Quality Control Quality assurance in laboratories May 2007

Quality Control Quality assurance in laboratories May 2007 Laboratory Training for Field Epidemiologists

Learning objectives • At the end of the presentation, participants should – Understand the principles of Quality control – Understand the importance for internal and external quality control schemes Laboratory Training for Field Epidemiologists

Quality is. . • invisible when GOOD • impossible to ignore when BAD Laboratory Training for Field Epidemiologists

Quality ? • = sum-total of all the characteristics of a product/service that has a bearing upon the utilization of the product/service to the entire satisfaction of the consumer Laboratory Training for Field Epidemiologists

Quality ? • Consistency – Accuracy – Precision • Right result – First time – Every time Laboratory Training for Field Epidemiologists

Objectives of quality in lab • Support provision of high quality health-care – Reduce morbidity – Reduce mortality – Reduce economic loss • Ensure credibility of lab • Generate confidence in lab results Laboratory Training for Field Epidemiologists

Consequences of poor quality • Inappropriate action – Over-investigation – Over-treatment – Mistreatment • Inappropriate inaction – Lack of investigation – No treatment • Delayed action • Loss of credibility of laboratory • Legal action Laboratory Training for Field Epidemiologists

Quality assurance = Internal quality control + External quality assessment Continuously and concurrently assessing lab work Laboratory Training for Field Epidemiologists Retrospectif and periodic

Assessment of Quality System Man-driven • Audit, On-site inspection Material-driven • Quality Assessment • Internal • External • Schematic way: External Quality Assessment Scheme (EQAS( • Accreditation Laboratory Training for Field Epidemiologists

1 - Internal quality control in laboratory • = set of procedures undertaken by the staff to ensure quality of reports • Total process beginning with sample collection up to final reporting Laboratory Training for Field Epidemiologists

Quality in labs is mutual responsibility of…. . • Laboratory specialists • Clinicians • Public health physicians Laboratory Training for Field Epidemiologists
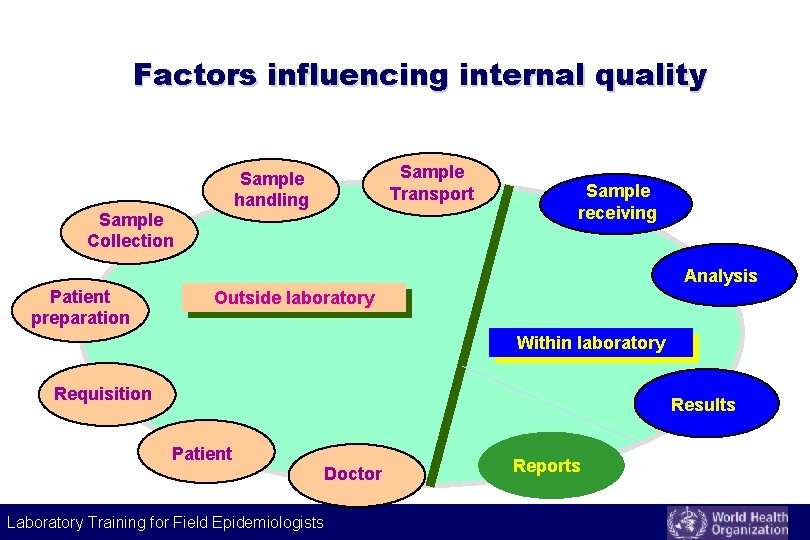
Factors influencing internal quality Sample Transport Sample handling Sample Collection Sample receiving Analysis Patient preparation Outside laboratory Within laboratory Requisition Results Patient Doctor Laboratory Training for Field Epidemiologists Reports

1. 1 - Factors influencing quality: Pre-analytical • Investigation – Ex: blood culture in the first week of enteric fever and not Widal • Specimen þRigh – Ex: No stool in SARS • Collection technique – Ex: Stool from bedpan→ collect stool in a clean container • Storage and transportation – t Laboratory Training for Field Epidemiologists Ex: Not kept in cold chain- overgrowth of other bacteria • Quantity – Ex: Not enough serum for serology • Labeling – Ex: Mismatch of sample • Laboratory – Ex: Not necessary test capacity
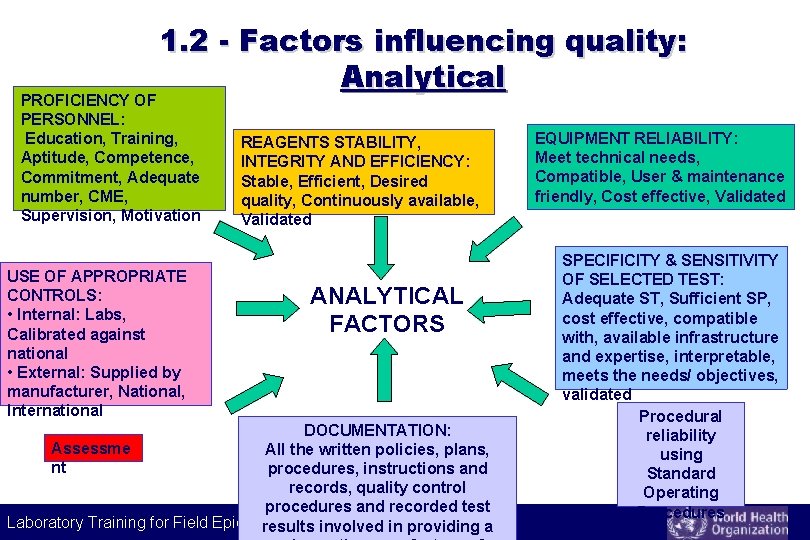
1. 2 - Factors influencing quality: Analytical PROFICIENCY OF PERSONNEL: Education, Training, Aptitude, Competence, Commitment, Adequate number, CME, Supervision, Motivation USE OF APPROPRIATE CONTROLS: • Internal: Labs, Calibrated against national • External: Supplied by manufacturer, National, International REAGENTS STABILITY, INTEGRITY AND EFFICIENCY: Stable, Efficient, Desired quality, Continuously available, Validated ANALYTICAL FACTORS DOCUMENTATION: Assessme All the written policies, plans, nt procedures, instructions and records, quality control procedures and recorded test Laboratory Training for Field Epidemiologists results involved in providing a EQUIPMENT RELIABILITY: Meet technical needs, Compatible, User & maintenance friendly, Cost effective, Validated SPECIFICITY & SENSITIVITY OF SELECTED TEST: Adequate ST, Sufficient SP, cost effective, compatible with, available infrastructure and expertise, interpretable, meets the needs/ objectives, validated Procedural reliability using Standard Operating Procedures

Documentation • If you have not documented it, • you have NOT done it … • If you have not documented, • it is a RUMOUR !!! Laboratory Training for Field Epidemiologists

Value of Documentation • Ensures processes and outcomes are traceable • Processes can be audited, thus external assessments can take place • Tool for training • Reminds you what to do next Laboratory Training for Field Epidemiologists

Standard Operating Procedures (SOP) • = comprehensively written document that describes the laboratory procedure and all other related issues • Essential for ensuring uniformity in laboratory procedures Laboratory Training for Field Epidemiologists SOP for Gram Staining

Validation • = is about determining whether something does what it is supposed to do Laboratory Training for Field Epidemiologists

Importance of validation • Validation - before you introduce something • Re-validation – after you have changed/modified – periodic • Validation is applied to: – SOP – reagents – equipment – software Laboratory Training for Field Epidemiologists

1. 3 - Factors influencing quality: Post-analytical • Right recording and reporting • Right interpretation – Range of normal values • Right turnaround time • Report to right user Laboratory Training for Field Epidemiologists

Reporting • Unequivocal message • Numerical value with units as and when required KISS ! Laboratory Training for Field Epidemiologists (Keep it short and simple)

Bottom-line • Quality costs , • but poor quality costs more … Laboratory Training for Field Epidemiologists € €

Training • The quality system is only as good as the staff who actually work with it • No matter how good the quality system is on paper, quality cannot be achieved if theory cannot be translated into practice • Training policy and plan • Training must include an understanding of why quality is important • Training should be need based, for all staff and reviewed Laboratory Training for Field Epidemiologists

2 - External Quality Assessment • 3 types, mainly 2: • An EQA organizer provides surveys in which identical material will be tested by all participating laboratories • ex: WHO/NHLS programme in Africa • Participating laboratories send specimens to EQA organizer for Rechecking • ex: Tuberculosis bacilloscopy quality control in Morocco • On-site visits with physical assessment) Laboratory Training for Field Epidemiologists

EQA • According to the ISO definition, EQA (also known as ‘proficiency testing’ (PT) or ‘EQ Control = EQC’) refers to: • a system of objectively checking laboratory results by means of an external agency • including comparison of a laboratory's result at intervals with those of other laboratories • the main objective being the establishment of trueness Laboratory Training for Field Epidemiologists

What makes microbiology different to other EQA schemes? • Microbiology samples are fundamentally non-uniform. • Microbiological taxonomy is fundamentally imprecise. • Microbiological samples are changing. • Traditional microbiological analysis depends upon behavior, not constitution. • Microbiology has many “right” answers. Laboratory Training for Field Epidemiologists

Objectives of EQA schemes for laboratories • Laboratory oriented objectives: • 1. Identifying possible deficiencies in laboratory practice, and guiding participants in any corrective actions to be taken for improvement; • 2. Identifying the reliability characteristics of particular methods, materials and equipment under routine conditions and suggest corrective actions as appropriate; • 3. Assessing and monitoring the impact of training; help for the preparation of future trainings Laboratory Training for Field Epidemiologists

Objectives of EQA schemes for laboratories • Public health oriented objectives: • 4. Providing the basis for the comparability of results during epidemiological surveillance and disease control • 5. Collecting information on laboratory measurements ( intra- and inter-laboratory) to alert professionals and/or government bodies about problems related to traceability and harmonization of results, and establish limits of acceptability of results as appropriate for a given purpose; • 6. Collecting information for the purpose of licensing or accreditation of laboratories; Laboratory Training for Field Epidemiologists

2. 1 Example of EQA organizer: CMPT, Canada Laboratory Training for Field Epidemiologists

2 -2 Rechecking (RC) • Participating laboratories send specimen to be rechecked on a regular basis to the EQA body • Targeted specimens and/or randomized specimens • Usually blind, can be single or multiple • Example of tuberculosis bacilloscopy in Morocco: – 180 centres in the country – All positives smears (targeted) – 10% of all negative smears (randomized) Laboratory Training for Field Epidemiologists

2. 3 On-site visits • Laboratory assessment • Laboratory licensing and/or accreditation • Combined with the other types of EQA • – After repeated problems (corrective action) – During on-site supervision (routine checking) – After training session (practical implementation of the training ) In addition to the assessment of the existing conditions, QC material can also be provided (slides, strain, sera, specimen for rapid tests …) Laboratory Training for Field Epidemiologists

The ideal situation: 2 types together and very targeted on-site visits • On-site visits: – Expensive, heavy – Only for one laboratory – Very much time consuming – Very effective if motivated staff – Very complementary to all other schemes especially rechecking • On-site visits should be used with « extreme » situations – « Initial » situation: laboratory assessment, licensing – « Bad » situation: repeated problems, failures in training – « Good » situation: accreditation Laboratory Training for Field Epidemiologists

3 - Accreditation • = process of inspection of laboratories and their licensing by a third party to ensure conformity to pre-defined criteria • Very very long task (As example, around 20% of French laboratories are accredited by COFRAC, it takes around 2 -3 years to follow the roadmap) • Last step of the entire process 1. Quality assurance (procedures, way of working) 2. IQC 3. EQC 4. Networking of the laboratories 5. … and then only accreditation if 1 -4 completed Laboratory Training for Field Epidemiologists

Carry home messages……. . • Quality assurance measures what a lab can do to improve reliability • As an epidemiologist, you may engage the laboratory in a dialogue and tactfully ask about QA measures in place • BE CAREFUL ! An epidemiologist is NOT in a position to assess the reliability of the lab or to evaluate its QA procedures as this requires a specific expertise Laboratory Training for Field Epidemiologists
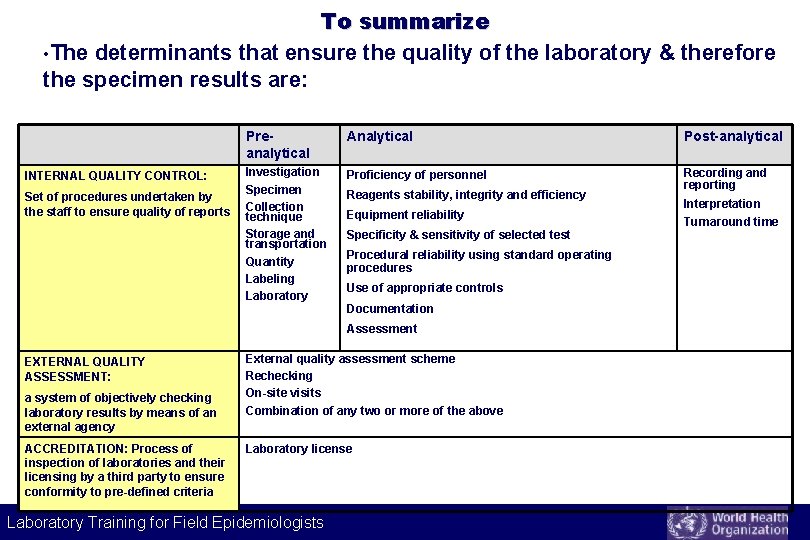
To summarize • The determinants that ensure the quality of the laboratory & therefore the specimen results are: INTERNAL QUALITY CONTROL: Set of procedures undertaken by the staff to ensure quality of reports Preanalytical Analytical Post-analytical Investigation Specimen Collection technique Storage and transportation Quantity Labeling Laboratory Proficiency of personnel Recording and reporting Reagents stability, integrity and efficiency Equipment reliability Specificity & sensitivity of selected test Procedural reliability using standard operating procedures Use of appropriate controls Documentation Assessment EXTERNAL QUALITY ASSESSMENT: a system of objectively checking laboratory results by means of an external agency ACCREDITATION: Process of inspection of laboratories and their licensing by a third party to ensure conformity to pre-defined criteria External quality assessment scheme Rechecking On-site visits Combination of any two or more of the above Laboratory license Laboratory Training for Field Epidemiologists Interpretation Turnaround time

Quality Control Developed by: The Department of Epidemic and Pandemic Alert and Response of the World Health Organization with the assistance of: European Program for Field Epidemiology Training Canadian Field Epidemiology Programme Thailand Ministry of Health Institut Pasteur Laboratory Training for Field Epidemiologists
- Slides: 36