Quality Control of Chagas diagnostics immunoassays Assay characteristics























- Slides: 24

Quality Control of Chagas diagnostics immunoassays: Assay characteristics and manufacturer’s reference panels. Gustavo A. Capriotti, Biochemist, R&D Manager WHO Consultation Meeting 27 -28 January 2009

Antigens used in Conventional Tests for T. cruzi infection: ü Whole Extracts or Semipurified Fractions of parasite (epimastigote) ü Purified Proteins ü Synthetic Peptides ü Recombinant Antigens WHO Consultation Meeting 27 -28 January 2009

Conventional serological tests • Indirect hemagglutination (IHA) § Parasite lysate • ELISA § Parasite lysate § Recombinant antigens WHO Consultation Meeting 27 -28 January 2009

Conventional serological tests Performance Method IHA ELISA Sensitivity > 97% > 98% WHO Consultation Meeting 27 -28 January 2009 Specificity > 98% > 99%

Kits ü Chagatest HAI screening A-V ü Chagatest ELISA (lysate) ü Chagatest ELISA recombinante v 3. 0 (FDA 510 k and CE) WHO Consultation Meeting 27 -28 January 2009

Kits ü New Chagatest ELISA recombinante v. 4. 0 (approved in LA/CE market) ü Chagatest ELISA recombinante for dried blood spot samples (approved in RA) ü Rapid test (in development) ü Colorimetric PCR (in development) ü Quantitative PCR (to start development this year) WHO Consultation Meeting 27 -28 January 2009

Chagatest ELISA recombinante v. 4. 0 Recombinant Antigens System with six recombinant Ags SAPA, Ag 1, Ag 2, Ag 13, Ag 30, Ag 36 • Highly sensitive and specific mixture • Proteins present in the trypomastigote stage • Proteins preserved in different parasite strains/linages WHO Consultation Meeting 27 -28 January 2009

Chagatest ELISA recombinante v. 4. 0 Recombinant Antigens Ag 1 Ag 2 Ag 30 Ag 13 Ag 36 Ag 2 Ag 13 SAPA Ag 13 Ag 36 SAPA Chronic Congenital Acute WHO Consultation Meeting 27 -28 January 2009

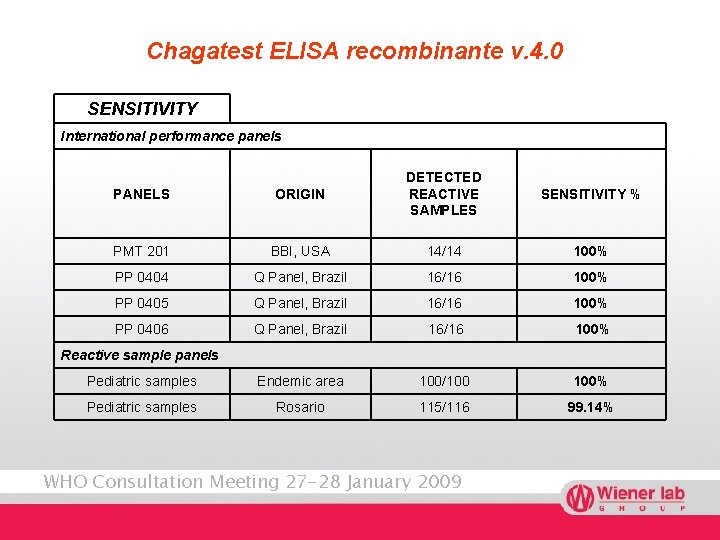
Chagatest ELISA recombinante v. 4. 0 SENSITIVITY International performance panels PANELS ORIGIN DETECTED REACTIVE SAMPLES PMT 201 BBI, USA 14/14 100% PP 0404 Q Panel, Brazil 16/16 100% PP 0405 Q Panel, Brazil 16/16 100% PP 0406 Q Panel, Brazil 16/16 100% Pediatric samples Endemic area 100/100 100% Pediatric samples Rosario 115/116 99. 14% SENSITIVITY % Reactive sample panels WHO Consultation Meeting 27 -28 January 2009

Chagatest ELISA recombinante v. 4. 0 SPECIFICITY SAMPLES SPECIFICITY % 1192 Blood Bank samples 99. 66 % 477 samples from different Health Centers 99. 57 % 474 samples from high prevalence population 98. 30 % 491 samples with different clinical conditions 98. 37 % WHO Consultation Meeting 27 -28 January 2009

Chagatest ELISA recombinante v. 4. 0 Interference with other pathologies Parasites Leishmaniasis Amebiasis Toxocariasis Toxoplasmosis Hydatidosis Teniasis 1/10 0/7 0/6 0/5 0/2 Infectious disease Hepatitis B Hepatitis C Syphilis HIV 0/22 0/28 0/19 0/25 Other pathologies Lymphoma 0/1 Myeloma 0/1 Lung tuberculosis 0/4 Autoimmune disorders 1/4 WHO Consultation Meeting 27 -28 January 2009

Recombinant antigens Advantages in serological diagnosis ü Standardized system uses a perfectly defined antigen composition. ü Antigens expressed in the infected trypomastigote stage of the parasite. ü Highly preserved antigens in different strains of the parasite. ü SAPA antigen, acute and congenital infection marker. WHO Consultation Meeting 27 -28 January 2009

How do we ensure Standardization? - At production level Recombinant antigens ü Recombinant antigens well characterized ü Perfectly defined antigen mix Parasitic Lysate ü Characterized lysate by WB ü Parasite culture under strict growth conditions. Well defined WCB & MCB WHO Consultation Meeting 27 -28 January 2009

How do we ensure Standardization? The recombinant antigens are tested separately using an ELISA technique with an internal panel of 9 positive samples specific for each antigen and 4 negative samples. WHO Consultation Meeting 27 -28 January 2009

How do we ensure Standardization? ELISAs Calibration 10 weak positive samples (IP between 1. 0 and 2. 0) 10 medium positive samples (IP between 2. 0 and 4. 0) 10 strong positive samples (IP < 4. 0) Note: samples diluted in negative or bovine serum may be used 20 negative samples WHO Consultation Meeting 27 -28 January 2009

How do we ensure Standardization? ELISAs Calibration A titer verifying the IP coefficient within a range of 0. 9 – 1. 2 must be selected. In addition, all negative samples must yield negative results. WHO Consultation Meeting 27 -28 January 2009

How do we ensure Standardization? ELISAs Titer verification - Internal panel of reactive samples evaluation: internal panel of 32 samples including weak, medium and strong. Acceptance criteria: the individual IP coefficient of each sample must be within a range of 0. 9 – 1. 2 WHO Consultation Meeting 27 -28 January 2009

How do we ensure Standardization? ELISAs Titer verification - Specificity: 200 sera / fresh plasmas Acceptance criteria: > 99%. If < 99%, the conjugate is diluted and retested. WHO Consultation Meeting 27 -28 January 2009

How do we ensure Standardization? ELISAs Final test - Internal panel: 2 positive control sera, 3 negative control sera, 6 weak positive, 10 medium and 10 strong samples. - Commercial panels: Chagas performance panel (QPanel, Brazil); BBI Panel WHO Consultation Meeting 27 -28 January 2009

How do we ensure Standardization? Hemagglutination - Titration of red blood cells sensitization. The titers are tested with an internal panel of 15 positive sera. Some of them diluted. Panel with 10 negative sera. Acceptance criteria: a titer where diluted sera match background titer of each sample is selected. Negative sera must yield negative results. WHO Consultation Meeting 27 -28 January 2009

How do we ensure Standardization? Hemagglutination Lot preparation - Internal panel of 20 positive sera - 100 negative sera WHO Consultation Meeting 27 -28 January 2009

How do we ensure Standardization? Hemagglutination Final verification - Chagas performance panel (QPanel) - BBI panel WHO Consultation Meeting 27 -28 January 2009

Reference Panel Considerations (proposal 2007): • Sample: SERUM (not plasma) • Number: 10 -12 (representing a range of reactivities from nonreactive to strongly reactive) • Not inactivated by heat (preferably aseptically filtered, photoinactivation, UV or g irradiation) • Preferable without preservatives • Representative from different disease stages and geographic regions • Selection made based on: IHA, ELISA, Immunoblot • For analytical sensitivity: diluted samples can be used • For clinical sensitivity: undiluted samples WHO Consultation Meeting 27 -28 January 2009

International Biological Reference Preparation for Chagas (2009) • A known reactivity standard is required to yield consistency lot to lot • To have a primary Standard of 2 reactive sera, as suggested, seems a good alternative. • This will allow to determine the analytical sensitivity for each lot, as being used for other international standards. WHO Consultation Meeting 27 -28 January 2009