a Point Charge 1) 2) 3) 4) 5) Neutral atoms are attracted by charges! Interaction strength ~ 1/r 5

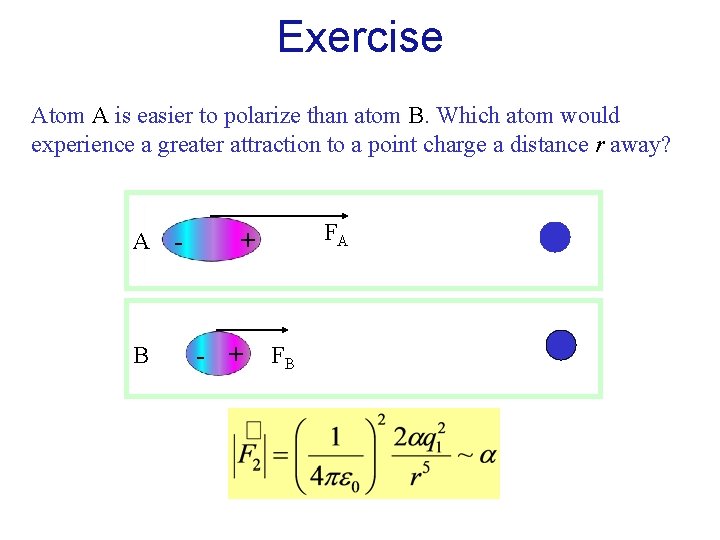
Exercise Atom A is easier to polarize than atom B. Which atom would experience a greater attraction to a point charge a distance r away? A B - FA + - + FB

Interaction of Charged Tapes and Neutral Matter