Quantum Numbers HONORS CHEMISTRY ONLY 3 a Quantum

Quantum Numbers

HONORS CHEMISTRY ONLY 3 a. Quantum Numbers • Electron energies are addressed in a similar way to a ZIP code. Many addresses in Ulster and northern Orange • county have 125 as the prefix, with the last two digits signifying the actual postal box. • For example, New Paltz is 12561, Wallkill is 12589, Newburgh is 12550, Pine Bush is 12566.

3 a. Quantum Numbers • There are four identifying characteristics of the energy of a specific electron in an atomic, each more specific than the last. • They are: – n (principal quantum number) = Principal Energy Level (1, 2, 3, 4, etc. ) – l (levarotary) = Sublevel (s, p, d, f) – m (magnetic) = Orbital – s (spin) = Spin (+ 1/2, - 1/2)

3 a. Quantum Numbers • n , principal quantum number – based on Bohr’s observations of line spectra for different elements – ‘n’ relates to the main energy of an electron – allowable values: n = 1, 2, 3, 4, … – electrons with higher ‘n’ values have more energy

3 a. Quantum Numbers • l , The Secondary Quantum Number – based on the observation that lines on line spectra are actually groups of multiple, thin lines – ‘l ’ relates to the shape of the electrons’ orbits – allowable values: l = 0 to l = n - 1 • i. e. for n = 4: l = 0, 1, 2, or 3 – the ‘l ’ values 0, 1, 2, and 3 correspond to the shapes we will call s, p, d and f, respectively

3 a. Quantum Numbers • ml , the Magnetic Quantum Number – based on the observation that single lines on line spectra split into new lines near a strong magnet – ‘ml ’ relates to the direction/orientation of the electrons’ orbits – allowable values: ml = - l to + l • i. e. for l = 2: ml = -2, -1, 0, 1, or 2 – electrons with the same l value but different ml values have the same energy but different orientations

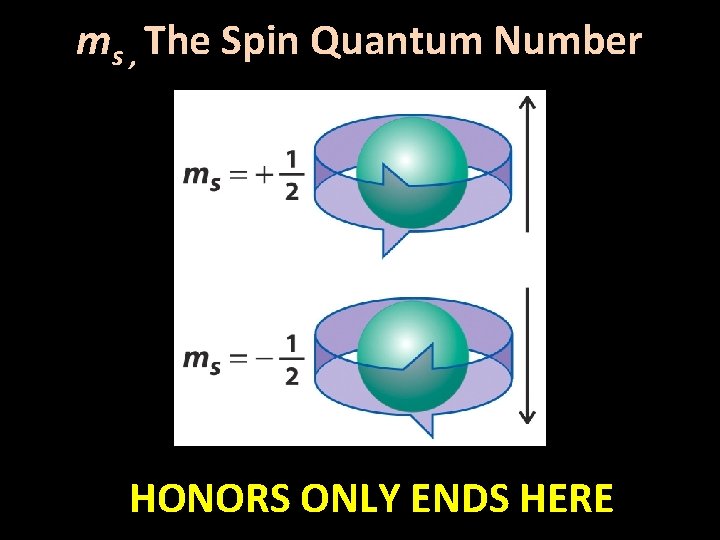
3 a. Quantum Numbers • ms , The Spin Quantum Number – based on the observation that magnets could further split lines in line spectra, and that some elements exhibit paramagnetism – ‘ms ’ relates to the ‘spin’ of an electron – allowable values: ms = - ½ or + ½ • i. e. for any possible set of n, l, and ml values, there are two possible ms values – when two electrons of opposite spin are paired, there is no magnetism observed; an unparied electron is weakly magnetic
ms , The Spin Quantum Number HONORS ONLY ENDS HERE

Click Below for the Video Lectures Quantum Mechanical Model
- Slides: 9