QUALITY ASSURANCE IN THE CLINICAL LABORATORY Why do

QUALITY ASSURANCE IN THE CLINICAL LABORATORY

Why do laboratory errors occur? Understaffed Inadequate Attention To Detail Poor Workload Management Time Pressures Poor Sample Control Poor Results Verification Non-validated Tests Quality Control & Assessment Monitoring all areas of the work in the laboratory will decrease errors

Definition q Quality assurance is the coordinate process of providing the best possible service to the patient and physician q Quality assurance includes monitoring and controlling: o The competence of personnel, o Quality of materials, o Methods, reagents and instruments, o and the reliable reporting of test results 3

WHO Definition q. Quality assurance has been defined by WHO as: • The total process whereby the quality of the laboratory reports can be guaranteed. q. It has been summarized as: • The Right result, • At the Right time, • On the Right specimen, • From the Right patient, • With the result interpretation based on Correct reference data, • and at the Right price. 4

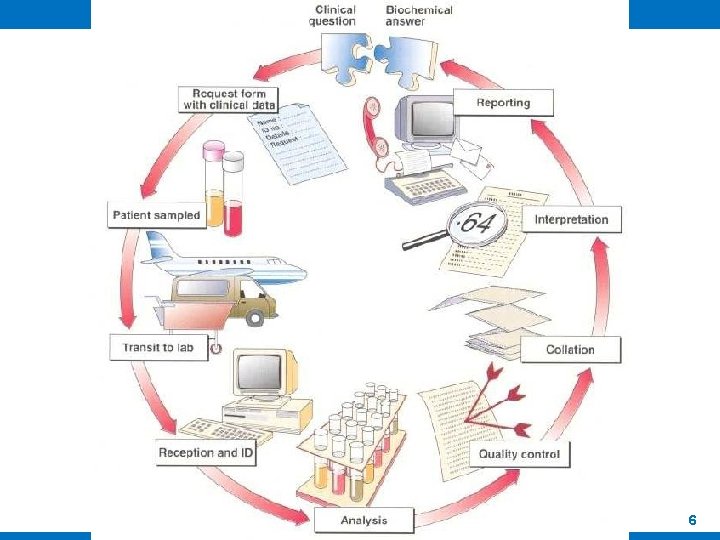
1 - Sources of Error q Erroneous results are at best a nuisance; at worst, they have potential for causing considerable harm q Errors can be minimized by: • Careful adherence to robust, agreed protocols at every stage of the testing process • This means a lot more than ensuring that the analysis is performed correctly q Errors can occur at various stages in the process: • Pre-analytical, occurring outside the laboratory, • Analytical, occurring within the laboratory, • Post-analytical, whereby a correct result is generated but is incorrectly recorded in the patient's record, 5
6

A- Preanalytical errors • This includes all the activities performed before the actual work (examination, analysis) is started PROCESS Test ordering Specimen acquisition POTENTIAL ERRORS • Inappropriate test • Handwriting not legible • Wrong patients ID • Special requirements not specified • • • Incorrect tube or container Incorrect patient ID Inadequate volume Invalid specimen (hemolysed or diluted) Collected at wrong time Improper transport conditions

B- Analytical errors PROCESS POTENTIAL ERRORS • Instrument not calibrated correctly • Specimens mix – up Analytical Measurement • Incorrect volume of specimen • Interfering substances present • Instrument precision problem

C- Post Analytical errors PROCESS POTENTIAL ERRORS • Previous values not available for comparison Test interpretation • Wrong patient ID Test reporting • Report not legible • Report delayed • Transcription error

2 - Aspects of a Good Quality Assurance Program q A good quality assurance program has three major aspects: A. Preventive activities B. Assessment Procedures C. Corrective actions 10

A- Preventive Activities q This helps to prevent error before it occurs by: o Improving accuracy and precision o Method selection o Careful laboratory design o Hiring of competent personnel o Development of comprehensive procedure manuals o Effective preventive maintenance programs 11

B- Assessment Procedures q Monitor the analytical process q Determine the type of error q Determine the amount of error q Determine the change in accuracy and precision q These activities include: o The testing of quality control material o Performing instrument function checks o Participating in proficiency testing programs (e. g. survey programs of accrediting agencies) 12

C- Corrective Actions q Correct errors after discovery q Communication with the users of laboratory's services q Review of work q Troubleshooting of instrument problems 13

3 - Quality Assurance versus Quality Control q Quality assurance is often confused with Quality control q Quality control involves the use of control samples to monitor the precision and accuracy of a test procedure q Control sample is processed along with the patient samples and the results are compared to the expected values and interpreted as acceptable or unacceptable q Quality control is an important part of a quality assurance program 14

A- Accuracy and Precision q Accuracy is the measure of "truth" of a result q Accurate results reflect the "true" or correct measure of an analyte or identification of a substance 15

A- Accuracy and Precision q Precision is the expression of the variability of analysis, reproducibility of a results, or an indication of the amount of random error q Precision is completely independent of accuracy or truth q A procedure can be precise, as determined by repeat analysis, but the result can be inaccurate q Three terms are widely used to describe the precision of a set of replicate data: o standard deviation; o variance; o coefficient of variation 16

A- Accuracy and Precision Good Accuracy Good Precision Only Neither Good precision Nor Accuracy 17

A- Accuracy and Precision q Both methods are equally precise, but in method D the mean value differs from the true value q The mean for method C is equal to the true value q Both methods are equally precise, but method C is more accurate 18

A- Accuracy and Precision • The graph shows the distribution of results for repeated analysis of the sample by different methods • The mean value is the same in each case, but the scatter about the mean is less in method A than in method B • Method A is, therefore, more precise 19

B- Types of Errors When Errors Occur ? q Errors occur when there is a loss of accuracy and precision q A primary goal of quality assurance is to reduce and detect errors or to obtain the best possible accuracy and precision 20

B- Types of Errors q Mistakes jeopardize patient care and must be detected and avoided at all times q An error is the difference between the result obtained and the result expected a. Random errors b. Systematic errors 21

a- Random Errors q Occur without prediction or regularity q Affect measurement of precision and causes data to be scattered more q Random errors occur as the result of: o Carelessness, o Inattention, o when taking short cuts in procedures, o Mislabeling specimens, o Incorrect filing of reports, o Reporting of wrong result to the wrong patient 22

b- Systematic Errors q Errors within the test system of methodology q Affect the accuracy of results o Causes the mean of a data set to differ from the accepted value q Examples include: o Incorrect instrument calibration o Unprecise or malfunctioning dilutors and pipettes o Reagents that lost their activity o Quantitative tests being read at an incorrect wavelength o Reagents are not prepared from sufficiently pure chemicals 23

b- Systematic Errors q Types of systematic errors A. Proportional systematic error or bias § It grows larger as the concentration of analyte grows B. Constant systematic error "constant bias" § A constant amount over the entire range of the analysis process § The magnitude of a constant error does not depend on the size of the quantity measured 24

b- Systematic Errors 25

c- Detection of Errors q Analyzing standard samples o The best way to estimate the bias of an analytical method is by analyzing standard reference materials, materials that contain one or more analytes at well-known or certified concentration levels q Using an independent analytical method o The independent method should differ as much as possible from the one under study to minimize the possibility that some common factor in the sample has the same effect on both methods q Performing blank determinations q Varying the Sample Size o As the size of a measurement increases, the effect of a constant error decreases. Thus, constant errors can often be detected by varying the sample size. 26

4 - Benefits of an Effective quality Assurance Program q Correct and timely presentation of data to the physician q Improvement of precision and accuracy q Early detection of mistakes q More efficient and cost effective use of materials and personnel q Meeting the requirements of inspection and accreditation agencies q Development of accurate and concise procedures and manuals q Measure of productivity of personnel and instrumentation. 27

PERSONNEL, STAFF DEVELOPMENT & QUALITY ASSURANCE

1 - Introduction q The most expensive and complex resource in any organization is its' employees q Choosing the appropriate individuals for the job and managing them effectively is one of the most difficult and powerful means available to prevent errors in the laboratory 29

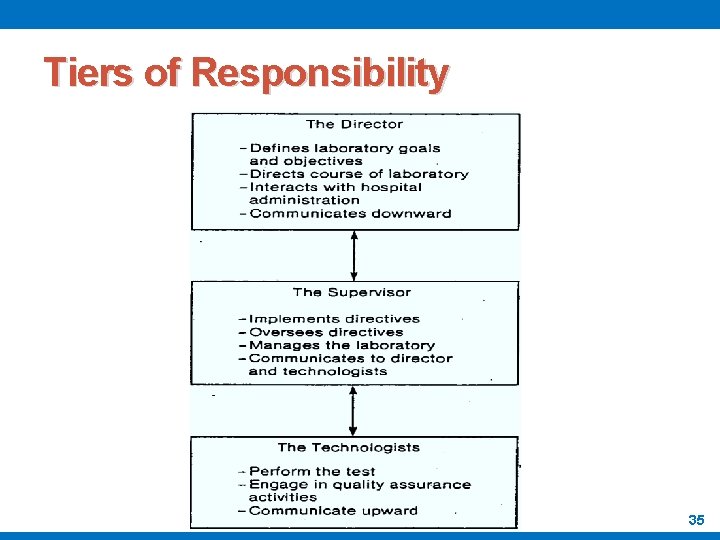
1 - Responsibility within a Quality Assurance Program q There are three tiers of responsibility in a well - designed quality assurance program: o The director, the supervisor, and technologist or technicians q These levels exist in all laboratory organizations regardless of size q A small hospital laboratory may have a director, one supervisor, and handful of technologists q The director in the laboratory service of a large medical center may have an associate and assistant directors as well as a support staff that works for and reports to him q The supervisor tiers in a larger organization can be divided into sections such as hematology, chemistry, or microbiology and the technical level can include numerous technologists and technicians 30

A- The director q His role is to direct and set policy q He is responsible for deciding the direction of the laboratory in the near and far future q He is responsible for defining goals, objectives and performance standards q He is responsible for communications with other levels concerning what is expected of them and keep them informed about the laboratory’s position in the hospital organization q He is responsible for communicating to the users of the laboratory's services, capabilities and limitations 31

B- The Supervisor q His job is to implement the objectives set forth by the q q q director He should be a competently certified technologist capable of understanding the role of the laboratory in providing health care His responsibilities include both administrative and technical ones He must be sure that each member of the technical staff is aware of his responsibilities He should maintain work conditions in which goals are fulfilled He is responsible for interviews, hiring, evaluation and disciplines the technical staff 32

B- The Supervisor q He is the direct connection between the director and the technical staff q His communications must be two-way: o Downward from director to the technologists, o And upward from the technologists to the director. q He should be capable of managing an effective quality assurance program to provide the required level of service 33

C- The Technical Staff q They should be trained how to operate the quality assurance program q They should be qualified and continue to update their level of competence by participating in continuing education programs q They perform the laboratory workload q The success of any laboratory quality assurance program depends on the dedication of the technical staff in providing a top - quality level of performance. 34
Tiers of Responsibility 35

2 - Tools of the Trade q There are some personnel management tools that should be part of the quality assurance system: A. Intralaboratory Communication B. Orientation and Trading C. Personnel Manuals D. Personnel Files E. Workload Recording 36

A- Intralaboratory Communication q Communication is an essential part of any enterprise q Laboratory communication can be improved by memorandums (memos), newsletters, laboratory log book q Memorandum is used to emphasize policy or information of general interest to the technical staff q Newsletters are used for making routine announcements, social events, giving credit for extraordinary work etc. . q Laboratory logbook is for every one left open, record daily events, serves as a record of the shift- to- shift, day - to - day life in the laboratory 37

B- Orientation and Training q A Well - informed technical staff is the best defense against q q serious errors The orientation of new employees to the laboratory's policies and procedures should be a well - planned and well executed quality assurance activity The orientation program should be well - structured and carried out within and appropriate time period For orientation, a new employee should have a written job description and a training checklist covering the important technical and non technical facets of the job. The completed checklist should be a part of the employee's permanent personnel file Participation in continuing education programs should be encouraged as there are rapid advances in technology and new discoveries in laboratory medicine 38

C. Personnel Manuals q Each laboratory should have available at all times a copy of the hospital's and laboratory personnel policies q These manuals should clearly outline work rules, disciplinary guidelines, sick and holiday benefits, work hours and the method by which compensation is calculated q Each new employee should be required to read the personnel manual during his orientation q If everyone is aware of work rules and employee rights, misunderstanding and unproductive attitudes can be avoided and a better opportunity to work in harmony will exist 39

D- Personnel Files q It is the record of the employee’s experience within the laboratory and should contain the following in a neat and organized manner: a) b) c) d) e) f) g) h) Job description Payroll information "rate of pay, vacations, sick leave". Resume "showing qualification, letters of recommendations etc. . . Periodic performance evaluation records Reports of any disciplinary actions Documentation of continuing education Training or orientation records, Records of physical examinations, immunization Records of radiation exposure q N. B. The personnel files should be treated with confidentiality by supervisor and employee 40

E- Workload Recording q A tool to document the time required to perform the laboratory workload q A unit value has been assigned for each procedure, based on the length of time required to prepare the specimen for analysis, to perform the analysis, and report the results q It can be applied for comparison within and between laboratories, it reflects the efficiency of the laboratory in performing its tasks 41
- Slides: 41