quaternary system without solubility e 1 e 2
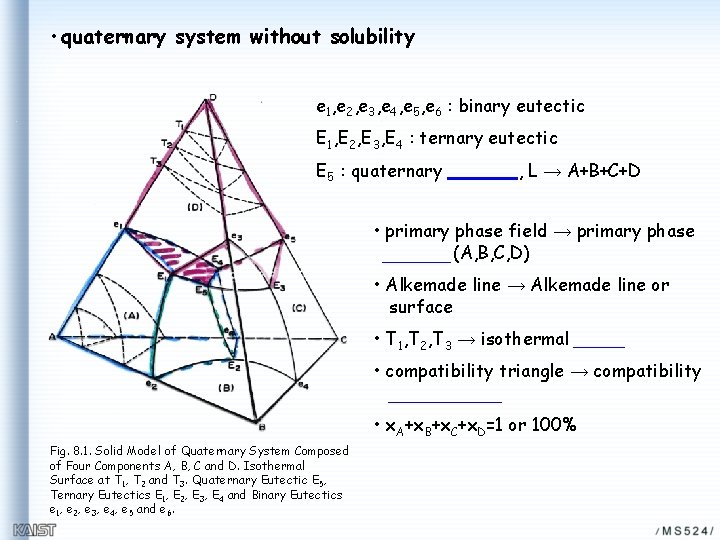
• quaternary system without solubility e 1, e 2, e 3, e 4, e 5, e 6 : binary eutectic E 1, E 2, E 3, E 4 : ternary eutectic E 5 : quaternary , L → A+B+C+D • primary phase field → primary phase (A, B, C, D) • Alkemade line → Alkemade line or surface • T 1, T 2, T 3 → isothermal • compatibility triangle → compatibility • x. A+x. B+x. C+x. D=1 or 100% Fig. 8. 1. Solid Model of Quaternary System Composed of Four Components A, B, C and D. Isothermal Surface at T 1, T 2 and T 3. Quaternary Eutectic E 5, Ternary Eutectics E 1, E 2, E 3, E 4 and Binary Eutectics e 1, e 2, e 3, e 4, e 5 and e 6.
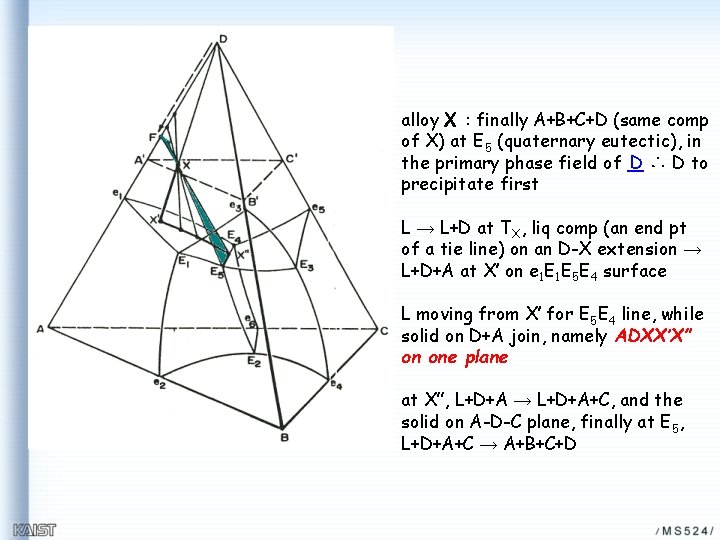
alloy X : finally A+B+C+D (same comp of X) at E 5 (quaternary eutectic), in the primary phase field of D ∴ D to precipitate first L → L+D at TX, liq comp (an end pt of a tie line) on an D-X extension → L+D+A at X’ on e 1 E 1 E 5 E 4 surface L moving from X’ for E 5 E 4 line, while solid on D+A join, namely ADXX’X” on one plane at X”, L+D+A → L+D+A+C, and the solid on A-D-C plane, finally at E 5, L+D+A+C → A+B+C+D

• the quaternary system of Si 3 N 4 -Si. O 2 -Al. N-Al 2 O 3 ( ) Si 3 N 4 1) used for advanced gas-turbine, may replace metal, easily developing on the Si. O 2 film 2) densification additives such as Al 2 O 3 being used in hot-pressing of Si 3 N 4 reciprocal salt system that is used in dealing with the typical salt in chemistry Si 3 N 4 -Al. N-Al 2 O 3 -Si. O 2 (Si-Al-O-N) % at comp unit ex) a. Si 3 N 4+b. Al. N+c. Si. O 2+d. Al 2 O 3
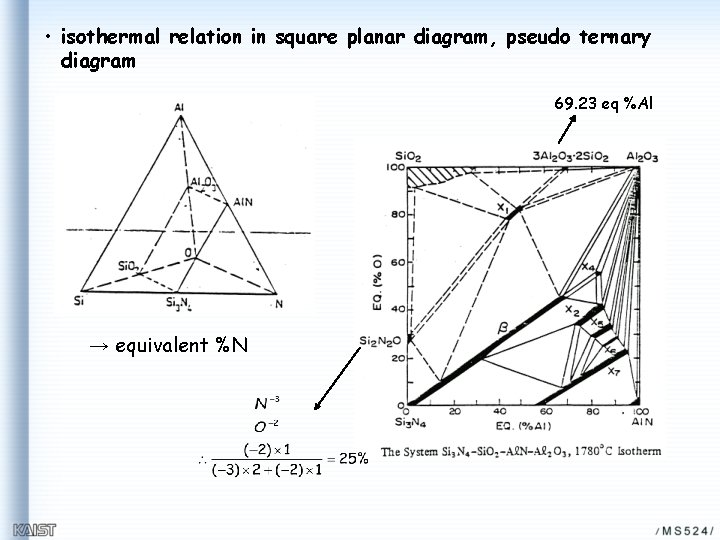
• isothermal relation in square planar diagram, pseudo ternary diagram 69. 23 eq %Al → equivalent %N
- Slides: 4