Quantum Numbers Quantum Numbers PRINCIPAL n energy level

Quantum Numbers

Quantum Numbers • PRINCIPAL: n energy level, the distance the orbital is from the nucleus (1, 2, 3, 4…) • ANGULAR MOMENTUM: l shape (s = 0, p = 1, d = 2, f = 3) • MAGNETIC: ml spatial orientation • 0 for s • -1, 0, +1 for p • -2, -1, 0, +1, +2 for d, etc. • SPIN: ms spin (+1/2 or -1/2)

st 1 Quantum # Principle Quantum # (n) Specifies the energy level that the electron is on.

nd 2 Quantum # (l) Specifies the shape of the sub level.

Quantum Numbers • The angular momentum quantum number has the symbol . = 0, 1, 2, 3, 4, 5, . . . . (n-1) = s, p, d, f, g, h, . . . . (n-1) • tells us the shape of the orbitals. • These orbitals are the volume around the atom that the electrons occupy 90 -95% of the time.

The 3 rd (m) and 4 th quantum numbers (s) … • Deals with the orbital within the sublevel and the spin of the electron.

Quantum Numbers • The symbol for the magnetic quantum number is m , representing the spatial orientation. m = - , (- + 1), (- +2), . . . 0, . . . . , ( -2), ( -1), • If = 0 (or an s orbital), then m = 0. • If = 1 (or a p orbital), then m = -1, 0, +1. y z x

• If = 2 (or a d orbital), then m = -2, -1, 0, +1, +2. • If = 3 (or an f orbital), then m = -3, -2, -1, 0, +1, +2, +3. • Theoretically, this series continues on to g, h, i, etc
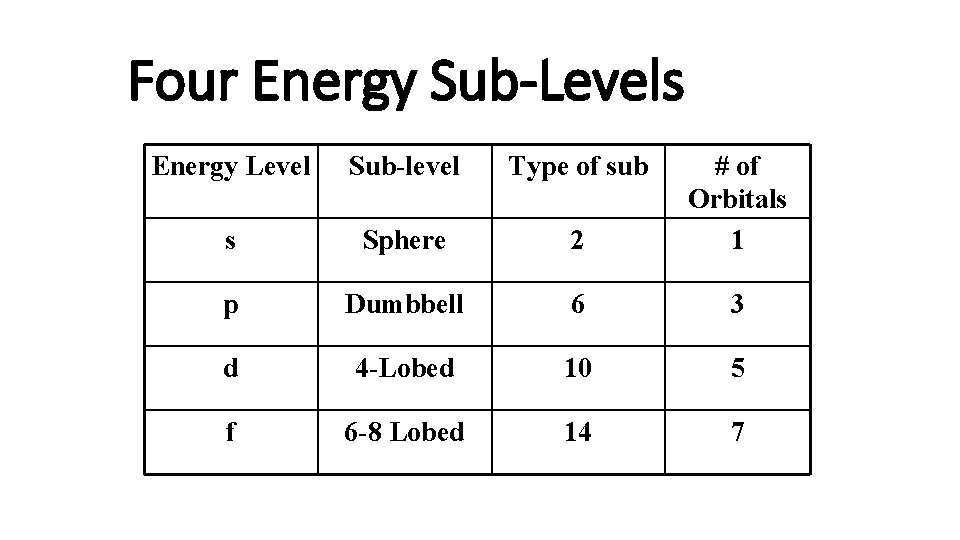
Four Energy Sub-Levels Energy Level Sub-level Type of sub s Sphere 2 # of Orbitals 1 p Dumbbell 6 3 d 4 -Lobed 10 5 f 6 -8 Lobed 14 7

Spin quantum number • The last quantum number is the spin quantum number which has the symbol ms. • The spin quantum number only has two possible values. • ms = +1/2 or -1/2
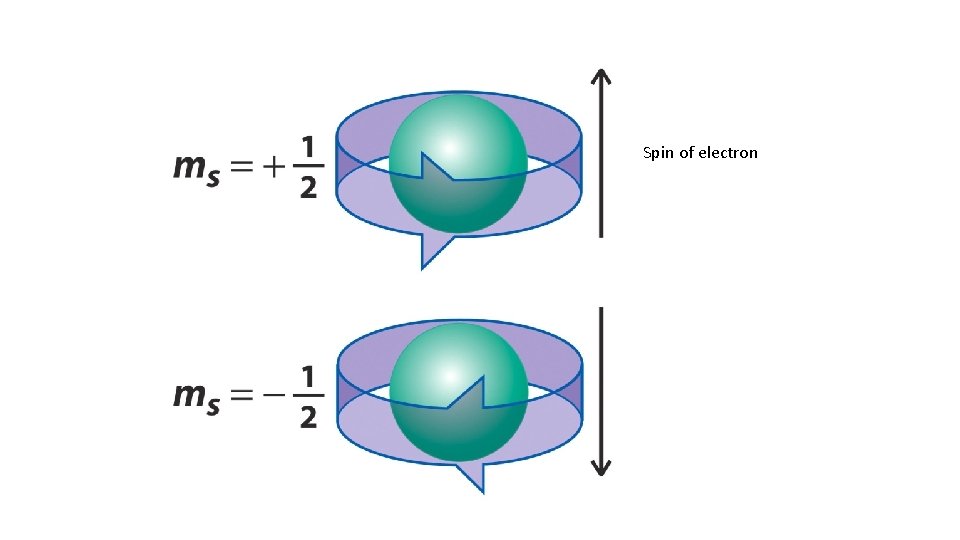
Spin of electron

Practice 1. When n = 3, l can have a value of? For the 3 d orbital, I has a value of? 2. What electron are we talking about if it has the quantum numbers of n = 2, l = 1, ml = -1 and spin of -1/2?
- Slides: 12