Quantum Numbers n l m and s Used

Quantum Numbers • n, l, m, and s – Used to describe an electron in an atom • Probable location • n – Principal Quantum Number – Represents main energy level of electron • Maximum # of electrons in an energy level = 2 n 2 • Example: What is the maximum number of electrons that can be in the 5 th main energy level? – 2(52) – Electrons in 7 th energy level? Episode 304

Quantum Numbers • l • The 2 nd quantum number – Angular momentum quantum number • Describes the orbital shape within an energy level • Number of orbital shapes possible in an energy level = n http: //www. youtube. com/watch? v=Kj. Ngq 16 j. EY&list=PLD 1 C 287 B 0 E 1484083&index=9&feature=plcp Episode 304
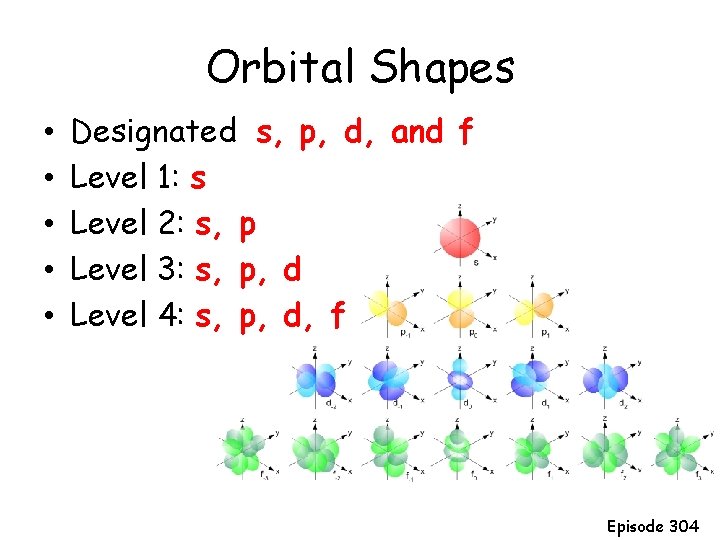
Orbital Shapes • • • Designated s, p, d, and f Level 1: s Level 2: s, p Level 3: s, p, d Level 4: s, p, d, f Episode 304

How many electrons can each sublevel hold? • • s = 1 orbital x 2 e-/orbital = p = 3 orbitals x 2 e-/orbital = d = 5 orbitals x 2 e-/orbital = f = 7 orbitals x 2 e-/orbital = Energy Level E sublevel n Type of orbital 1 2 s s p 2 e 6 e 10 e 14 e- # of orbitals # e- in orbital # e- = 2 n 2 1 1 2 2(12) = 2 2 6 2(22) = 8 3 Episode 304
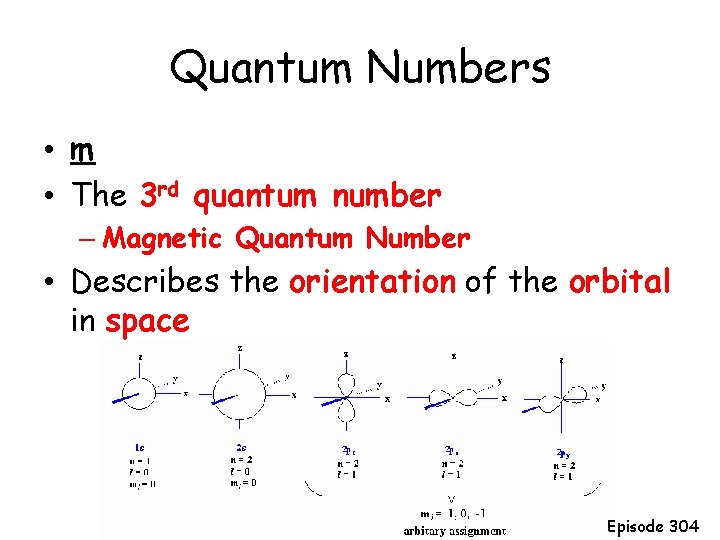
Quantum Numbers • m • The 3 rd quantum number – Magnetic Quantum Number • Describes the orientation of the orbital in space Episode 304

Quantum Numbers • The 4 th quantum number – Spin Quantum Number http: //www. youtube. com/watch? v=2 yp. C 7 rn. FXLU&list=PLD 1 C 287 B 0 E 1484083&index=1&feature=plcp • Describes the spin of the electron in orbital • Ground state • Lowest energy arrangement of electrons – Aufbau Principle Episode 304

Diagonal Rule Examples: • Hydrogen – 1 electron – 1 s 1 • Lithium – 3 electrons – 1 s 2 2 s 1 • Nitrogen – 7 electrons – 1 s 2 2 p 3 Episode 304

• Electron Configurations – Describes the electron distribution within an atom • Longhand electron configuration – Nitrogen 1 s 2 2 p 3 • Orbital Notation – Uses arrows to represent electrons • Examples: – Hydrogen 1 s 1 1 s Episode 304

Nitrogen • 1 s 2 2 p 3 1 s 2 s 2 p • Hund’s Rule • Orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second electron (spinning in opposite direction) • Pauli Exclusion Principle • No two electrons in the same atom can have the same set of 4 quantum numbers Episode 304
- Slides: 9