Quantum Numbers Electron Configuration and Orbital Diagrams What

Quantum Numbers, Electron Configuration, and Orbital Diagrams

What are quantum numbers? �Describes the location of an electron �Kind of like an electron’s address

st 1 quantum number � Symbol n � Called the principal quantum number � Describes which energy level the electron is located on � Formula to figure out how many electrons an energy level can hold is …
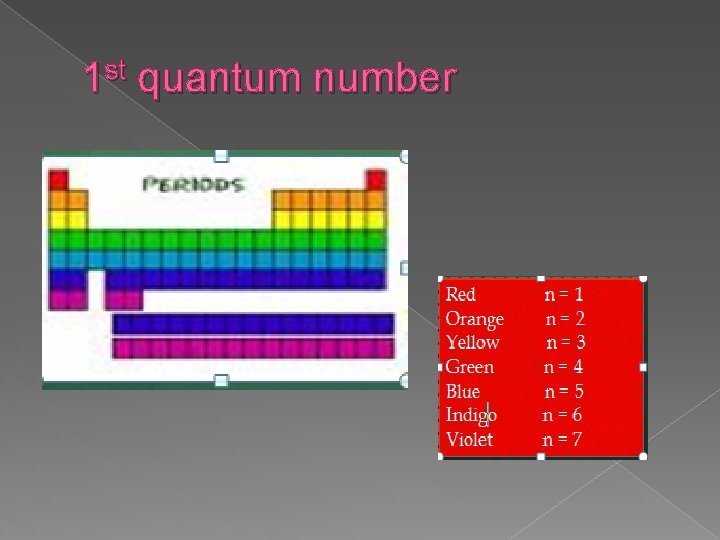
1 st quantum number
nd 2 quantum number � Symbol---- l � Called the azimuthal quantum number � Describes which sublevel the electron is located on � There are 4 sublevels �S �P �D �F

2 nd quantum number(cont’d) �L values � L=0 (s sublevel) � L =1 ( p sublevel) � L = 2 ( d sublevel) � L = 3 ( f sublevel)

rd 3 quantum number � Symbol----m � Called the magnetic quantum number � Describes the orientation or which “orbital” the electron is located in � S--- has 1 orbital � P---has 3 orbitals � D—has 5 orbitals � F– has 7 orbitals
Orbitals � The highest probability of finding an electron. � Each orbital can only hold 2 electrons. � S--- _____ � P---______ � D-- _______ ______ � F--______ _____
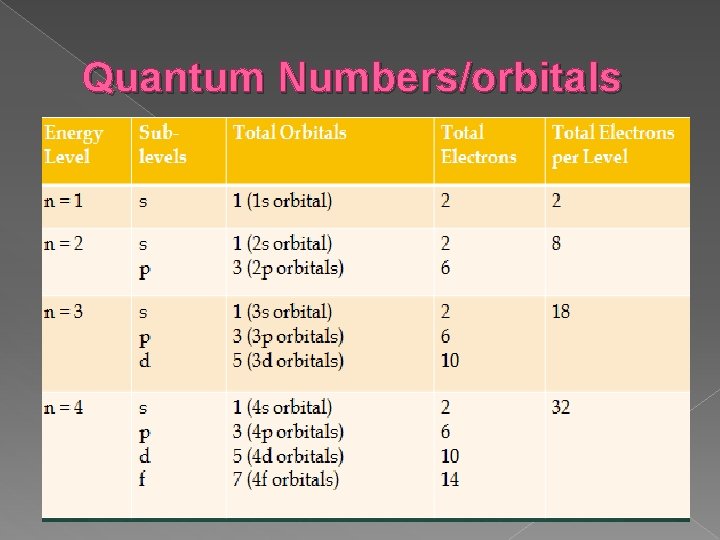
Quantum Numbers/orbitals

4 th quantum number � Symbol—s � Called the spin quantum number � Describes the spin of the electron � The values are either +1/2 or -1/2 � 2 electrons are in each orbital spinning in opposite directions

Electron Configuration � Describes the specific way the orbitals are filled. � Always start at the lowest level “ 1 s” and fill each orbital completely before proceeding to the next one. � Continue until you reach your desired element and all elements up to that point have been accounted for

No more than 2 electrons in any orbital… ever!!
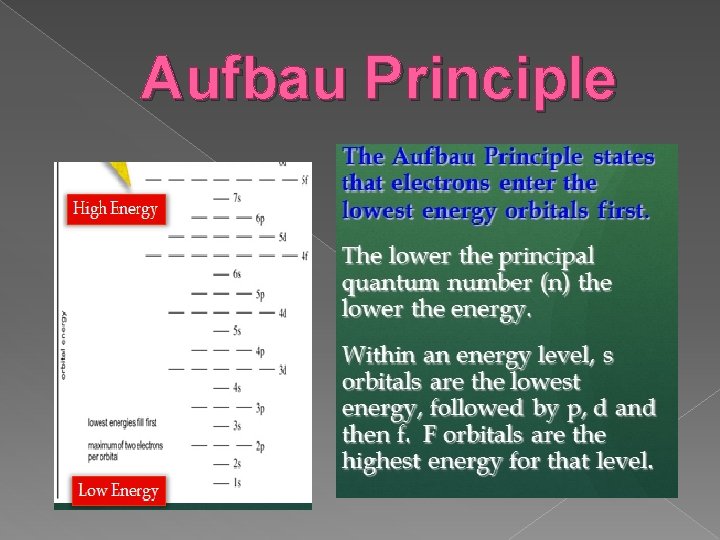
Aufbau Principle

Hund’s Rule
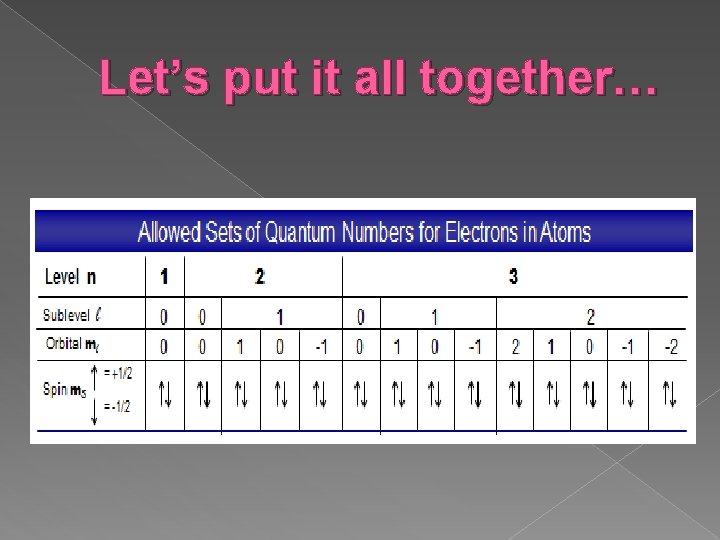
Let’s put it all together…
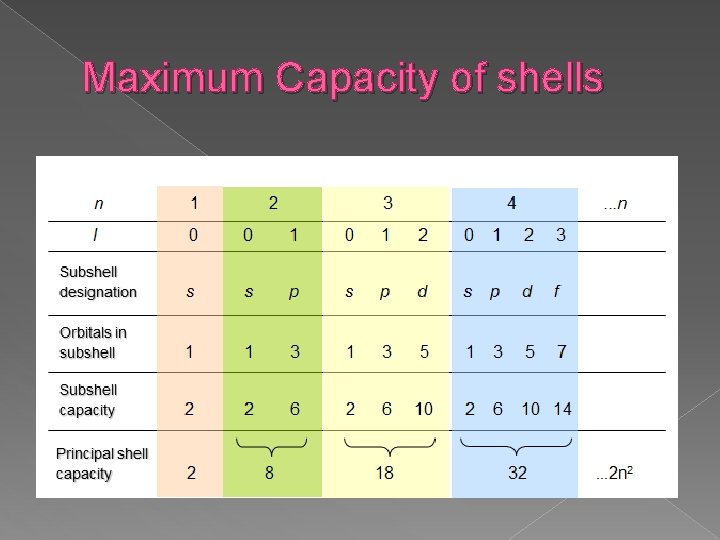
Maximum Capacity of shells
- Slides: 16