Quantum numbers are used to describe where an

Quantum numbers are used to describe where an e– is in an atom. 1. principal quantum number, n (n = integers 1, 2, 3, . . . ) -- correspond to the energy level of the electrons -- All orbitals having the same n are called an electron shell (e. g. , 2 s and 2 p). 2. angular momentum quantum number, l (l = integers from 0 up to (n – 1)) -- This number defines the type of subshell: s = 0, p = 1, d = 2, f = 3 -- For a given shell, the energies of orbitals go: s<p<d<f
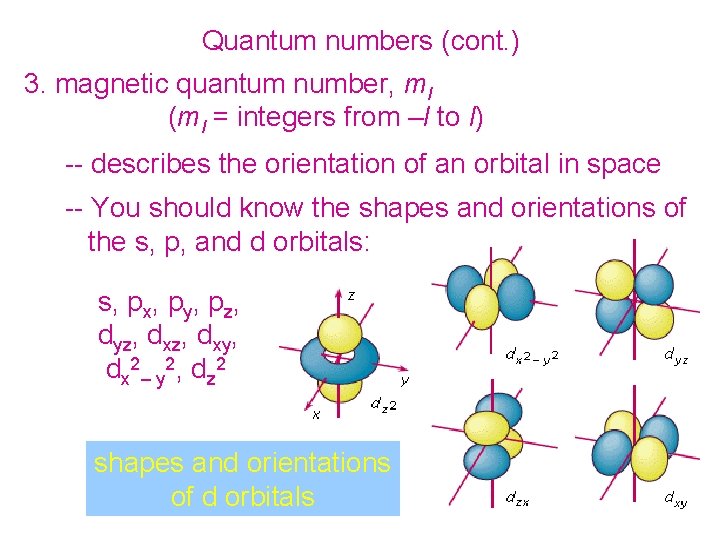
Quantum numbers (cont. ) 3. magnetic quantum number, ml (ml = integers from –l to l) -- describes the orientation of an orbital in space -- You should know the shapes and orientations of the s, p, and d orbitals: s, px, py, pz, dyz, dxy, dx 2– y 2, dz 2 shapes and orientations of d orbitals

4. electron spin quantum number, ms -- only two values: +½ or –½ (“spin-up” and “spin-down”) -- Pauli exclusion principle: No two electrons in an atom may have the same set of four quantum numbers (i. e. , an orbital may hold only two electrons, and they must have opposite spins). Wolfgang Pauli (1900– 1958)

(a) Predict the number of subshells in the third shell. n = 3, so… l could be 0, 1, or 2 3 subshells (b) Give the label for each of these subshells. For: l = 0… 3 s l = 1… 3 p l = 2… 3 d (c) How many orbitals are in each of these subshells? For: l = 0… ml = 0 l = 1… ml = – 1, 0, +1 l = 2… ml = – 2, – 1, 0, +1, +2 3 s: 1 orbital 3 p: 3 orbitals 3 d: 5 orbitals

What are the values of n and l for the following sublevels? 2 s 3 d 4 p 5 s 4 f n=2 l=0 n=3 l=2 n=4 l=1 n=5 l=0 n=4 l=3 Write the possible sets of the four quantum numbers for a 4 p electron. 4 p orbitals ml : – 1 0 +1 4, 1, – 1, +½ 4, 1, 0, +½ 4, 1, –½ 4, 1, 0, –½ 4, 1, +1, –½

Write the four quantum numbers of each of the six 3 d electrons of an iron atom. 3 d orbitals ml : – 2 – 1 0 +1 +2 3, 2, – 2, +½ 3, 2, – 1, +½ 3, 2, 0, +½ 3, 2, +1, +½ 3, 2, ? , –½ ?

Brief Review of the Periodic Table metals: left side of Table; form cations properties: lustrous (shiny) ductile (can pull into wire) good conductors (heat and electricity) malleable (can hammer into shape)

Brief Review of the Periodic Table (cont. ) nonmetals: right side of Table; form anions properties: good insulators gases or brittle solids neon sulfur iodine bromine Ne S 8 I 2 Br 2
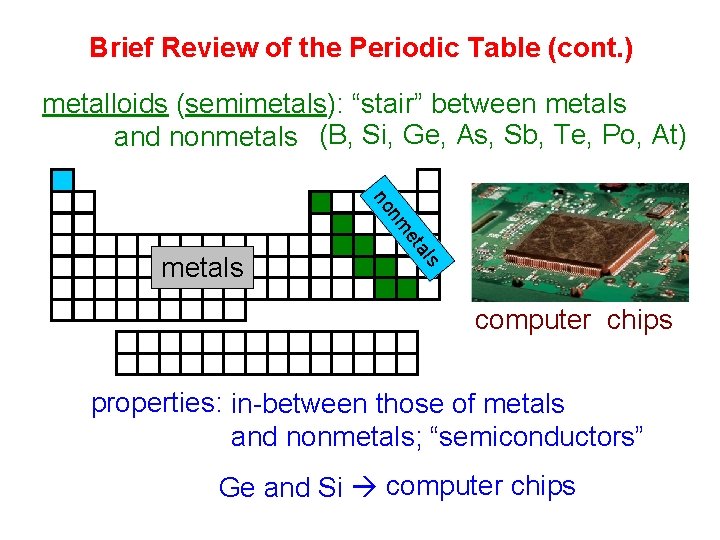
Brief Review of the Periodic Table (cont. ) metalloids (semimetals): “stair” between metals and nonmetals (B, Si, Ge, As, Sb, Te, Po, At) als et nm no metals Si and Ge computer chips properties: in-between those of metals and nonmetals; “semiconductors” Ge and Si computer chips

alkali metals: group 1 (except H); 1+ charge; very reactive alkaline earth metals: group 2; 2+ charge; less reactive than alkalis transition elements: groups 3– 12; variable charges chalcogens: group 16; 2– charge; reactive halogens: group 17; 1– charge; very reactive noble gases: group 18; no charge; unreactive lanthanides: elements 58– 71 actinides: elements 90– 103 contain f orbitals main block (representative) elements: groups 1, 2, 13– 18
What family of elements has an ns 2 valence electron configuration? alkaline earth metals

Anomalies in the Electron Configurations Your best guide to writing e– configs is “The Table, ” but there a few exceptions. e. g. , Cr: [ Ar ] 4 s 1 3 d 5 Cu: [ Ar ] 4 s 1 3 d 10 These exceptions are due to the closeness in energy of the upper-level orbitals. Other exceptions are… Mo, Ru, Rh, and Ag. All of these exceptions have a single valence-level s electron. “Ru. Rh…!!”
- Slides: 12