Qualitative Tests for Amino Acids There a number

Qualitative Tests for Amino Acids • There a number of qualitative tests to detect the presence of amino acids • These are largely dependent on the nature of R-group.

Exp. 1 Ninhydrin Reaction • A color reaction given by amino acids and peptides on heating with the chemical ninhydrin. • The technique is widely used for the detection and quantitation (measurement) of amino acids and peptides. • Ninhydrin is a powerful oxidizing agent which reacts with all amino acids between p. H 4 -8 to produce a purple colored-compound. • The reaction is also given by primary amines and ammonia but without the liberation of Co 2 • The amino acids proline and hydroxyproline also reacts but produce a yellow color.

Exp. 1 Ninhydrin Reaction ■ Method: • 1 ml AA + 1 ml NH • heat in boiling WB for 5 min. • Purple color.

Exp. 1 Ninhydrin Reaction ■ Method: • α-amino acid + 2 ninhydrin ---> CO 2 + aldehyde + final complex (purple) + 3 H 2 O • In summary, ninhydrin, which is originally yellow, reacts with amino acid and turns deep purple. It is this purple color that is detected in this method.
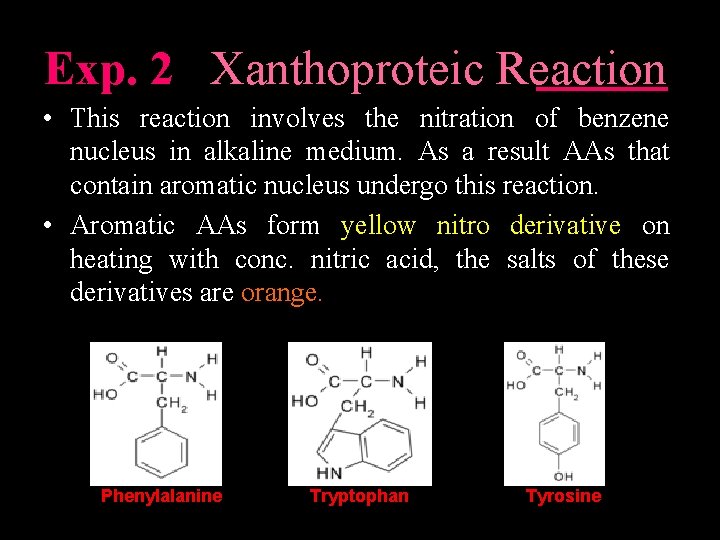
Exp. 2 Xanthoproteic Reaction • This reaction involves the nitration of benzene nucleus in alkaline medium. As a result AAs that contain aromatic nucleus undergo this reaction. • Aromatic AAs form yellow nitro derivative on heating with conc. nitric acid, the salts of these derivatives are orange. Phenylalanine Tryptophan Tyrosine

Exp. 2 Xanthoproteic Reaction ■ Method: • • 1 ml AA + 1 ml conc. HNO 3 heat the mixture in WB for 30 s cool add drop-wise 40% Na. OH to render the solution alkaline • Yellow to orange color.
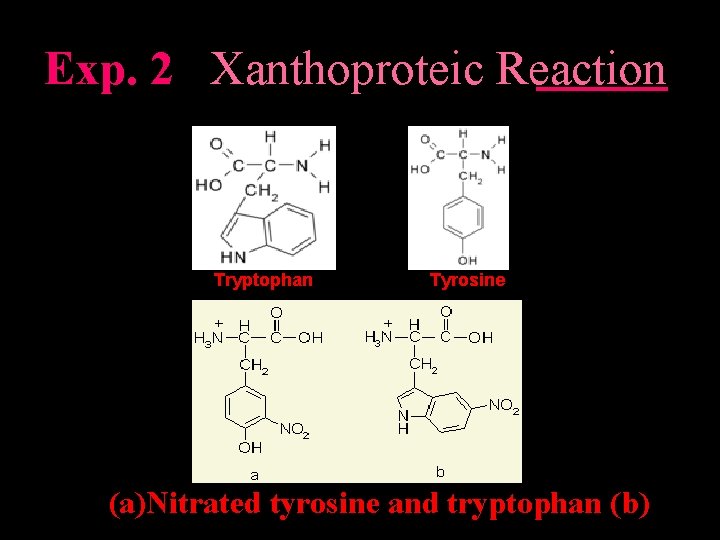
Exp. 2 Xanthoproteic Reaction Tryptophan Tyrosine (a)Nitrated tyrosine and tryptophan (b)

Exp. 3 Millon Reaction • This reaction is used to detect the presence of phenol (hydroxybenzene) which reacts with Millon's reagent to form red complexes. • The only phenolic AA is tyrosine. Tyrosine

Exp. 3 Millon Reaction ■ Method: • • • 1 ml AA + 5 drops of Millon reagent heat the mixture in BWB for 10 min cool too room temp add 5 drops of Na. NO 2 Brick red color.

Exp. 4 Hopkin-cole Reaction • This reaction is used to detect the presence of indol group • The indol group of tryptophan reacts with glyoxalic acid in the presence of conc. H 2 SO 4 to give purple color. Tryptophan

Exp. 4 Hopkin-cole Reaction ■ Method: • 1 ml AA + 1 ml Hopkin-cole reagent • mix well • Carefully pour conc. H 2 SO 4 down the side of the tube so as to form two layers • Purple ring at the interface.

Exp. 5 Sulfur Reaction • This reaction is specific to detect the presence of sulfur. • The sulfur of cystein and cystine is converted to inorganic sulfide with conc. Na. OH. Lead acetate is added and a ppt of black lead sulfide indicates a +ve reaction. Cystein

Exp. 5 Sulfur Reaction • 2 ml AA + 1 ml 40% Na. OH + 1 -3 drops of lead acetate solution • heat the mixture in WB for 3 min • Cool • observe any change • Black ppt.

Exp. 6 Sakaguchi Reaction • This reaction is used to detect the presence of guanidine group. • The only AA that contains guanidine group is arginine which reacts with α-naphthol and an oxidizing agent such as bromide water to give a red color. Arginine

Exp. 6 Sakaguchi Reaction • 2 ml AA + 1 ml 2 M Na. OH + 1 ml ethanolic 0. 02% α-naphthol • mix well • cool in ice • add 1 ml of alkaline hypochlorite solution • Red color
- Slides: 15