Quality and Compliance Challenges for Biopharmaceutical Products Ramon

Quality and Compliance Challenges for Biopharmaceutical Products Ramon Rivera Gonzalez, Ph. D. Director Quality Assurance Amgen Manufacturing, Limited

Outline § Background Information § Regulatory and Process Challenges § Quality by Design § Risk Management § Process Analytical Technology § Key Quality Systems • Deviations/Nonconformities • Corrective and Preventive Actions § Biological Product Deviations § FDA and EMEA Observations 2

Biotechnology is a set of scientific techniques used to derive valuable products from living organisms Applications § biopharmaceutical drugs § agriculture § waste management 3

Background information § FDA- “sterile drugs should be manufactured by aseptic processing only when terminal sterilization is not feasible”. § Parenterals -Drug administration other than by the mouth or rectum- ex. Injection, infusion or implantation. – Biological products (vials or syringes) • solubility, stability, maintain activity 4
Biotechnology CHEMISTRY ENGINEERING BIOLOGY 5

Biological Products Manufacturing 6

Biological Products Manufacturing 7
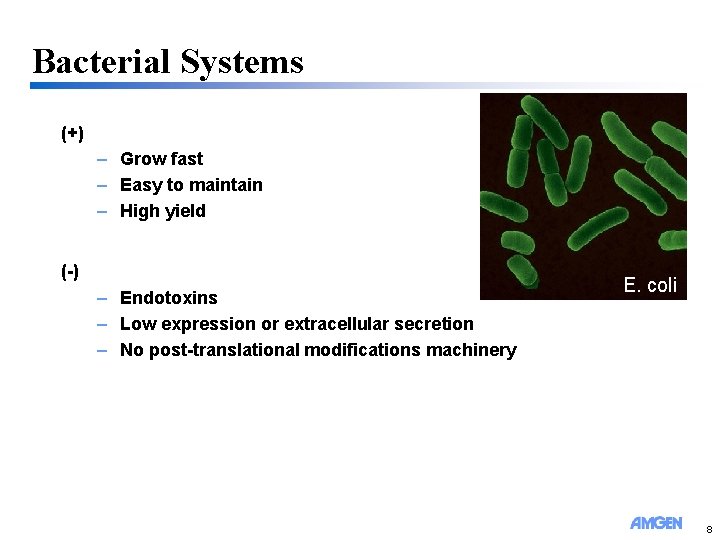
Bacterial Systems (+) – Grow fast – Easy to maintain – High yield (-) – Endotoxins – Low expression or extracellular secretion – No post-translational modifications machinery E. coli 8
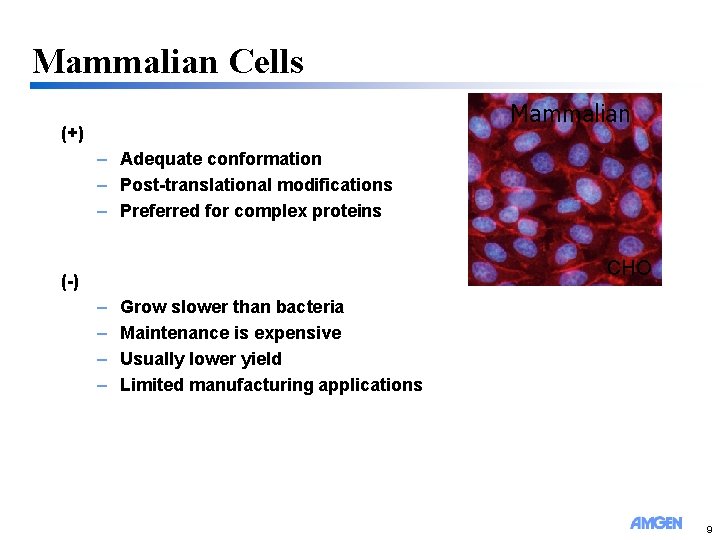
Mammalian Cells Mammalian (+) – Adequate conformation – Post-translational modifications – Preferred for complex proteins CHO (-) – – Grow slower than bacteria Maintenance is expensive Usually lower yield Limited manufacturing applications 9

Quality and the Product Life Cycle § Adherence to regulations § Control and maintenance of documentation § Quality of suppliers, components and raw materials § Reliability and consistency § Monitor/audit of the manufacturing process § Deviations, unexpected situations - product impact § Correction and prevention - CAPA § Lot release/rejection decision § Customer complaints § Continuous improvement 10

Quality Systems Emphasis § Quality Management § Quality Assurance § Risk Management – Evaluation analysis and quality risk management tools § Preventive Action § Promote product and process improvement (i. e. , continuous improvement) § “Continuous Improvement” of the Quality System 11

Regulatory Environment Challenges § Transfer of CBER products to CDER § GMPs for the 21 st. Century § Aseptic Processing Guideline § Risk management § Increased scrutiny of product insert claims § Focus on patient safety § Process Analytical Technology (PAT) 12

Regulatory Environment Challenges § Quality by Design § Bioterrorism § Animal-derived materials § Country-specific regulatory requirements – Mexico, Brazil, Saudi Arabia, Japan § Biogenerics in EU § State of the art technology – Isolators 13

Major Process Challenges • Sterile vs. Aseptic • Requires the application of microbiological contamination control to prevent infectious organisms to be present in the sterile product • Demonstrate “CONTROL” of the process, while technical complexity increases • Characterization to identify variability components • Application of science and new technologies • Maintenance of the cell lines • Contamination risks • Personnel as “incubators” • Source of microbial load 14

Quality by Design § “Quality can not be tested into products; it has to be built in by design” § Product quality and performance requires efficient design of manufacturing processes § Product specifications based on deep understanding of how formulation and process factors impact product performance § It provides a framework for continuous "real time" assurance of quality and continuous Improvement 15

The importance of Design § Multidimensional combination and interaction of input variables and process parameters that have been demonstrated to provide an assurance of quality § Operating within design parameters will produce a product meeting designed quality attributes § Working within the design parameters is not considered a reportable change § Movement outside of design space is considered a change – subject to regulatory approval 16

Risk Management § What are the potential hazards to process and product ? • Identify potential hazards both prospectively and in a reactive mode § Applies to components, container closure, raw materials, dosing devices, manufacturing process, drug substance, intermediates § How these factors influence variability of process, product performance, product safety and efficacy? 17

Risk Management § Risk management is a regulatory expectation for a modern quality system § Risk assessment, risk control, risk communication and risk review throughout product lifecycle § Decisions should be based upon process and product understanding • Balance between the use of risk management and compliance with GMPs § Always include intended use, patient safety and availability 18

Process Analytical Technology § “…any system for continuous analysis and/or control of manufacturing processes based on real-time measurements, or rapid measurements during processing. ” • Source; FDA Human Drug c. GMP Notes, Q 1 2002 19

Process Analytical Technology § “At line” - the sample is removed, isolated from, and analyzed in close proximity to the process stream § “On-line” - the sample is diverted from the manufacturing process, and may be returned to the process stream § “In-line” – the sample is not removed from the process stream and can be invasive or non-invasive • Source: FDA PAT Guidance 20

PAT - Benefits § Enhancement of process understanding § Sources of variability identified and explained § Meet requirements for validating and controlling process § Quality attributes can be accurately and reliably predicated § Continuous improvement § Integration of development, manufacturing, QA and knowledge management § Acceptability of in-process materials and final product based on process data 21

Management Controls § Written quality policy and objectives § Management reviews – regulations and quality objectives – attendance documented – results, action plans/corrective actions documented § Internal audits – auditors no direct responsibility for matters being audited 22

Deviations/Nonconformities § Classification based on impact/risk § Investigations need to be thorough, stand-alone – Root cause analysis – Product impact • Stability data, intrinsic vs. extrinsic, historical data – Toxicological/health hazard evaluations based on route of administration to patient – Corrective and Preventive actions 23

Corrective and Preventive Actions (CAPA) § Corrective = correct existing nonconformity § Prevention = potential recurrence of nonconformity Regulatory expectations: – Identify sources of problems/nonconformities • Unfavorable trends – – – Prioritize based on risk Defined action plans Timely implementation Measure and document effectiveness Reviewed by Management 24

If a deviation/nonconformity involves distributed product… § Evaluate per 21 CFR 600. 14 – Biological Product Deviation (BPD) § “must report any event and any information relevant to the event associated with the manufacturing, to include testing, processing, packing, labeling or storage, or with the holding or distribution, of a licensed biological product if the event meets the following criteria: (1) Either; (i) Represents a deviation from c. GMP, applicable regulations, applicable standards, or established specifications that may affect the safety, purity or potency of that product; or (ii) Represents an unexpected or unforeseeable event that may affect the safety, purity, or potency of that product; and (2) Occurs in your facility or a facility under contract with you; and (3) Involves a distributed biological product 25

Some points regarding BPDs § Distributed = biological product has left the control of the licensed manufacturer • Contract manufacturing § The decision to report should be based on whether the event had the potential to affect safety, purity, or potency of the product § The license holder is required to file at a date not to exceed 45 calendar days from the date that you, your agent, or another person, acquire information reasonably suggesting that a reportable event has occurred § Must report even if the investigation determined that there was no impact 26

BPD- Reportable Examples § Raw materials that failed specifications used in manufacturing § Aseptic processing not performed according to procedures § Stability testing not performed at required interval § Missing information in label (product type, lot number, storage temperature, concentration) § Distributed product prior to completion of required testing § Product release prior to validation of manufacturing process § Biological Drug Substance stored at the incorrect temperature 27

BPD – Non-reportable Examples § Raw material did not meet specification and was rejected prior to its use § Testing performed incorrectly, invalidated and repeated and found acceptable prior to distribution § Product labeled with a shortened expiration date (not due to failure to meet specification) § Product shipped to the incorrect facility § Customer order filled incorrectly (wrong product, wrong amount), but labeled correctly 28

EMEA Observations § Critical = give rise to product that could be harmful to patient § Major = result in product not meeting marketing authorization, major deviation from EU GMP § Other = usually lack information to be classified 29

EMEA 1995 - 2005 § Critical • Design and maintenance of premises • Potential for microbiological or chemical/physical contamination § Major/Other • Potential for microbial contamination • Documentation- Quality system elements/procedures • Unauthorized activities requiring regulatory filing • Design and maintenance of equipment and premises 30

FDA Observations (1996 -2006) § Failure Investigations (Nonconformities) – Failure to conduct investigations – Failure to extend investigations to related batches and products – Averaging OOS results with in-specification results to support release – Delays in starting the investigation – Incomplete investigation into root cause – Failure to take immediate corrective action – Failure to address product impact 31

FDA Observations (1996 -2006) § Record keeping – Batch Production and Control records do not include complete information – “post-it” notes with hand-written original data – Lack of procedures – No revision history for procedures § Validations – – Discrepancies against Master Validation Plan Inadequate cleaning validation Inadequate process validation Inadequate validation of analytical methods 32

In Summary § The successful application of quality systems for the manufacturing of biological products requires the comprehensive synergy between: regulations, science/new technologies, process knowledge, Management accountability and efficient risk management 33

Questions? 34
- Slides: 34