Quantum History Columbus discovers America Pasteurization 1492 Max
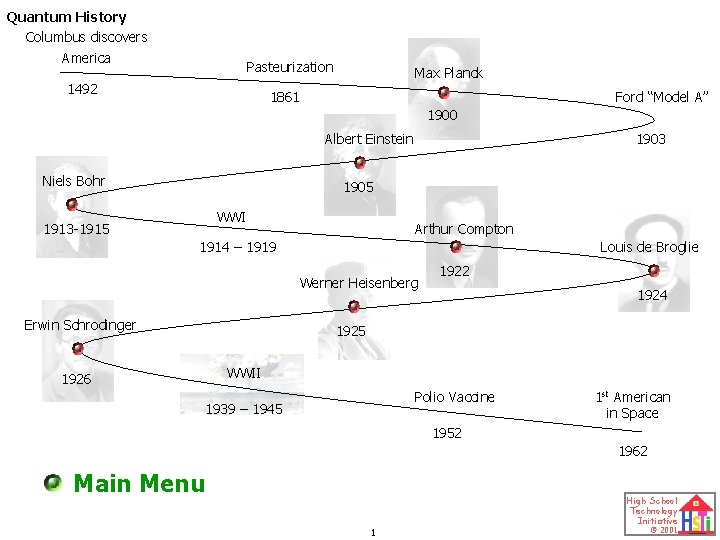
Quantum History Columbus discovers America Pasteurization 1492 Max Planck 1861 Ford “Model A” 1900 Albert Einstein Niels Bohr 1903 1905 WWI 1913 -1915 Arthur Compton 1914 – 1919 Louis de Broglie Werner Heisenberg Erwin Schrodinger 1926 1922 1924 1925 WWII Polio Vaccine 1939 – 1945 1 st American in Space 1952 1962 Main Menu 1 High School Technology Initiative © 2001
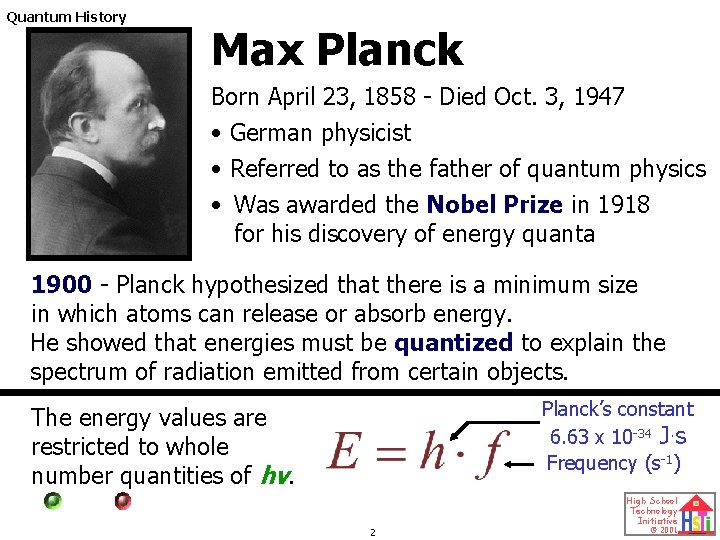
Quantum History Max Planck Born April 23, 1858 - Died Oct. 3, 1947 • German physicist • Referred to as the father of quantum physics • Was awarded the Nobel Prize in 1918 for his discovery of energy quanta 1900 - Planck hypothesized that there is a minimum size in which atoms can release or absorb energy. He showed that energies must be quantized to explain the spectrum of radiation emitted from certain objects. Planck’s constant 6. 63 x 10 -34 J. s Frequency (s-1) The energy values are restricted to whole number quantities of hv. 2 High School Technology Initiative © 2001

Quantum History Albert Einstein • Born March 14, 1879 - Died April 18, 1955 • German physicist • Awarded the Nobel Prize in Physics in 1921 for his discovery of the photoelectric effect 1905 - Einstein used Planck’s quantum theory to explain the photoelectric effect. • Every metal has a minimum frequency of light for which it will emit electrons. He hypothesized that the light striking the surface of the metal is a stream of tiny energy particles he called photons. The energy of each photon must be proportional to the frequency of the light. 3 High School Technology Initiative © 2001

Quantum History Niels Bohr • Born Oct. 7, 1885 - Died Nov. 18, 1962 • Danish physicist • Awarded the Nobel Prize in Physics in 1922 for his investigation of atomic structure and the radiation they emanate 1913 - Bohr developed a model of the hydrogen atom that explains its emission line spectrum. Bohr borrowed Planck’s idea that energies are quantized and proposed that only orbits of certain radii corresponding to defined energies are permitted. 4 High School Technology Initiative © 2001

Quantum History • Bohr assumed that electrons could “quantum jump” from one allowed orbit to another by absorbing or emitting photons of light with specific frequencies. • A photon is absorbed when an electron moves to an orbit with a higher energy state and is emitted when an electron moves to an orbit with a lower energy state. • Bohr’s model states that only photons of specific frequencies can be absorbed or emitted by the atom. these frequencies must correspond to the energy difference between two orbits. • Bohr’s model was important because it introduced the idea of using quantized energy stated for electrons in atoms. However, his model is only accurate for atoms or ions with a single electron. 5 High School Technology Initiative © 2001

Quantum History Arthur H. Compton • Born Sept. 10, 1892 - Died March 15, 1962 • American physicist • Awarded the Nobel Prize in Physics in 1927 for his discovery of the Compton Effect 1922 - Discovered that the wavelength of x-rays would increase when they are collided with and scattered by free electrons in matter. - This increase in wavelength means that the x-ray photons can transfer quanta of energy to the electron upon collision. This was a strong verification for quantum theory because explanation of the effect required that the x-rays be looked at as particles rather than as waves. 6 High School Technology Initiative © 2001

Quantum History Louis de Broglie • Born Aug. 15, 1892 - Died Mar. 19, 1987 • French physicist • Awarded the Nobel Prize in Physics in 1929 for his discovery of the wave nature of electrons. 1924 - De Broglie proposed that an electron in its orbit around the nucleus has a particular wavelength associated with it. Planck’s constant 6. 63 x 10 -34 J. s The wavelength of any particle is dependent on its mass and velocity. Mass (kg) Velocity (m/s) 1927 - Wave nature of the electron was experimentally proven using electron diffraction by Davisson and Germer. 7 High School Technology Initiative © 2001

Quantum History Werner Heisenberg • Born Dec. 5, 1901 - Died Feb 1, 1976 • German physicist • Awarded the Nobel Prize in 1932 for the creation of quantum mechanics. 1925 - Published his theory of quantum mechanics • Formulated his new theory in terms of matrix equations. 1927 - Uncertainty Principle • It is impossible to know both the exact momentum of an electron and its exact location in space at the same time. 8 High School Technology Initiative © 2001

Quantum History Erwin Schrodinger • Born Aug. 12, 1887 - Died Jan. 4, 1961 • Austrian physicist • Awarded a share of the Nobel Prize in Physics in 1933 for the development of his wave equation. 1926 - Wave Equation • Schrodinger’s wave equation includes both the wave-like and particle-like behavior of the electron. • The square of the wave function represents the probability that an electron will be in a certain place at a given instant of time. 9 High School Technology Initiative © 2001

Quantum History 10 High School Technology Initiative © 2001
- Slides: 10