Quantum Numbers Quantum Numbers n Principal q n

Quantum Numbers

Quantum Numbers • n = Principal q. n. • Which energy level the electron is on. • Can have values of 1, 2, 3…. .

Quantum Numbers • l – angular momentum q. n. • Tells us about the shape of the orbitals. • s , p, d and f orbitals

Quantum Numbers • • • m – magnetic q. n. Number of orientations of the orbitals s orbital = 1 p orbital = 3 d orbital = 5 f orbital = 7

Image of Orbitals

Quantum Numbers • ms – spin q. n. • We use the model that electrons spin on its axis just as the earth spins on its axis. • Electrons can spin clock wise or counter clock wise. • We use (+ ½) spin or a (- ½) spin to distinguish the two.

Quantum Rules • There are three rules that must be followed when doing electron configuration. • The aufbau principle • Pauli’s exclusion principle • Hund’s rule

The aufbau principle • The aufbau principle says that we always start from the bottom and work our way up. In other words we begin with the first energy level and then fill orbitals from the bottom up.

The aufbau principle • The first energy level contains only 1 s orbital. Once it is filled the next electron must go up to the second energy level. • The second energy level contains the 2 s and 2 p orbitals. Once those orbitals are filled the next electron must go up to the third energy level. • The third energy level contains the 3 s, 3 p and 3 d orbitals. Are you beginning to see a pattern here?
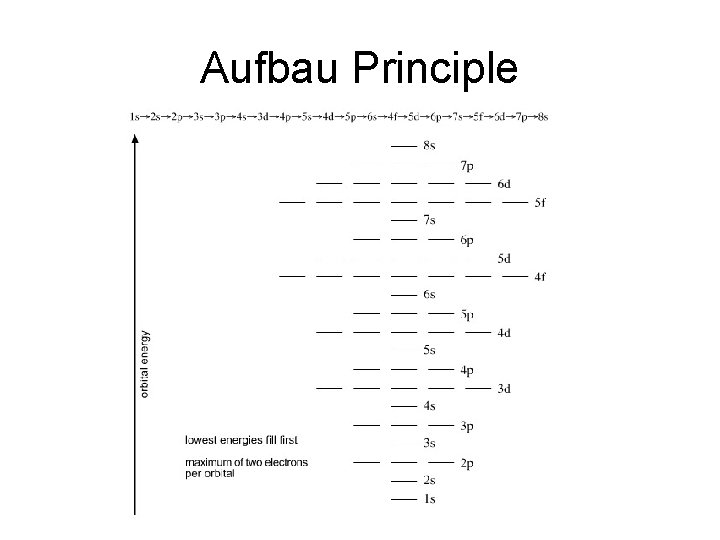
Aufbau Principle

Pauli Exclusion Principle • Pauli exclusion principle states that no two electrons can have the same four quantum numbers.

Pauli Exclusion Principle • Consider helium. Helium has two electrons. • Both electrons are on the same energy level. • Both electrons are in a s orbital. • Both electrons have the same orientation in space.

Pauli Exclusion Principle • According to the exclusion principle the fourth quantum number, spin, must be different or it will violate the exclusion principle. • The electrons in helium must have opposite spin. • The electron configuration for helium is 1 s 2

Pauli Exclusion Principle • Essentially the exclusion principle tells us that no more than two electrons can be in any one orbital. If there are more than two electrons in an orbital the exclusion principle will be violated.

Hund’s Rule • Hund’s Rule states that when electrons are filling the p orbitals, electrons must fill an empty orbital before they double up.

Electron Configuration • We use the quantum numbers to tell us where the electrons are located around an atom. • Let’s start with hydrogen. It has only one electron • Hydrogen’s electron is on the first energy level, therefore the n q. n. is equal to 1.

Electron Configuration • The 1 electron forms an s orbital. Therefore the l q. n. is s. • The s orbital looks the same no matter which direction we are looking at it. Therefore the m q. n. is just 1 • The single electron can spin either clockwise or counterclock wise. Therefore it can have a value of either +1/2 or -1/2.

Electron Configuration • We use electron configuration to put these numbers together. • The hydrogen atom has a 1 s electron configuration. • The 1 tells us the electron is on the first energy level. • The s tells us the shape of the orbital • Because the s orbital has only 1 orientation we don’t need indicate that • The electron can spin either clock wise or counter clock wise. We don’t really care which.

Electron Configuration • The electron configuration of hydrogen is: • 1 s 1

Electron Configuration • Let’s try helium. • Helium has 2 electrons. • The electrons are both on the first energy level. • They are both in an s orbital. • They both have the same orientation. • They must have opposite spin.

Electron Configuration • Helium has an electron configuration of • 1 s 2 • The 2 above the s indicates 2 electrons are in the s orbital.
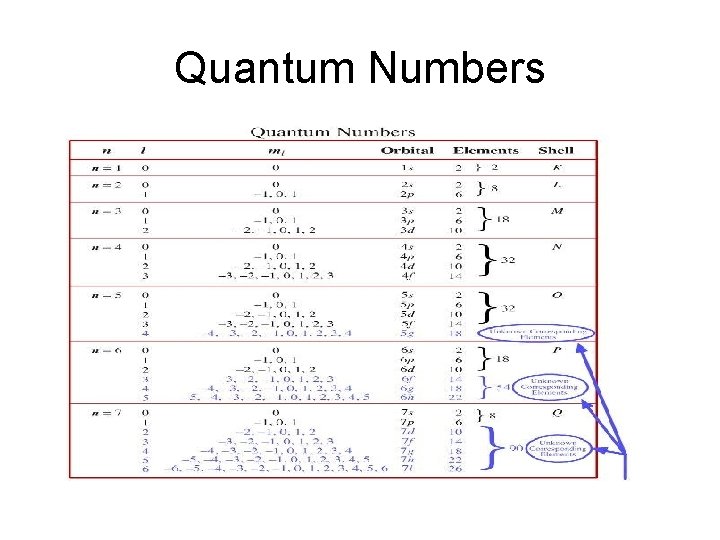
Quantum Numbers
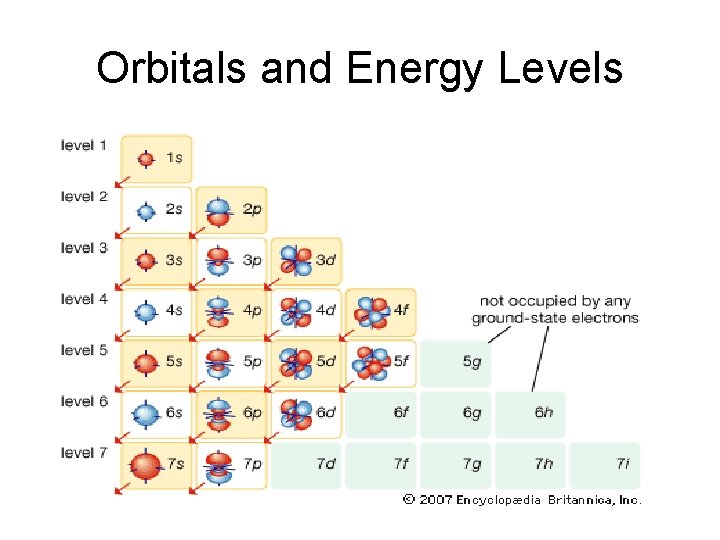
Orbitals and Energy Levels

Electron Configuration
- Slides: 24