Quality Management Beyond the PMBOK Christopher E Maddox

Quality Management Beyond the PMBOK® Christopher E. Maddox, PMP Vice President, Program Management Legacy Pharmaceuticals International Project Management Institute Baltimore Chapter 20 January 2011 This Presentation © 2011 Christopher E. Maddox. For use only by PMI members for non-profit educational purposes without express written consent of the copyright holder. Dilbert cartoons © United Features Syndicate and are used under Fair Use doctrine for educational purposes.

An Opening Thought

Objectives • Review Quality Management Definitions and Tools • Quality and Risk • The Economics of Quality • Understanding Causes and Effects • Statistical Tool Cautions and Tips

Quality Management Definitions QUALITY “the degree to which a set of inherent characteristics fulfill requirements” GRADE “category assigned to products or services having the same functional use but different technical characteristics” PMBOK® Guide 4 ed p 190

Quality Management Definitions WHICH IS HIGHER QUALITY? Hyundai Accent Mercedes Brabus SL

Quality Management Definitions WHICH IS HIGHER QUALITY? “Zero to 60 in under 6 seconds”

Quality Management Definitions WHICH IS HIGHER QUALITY? “Better than 25 MPG”

Quality Management Definitions QUALITY ASSURANCE “The process of auditing the quality requirements and the results from quality control measurements to ensure appropriate quality standards and operational definitions are used” QUALITY CONTROL “The process of monitoring and recording results of executing the quality activities to assess performance and recommend necessary changes” PMBOK® Guide 4 ed p 189

PMBOK® Quality Tools and Techniques • Cause and Effect Diagrams (Fishbone) • Control Charts • Flowcharting • Histogram • Pareto Chart • Run Chart • Scatter Diagram • Statistical Sampling • Inspection

Quality and Risk • Quality management ALWAYS involves assessment of risk • Risks to consider – Risk related to poor quality (scrap, recalls, reputation, liability, etc) – Risk related to sampling/inspection error • Complete elimination of risk is impractical in terms of time and cost; risk-based mitigation strategies are at the heart of effective QM • Understanding risks is the first step towards prevention of quality problems

Quality, Risk and Cost • Define Quality and Cost – Define Acceptable Quality Level – Define Cost of Quality and Risk/Cost of Poor Quality • Acceptable Quality Level 1. What are the user requirements / specifications? 2. What is needed to meet them? • Cost of Quality and Risk/Cost of Poor Quality 1. What is the cost of compliance (process, QA, QC)? 2. What is the cost and risk of poor quality (scrap, returns, etc)?
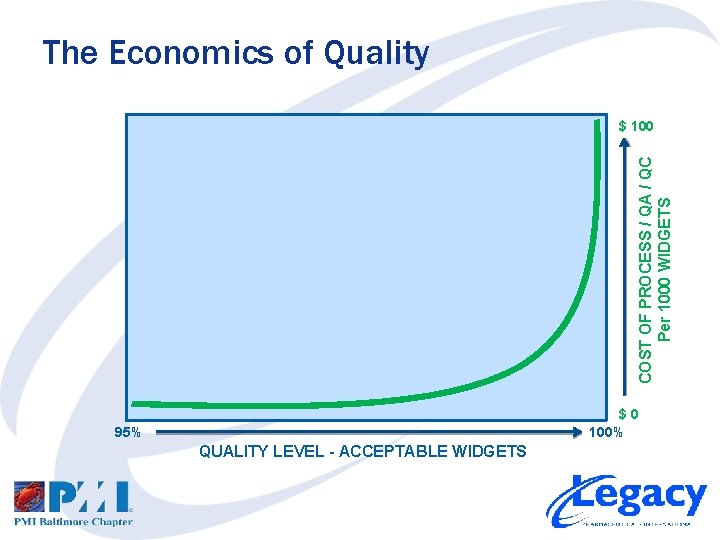
The Economics of Quality COST OF PROCESS / QA / QC Per 1000 WIDGETS $ 100 $0 100% 95% QUALITY LEVEL - ACCEPTABLE WIDGETS
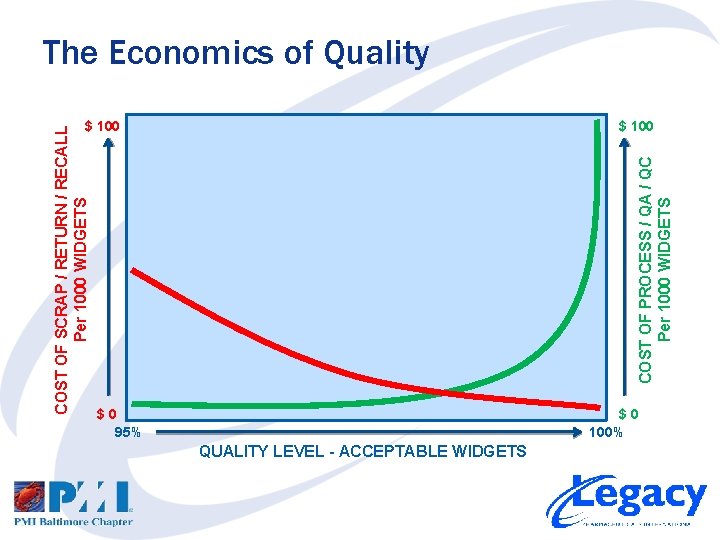
COST OF PROCESS / QA / QC Per 1000 WIDGETS COST OF SCRAP / RETURN / RECALL Per 1000 WIDGETS The Economics of Quality $ 100 $0 95% $0 100% QUALITY LEVEL - ACCEPTABLE WIDGETS
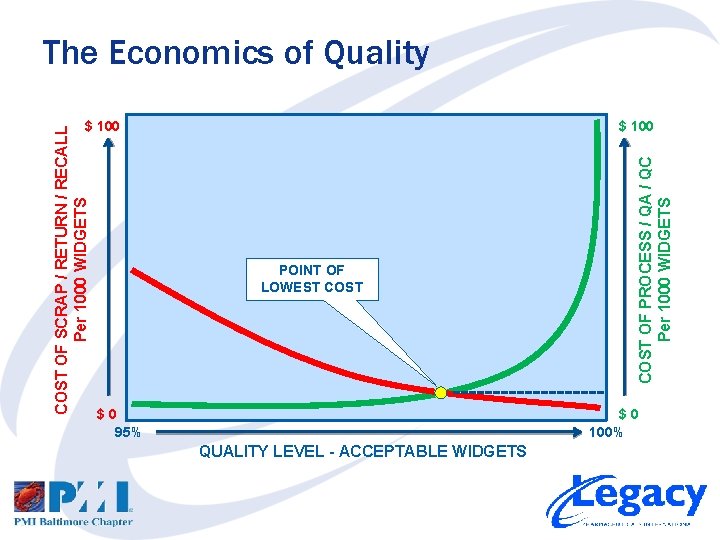
The Economics of Quality $ 100 POINT OF LOWEST COST $0 95% COST OF PROCESS / QA / QC Per 1000 WIDGETS COST OF SCRAP / RETURN / RECALL Per 1000 WIDGETS $ 100 $0 100% QUALITY LEVEL - ACCEPTABLE WIDGETS
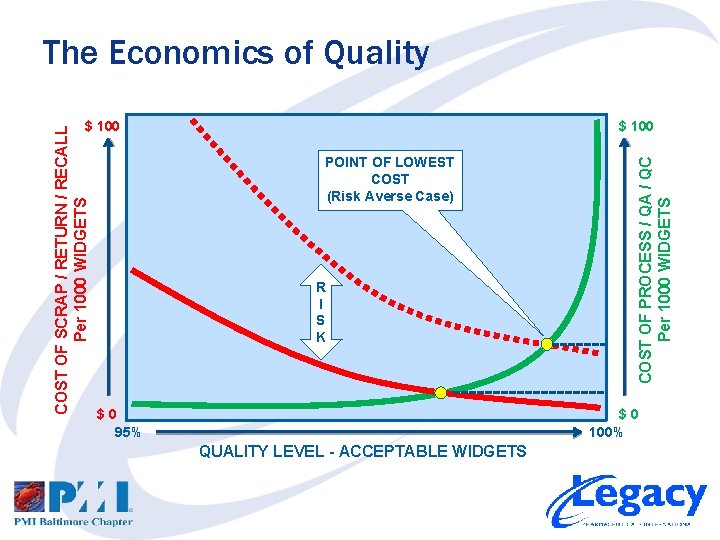
The Economics of Quality $ 100 POINT OF LOWEST COST (Risk Averse Case) R I S K $0 95% COST OF PROCESS / QA / QC Per 1000 WIDGETS COST OF SCRAP / RETURN / RECALL Per 1000 WIDGETS $ 100 $0 100% QUALITY LEVEL - ACCEPTABLE WIDGETS

The Economics of Quality • Investing in Quality – FIRST invest in capable processes – NEXT invest in Quality Assurance – LAST invest in Quality Control • Consider in your Risk Assessment… – Cost of lost customers – Cost of lost reputation – Cost of liability • Accepting less than Six Sigma Quality may not be sufficient to mitigate your risks!

So… what the heck is Six Sigma?

Six Sigma Quality • A measure of process capability • The simple definition – A process capable of producing not more than 3. 4 defects per 1, 000 opportunities – That’s 99. 9997% “Good” • The slightly-more-complex definition – Acceptance range of the process is 3 standard deviations (SD, Sigma or σ) of the process’ normal distribution on either side of the desired specification point
NUMBER OF OBSERVATIONS Six Sigma Quality DEVIATION BELOW SPECIFICATION DEVIATION ABOVE SPECIFICATION
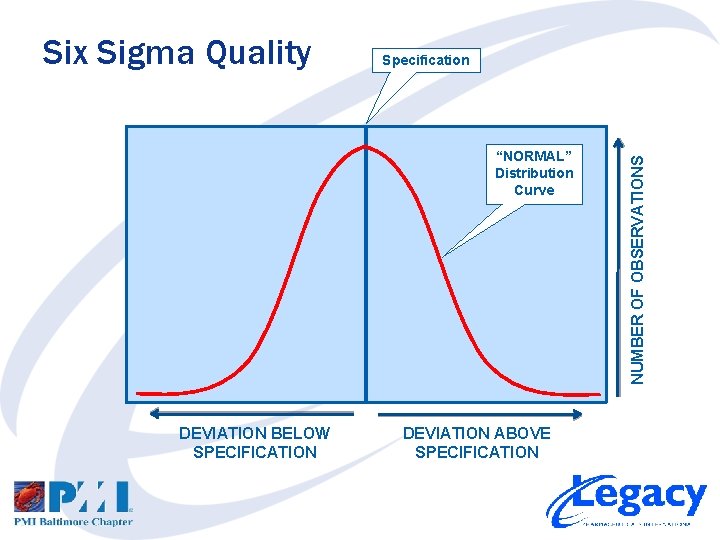
Specification “NORMAL” Distribution Curve DEVIATION BELOW SPECIFICATION DEVIATION ABOVE SPECIFICATION NUMBER OF OBSERVATIONS Six Sigma Quality
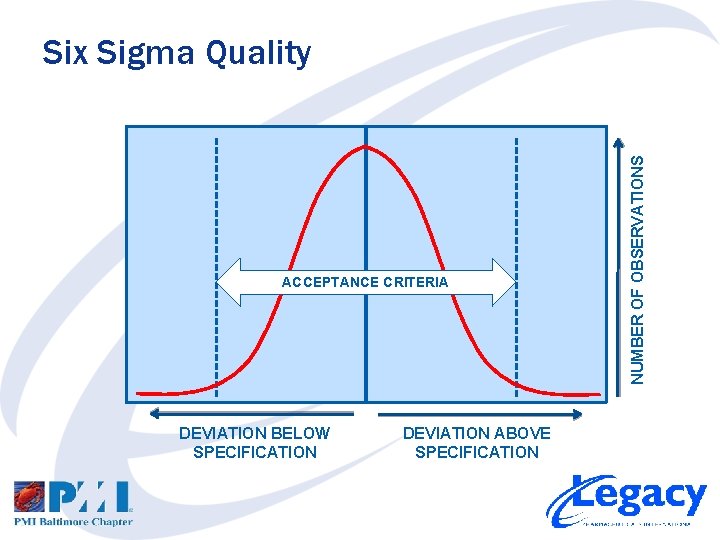
ACCEPTANCE CRITERIA DEVIATION BELOW SPECIFICATION DEVIATION ABOVE SPECIFICATION NUMBER OF OBSERVATIONS Six Sigma Quality
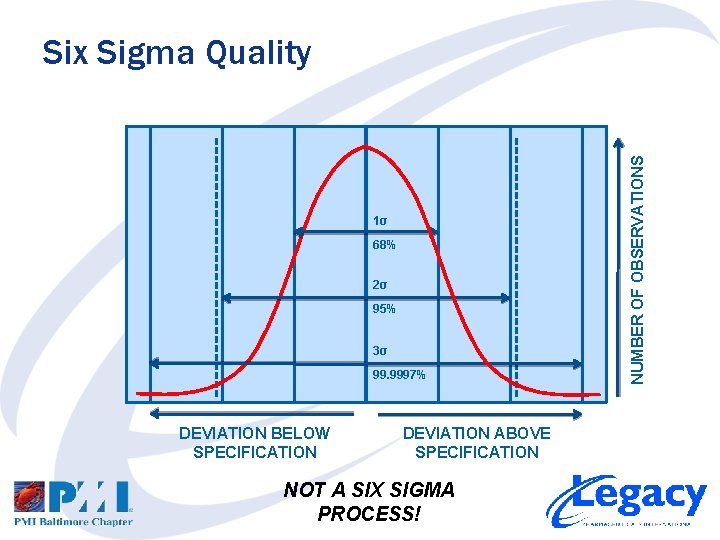
1σ 68% 2σ 95% 3σ 99. 9997% DEVIATION BELOW SPECIFICATION DEVIATION ABOVE SPECIFICATION NOT A SIX SIGMA PROCESS! NUMBER OF OBSERVATIONS Six Sigma Quality

1σ 68% 2σ 95% 3σ 99. 9997% DEVIATION BELOW SPECIFICATION DEVIATION ABOVE SPECIFICATION IMPROVE PROCESS CAPABILITY NUMBER OF OBSERVATIONS Conforming to Six Sigma
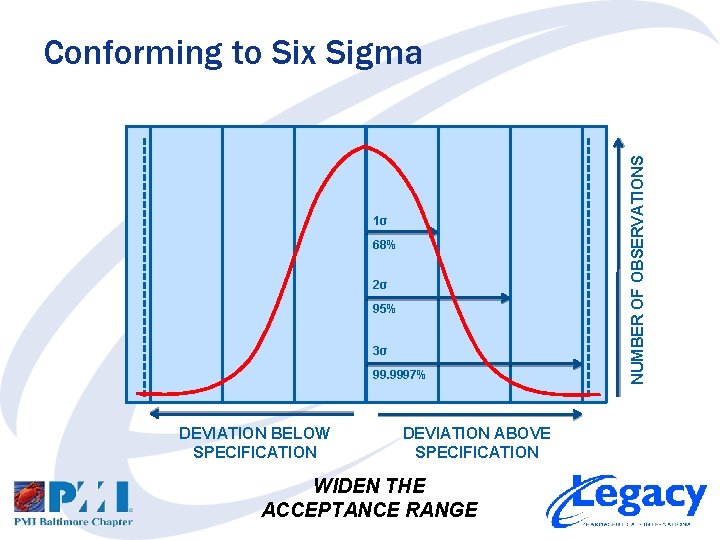
1σ 68% 2σ 95% 3σ 99. 9997% DEVIATION BELOW SPECIFICATION DEVIATION ABOVE SPECIFICATION WIDEN THE ACCEPTANCE RANGE NUMBER OF OBSERVATIONS Conforming to Six Sigma

Causes and Effects of Quality Problems

Cause and Effect of Quality Problems • Ishikawa (Fishbone) Diagram – Featured in the PMBOK® - a basic tool of investigating defects – Qualitative but not quantitative – Does not always help determine causality • Other Tools – Causal Circle explores cause-effect relationships of undesirable effects (UDEs) – suited to complex systems – Failure Mode and Effect Analysis (FMEA) systematically analyzes and prioritizes potential failures, consequences and preventative/corrective actions.

Causal Circle UDEs (Tablet Compression Project) 1. Tablet chipping 2. Tablet hardness variation 3. Coating is not uniform 4. Tablets under weight 5. Press force drifting during process 6. Granulation density low 7. Press process too slow 8. Speed control past limit 1. List and Number UDEs
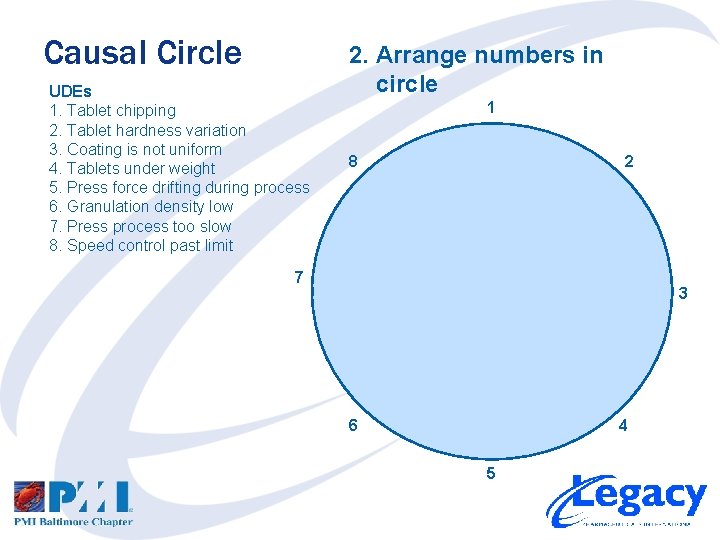
Causal Circle UDEs 1. Tablet chipping 2. Tablet hardness variation 3. Coating is not uniform 4. Tablets under weight 5. Press force drifting during process 6. Granulation density low 7. Press process too slow 8. Speed control past limit 2. Arrange numbers in circle 1 8 2 7 3 6 4 5
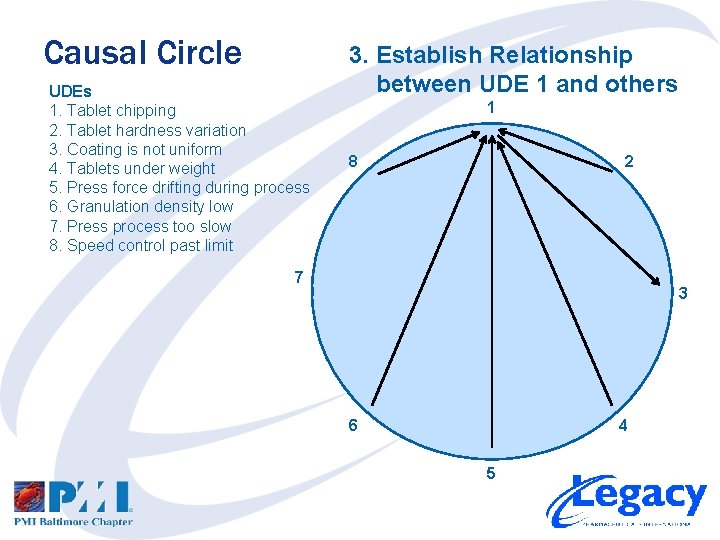
Causal Circle UDEs 1. Tablet chipping 2. Tablet hardness variation 3. Coating is not uniform 4. Tablets under weight 5. Press force drifting during process 6. Granulation density low 7. Press process too slow 8. Speed control past limit 3. Establish Relationship between UDE 1 and others 1 8 2 7 3 6 4 5
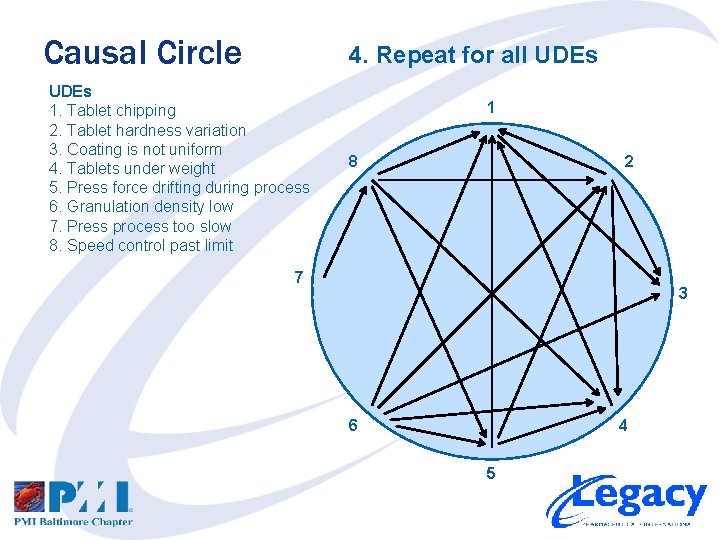
Causal Circle 4. Repeat for all UDEs 1. Tablet chipping 2. Tablet hardness variation 3. Coating is not uniform 4. Tablets under weight 5. Press force drifting during process 6. Granulation density low 7. Press process too slow 8. Speed control past limit 1 8 2 7 3 6 4 5
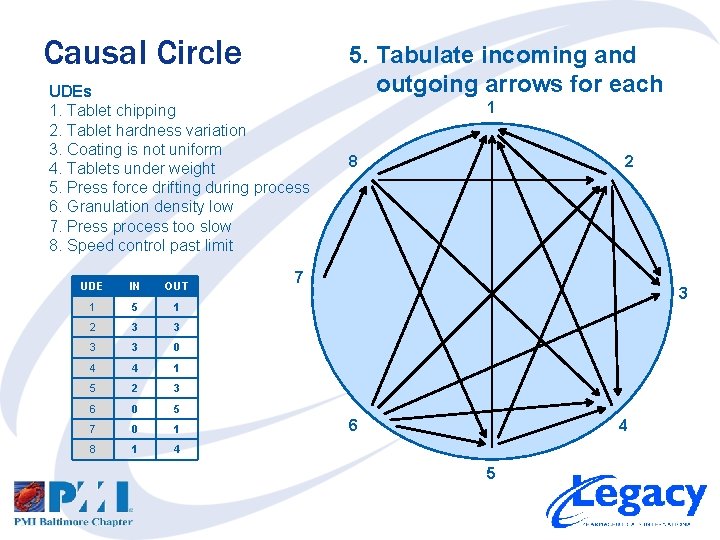
Causal Circle UDEs 1. Tablet chipping 2. Tablet hardness variation 3. Coating is not uniform 4. Tablets under weight 5. Press force drifting during process 6. Granulation density low 7. Press process too slow 8. Speed control past limit UDE IN OUT 1 5 1 2 3 3 0 4 4 1 5 2 3 6 0 5 7 0 1 8 1 4 5. Tabulate incoming and outgoing arrows for each 1 8 2 7 3 6 4 5
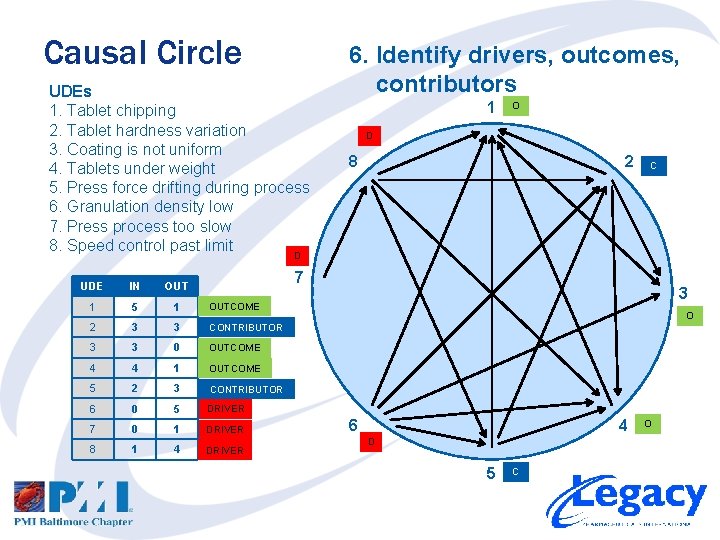
Causal Circle UDEs 1. Tablet chipping 2. Tablet hardness variation 3. Coating is not uniform 4. Tablets under weight 5. Press force drifting during process 6. Granulation density low 7. Press process too slow 8. Speed control past limit 6. Identify drivers, outcomes, contributors 1 O D 8 2 C D 7 UDE IN OUT 1 5 1 OUTCOME 2 3 3 CONTRIBUTOR 3 3 0 OUTCOME 4 4 1 OUTCOME 5 2 3 CONTRIBUTOR 6 0 5 DRIVER 7 0 1 DRIVER 8 1 4 DRIVER 3 O 6 4 D 5 C O

Failure Mode and Effect Analysis 1. 2. 3. 4. 5. 6. 7. 8. 9. Study the system, product or process Brainstorm possible range of failure modes for each process step List potential consequences of each failure mode Assign Severity (SEV) scores for each failure mode Identify cause(s) of each failure mode Assign Occurrence probability (OCC) scores for each cause Identify controls to detect the failure modes Assign a detection escape (DET) score for each control Calculate Risk Priority Number (RPN) for each FMEA line [RPN = SEV x OCC x DET] 10. Prioritize failure modes and causes based on RPN 11. Determine actions to be taken on failure and plans for prevention 12. Recalculate RPN based upon actions/plans and re-prioritize
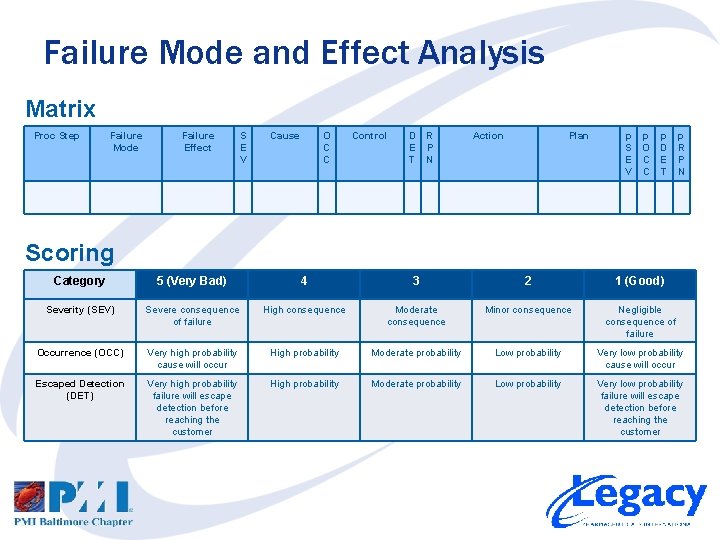
Failure Mode and Effect Analysis Matrix Proc Step Failure Mode Failure Effect S E V Cause O C C Control D E T R P N Action Plan p S E V p O C C p D E T p R P N Scoring Category 5 (Very Bad) 4 3 2 1 (Good) Severity (SEV) Severe consequence of failure High consequence Moderate consequence Minor consequence Negligible consequence of failure Occurrence (OCC) Very high probability cause will occur High probability Moderate probability Low probability Very low probability cause will occur Escaped Detection (DET) Very high probability failure will escape detection before reaching the customer High probability Moderate probability Low probability Very low probability failure will escape detection before reaching the customer

PMBOK® Statistical Tool Cautions & Tips • Fishbone: Beware that correlation ≠ causality (use Causal Circle for complex systems) • Control Charts: Look for off-center trends; plot a distribution if you have enough data • Pareto Charts: Beware that all items must be a direct cause of the defect or you can draw invalid conclusions • Scatter Plots: Do these in Excel and you can easily plot trend lines, derive formulas and R-squared values • Statistical Sampling: Subject to errors; know how to calc margin of error so you can create a valid sampling plan

Summary • Understand what quality is, and who defines it • Good quality management is dependent on good risk management • Understanding economics of quality is a competitive advantage • Process capability is the most important factor in quality and usually the most cost effective in the long run • Knowing cause, effect & failure mode is the only way to anticipate and investigate failure and continuously improve • Understand what tools and statistics are telling you • Quality is profitable unless it is just a buzzword – then it costs!

Questions? Comments?

Thank You! Chris Maddox chris. maddox@legacypharm. com +1 443 2220 This Presentation © 2011 Christopher E. Maddox. For use only by PMI members for non-profit educational purposes without express written consent of the copyright holder. Dilbert cartoons © United Features Syndicate and are used under Fair Use doctrine for educational purposes.
- Slides: 38