Qualitative Inorganic Analysis By Tedi Hadiana SMK Negeri

Qualitative Inorganic Analysis By : Tedi Hadiana SMK Negeri 13 Bandung
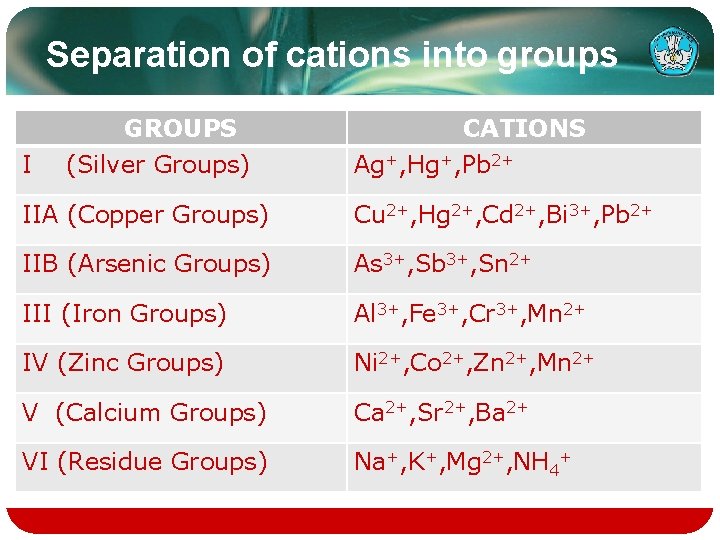
Separation of cations into groups GROUPS I (Silver Groups) CATIONS Ag+, Hg+, Pb 2+ IIA (Copper Groups) Cu 2+, Hg 2+, Cd 2+, Bi 3+, Pb 2+ IIB (Arsenic Groups) As 3+, Sb 3+, Sn 2+ III (Iron Groups) Al 3+, Fe 3+, Cr 3+, Mn 2+ IV (Zinc Groups) Ni 2+, Co 2+, Zn 2+, Mn 2+ V (Calcium Groups) Ca 2+, Sr 2+, Ba 2+ VI (Residue Groups) Na+, K+, Mg 2+, NH 4+
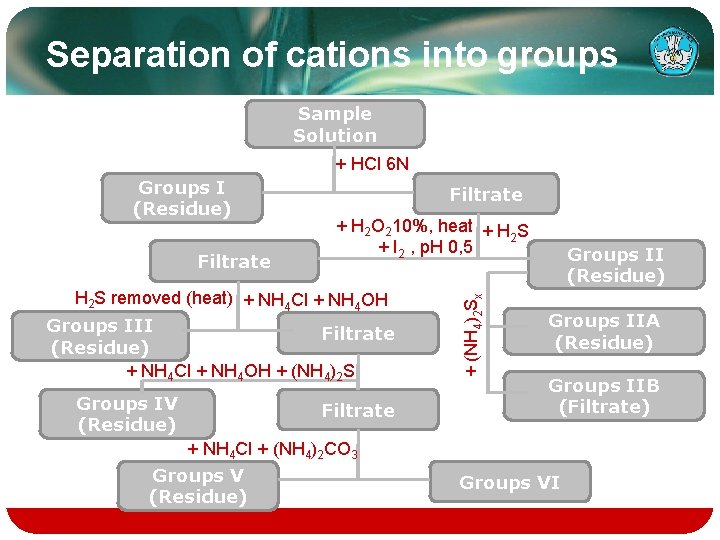
Separation of cations into groups Sample Solution + HCl 6 N Filtrate + H 2 O 210%, heat + H S 2 + I 2 , p. H 0, 5 H 2 S removed (heat) + NH 4 Cl + NH 4 OH Groups III Filtrate (Residue) + NH 4 Cl + NH 4 OH + (NH 4)2 S Groups IV (Residue) Filtrate + (NH 4)2 Sx Groups I (Residue) Groups IIA (Residue) Groups IIB (Filtrate) + NH 4 Cl + (NH 4)2 CO 3 Groups V (Residue) Groups VI

Separation and Identification of Groups 1 Residue Groups I may contain: Ag. Cl(white), Hg 2 Cl 2(white), Pb. Cl 2(white) Filtrate may contain : Pb. Cl 2 Identification: 1. Cool a portion of the solution white crystalline residu of Pb. Cl 2 2. Add 1 M K 2 Cr. O 4 solution yellow residue of Pb. Cr. O 4, soluble in Na. OH solution 3. Add dilute 2 M H 2 SO 4 (+alcohol) white residue of Pb. SO 4 soluble in NH 4 OAc solution Residue : Hg 2 Cl 2 If black, consists of Hg(NH 2)Cl + Hg. Hg 22+ present Wash the residu with 2 m. L of 2 M HCl, then 2 -3 times with 1 m. L portions of cold water then add 2 m. L hot water and heating for 2 -3 minutes Residue may contain : Ag. Cl, Hg 2 Cl 2 Pour 1 m. L dilute 2 M NH 4 OH solution Filtrate may contain : [Ag(NH 3)2]+ Identification: 1. 2. Acidity with dilute HNO 3 white residue of Ag. Cl Add a few drops of KI solution pale yellow residue of Ag. I

Separation and Identification of Groups 1 Residue : Hg 2 Cl 2 If black, consists of Hg(NH 2)Cl + Hg. Hg 22+ present Add 1 m. L aqua regia (HNO 3 : HCl = 1: 3) and heat cautiously until the mixture is dry Then add 1 m. L 2 M HNO 3 (centrifuge) Filtrate may contain : Hg(NO 3)2 Identification: 1. 2. Place 1 drop filtrate upon Sn. Cl 2 paper greyish-black spot of Hg Place drop filtrate on a glossy copper surface silverish spot of Hg

Separation and Identification of Groups 2 A Residue Groups II A may contain : Hg. S(black), Cu. S(black), Cd. S(yellow), Bi 2 S 3{black-brown), Pb. S (black) Residue may contain: Hg. S 1. Wash the residue with aquadest (2 -3 times) 2. Add 1 m. L aqua regia and heat cautiously until the mixture in dry 3. Add 1 m. L water, stir Identification, see Hg+ of groups 1 Wash the residue with water to be free of chloride (2 -3 times) add 2 m. L dilute HNO 3 1: 1, boil gently for 2 -3 minutes (centrifuge) Filtrate may contain : Cu, Cd, Bi, Pb Add 5 drops of H 2 SO 4 concentrated and heat cautiously until the mixture is dry Add 1 m. L water (centrifuge) Filtrate may contain : Cu , Cd , Bi Residue may contain : Pb. SO 4 Wash the residue with aquadest (2 -3 times) Dissolved residue with 10 drops 2 M NH 4 OAc Filtrate my contain : Pb 2+ Identification: Add a few drop of 1 M K 2 Cr. O 4 solution yellow residue of Pb. Cr. O 4

Separation and Identification of Groups 2 A Filtrate may contain : Cu , Cd , Bi Add 6 M NH 4 OH solution (centrifuge) Residue may contain : Bi(OH)3 Wash the residue with water dissolve in the minimum volume of 2 M HCl solution Identification: 1. Pour in to Sn. Cl 2 solution and more of 2 M Na. OH black brown residue of Bi 2. Dissolve a little residue in drop of dilute 2 M Na. OAc then place upon filter paper moistened with cinchonine-KI reagent orange-red spot Filtrate may contain : [Cu(NH 3)4]2+ and [Cd(NH 3)4]2+ Identification: 1. If deep blue in colour Cu 2+ present 2. Confirm Cu 2+ by acidifying a portion of the filtrate with dilute 2 M HCl and add K 4[Fe(CN)6] reddish-brown residue of Cu 2 Fe(CN)6 3. Drop of the filtrate upon filter paper and give a NH 3 vapor blue spot 4. Pour the filtrate on H 2 S liquid (If Cu present, add 2 M KCN solution until blue color is discharged) yellow residue of Cd. S

Separation and Identification of Groups 2 B Filtrate Groups II B may contain : (NH 4)3 As. S 4, (NH 4)2 Sb. S 4, (NH 4)2 Sn. S 3 Add concentrated 6 M HCl dropwise Warm gently for 3 minutes (centrifuge) Residue Group II B may contain : As 2 S 5 (yellow), Sb 2 S 5 (orange), Sn. S 2 (yellow) Filtrate may contain : Sb. Cl 3 and Sn. Cl 4 Wash the residue with aquadest (2 -3 times) Add 6 M HCl and warm gently for 3 minutes (centrifuge) Residue may contain : As 2 S 5 Wash the residue with water (1 -2 times) Dissolve residue with 1 m. L of 6 M Na. OH solution Filtrate Identification: 1. Add 6 M Na. OH solution and add 2 piece metallic Aluminium then gases identification with Hg. Cl 2 grey spot from Hg 2. Add H 2 O 2 3% solution and warm for a few minutes oxidize arsenit to arsenat a. 2 drops of the filtrate + 2 drops 2 M HCl solution and add Mgmixture white residue of Mg. NH 4 As. O 4 b. 2 drops of the filtrate with HNO 3 concentrated and add 10 drops of molibdat reagent yellow residue of NH 4 -arsenat molibdat

Separation and Identification of Groups 2 B Filtrate may contain : Sb. Cl 2 and Sn. Cl 4 Add metallic iron in to the filtrate Warm gently for 3 minutes Add 10 m. L of water (centrifuge) Filtrate may contain : Sb 3+ Identification: 1. To 2 drops of the filtrate add KNO 2 (solid) mixing until gas become steam and then 2 drops of Rhodamine. B reagent (red color) violet solution Filtrate : Sn 2+ Identification: 1. Dissolve the filtrate with Hg. Cl 2 5 % solution white residue of Hg 2 Cl 2 or grey residue of Hg (Sn present) 2. Place 1 drop the filtrate upon cacothilin paper red or purple spot (Sn present)

Separation and Identification of Groups 3 Residue Groups III may contain : Fe(OH)3 (brown), Al(OH)3 (white), Cr(OH)3 (green), Mn. O 2. x. H 2 O (brown) Wash the residue with a little NH 4 NO 3 solution (2 -3 times) Add 1, 5 m. L of 2 M Na. OH and 0. 5 m. L 6% H 2 O 2 Boil gently until the evolution of O 2 ceases (2 -3 minutes)-(centrifuge) Residue may contain : Fe(OH)3, Mn. O 2. x. H 2 O Filtrate may contain : Na. Al. O 2, Na 2 Cr. O 4 Wash the residue with 1 m. L of NH 4 NO 3 solution. Dissolve residue in to 5 drops of HCl concentrated then dilute solution until 1 m. L Filtrate may contain : Fe 3+ Identification: 1. To the filtrate add a few drops of 2 M KSCN solution deep red colouration (Fe present) 2. To the filtrate add K 4[Fe(CN)4] solution blue residue (Fe present) Filtrate may contain : Mn 2+ Identification: 1. To the filtrate add 5 drops of 6 M HNO 3 solution and a few drops of Na. Bi. O 3/KIO 4/Pb. O 2 warm for a few minutes violet solution (Mn present) 2. Heat 2 drops of the filtrate until the mixture is dry add Na 2 CO 3 solid and KCl. O 3 solid then melted green solution of K 2 Mn. O 4 (Mn present)

Separation and Identification of Groups 3 Filtrate may contain : Na. Al. O 2, Na 2 Cr. O 4 Acidify with HOAc until just neutral (test with litmus paper) (centrifuge) Residue may contain : Al(OH)3 Filtrate may contain : Na 2 Cr. O 4 Wash the residue with a few water Dissolve with a few 2 M HCl solution Filtrate may contain : Cr 3+ Filtrate may contain : Al 3+ Identification: 1. To the filtrate add 2 drops of 6 M NH 4 OH solution white residue of Al(OH)3 2. To 5 drops of the filtrate add 2 drops of 6 M NH 4 OAc solution and 3 drops of the aluminon reagent then heat for 5 minutes. Add (NH 4)2 CO 3 until just alkaline and add 3 drops later red residue Identification: 1. Acidity with acetic acid and add Pb(OAc)2 solution yellow residue of Pb. Cr. O 4 2. To 5 drops of the filtrate add 2 drops of Ag. NO 3 solution red residue of Ag 2 Cr. O 4

Separation and Identification of Groups 4 Residue Groups IV may contain : Co. S (black), Ni. S (black), Mn. S (brown), Zn. S (putih) Wash the residue with a little NH 4 OH solution (2 -3 times) Add 1 m. L water and 6 drop 2 M HCl solution If black Residue may contain : Co. S, Ni. S Filtrate may contain : Zn. Cl 2, Mn. Cl 2 Wash the residue with 1 m. L of acetic acid solution. Dissolve the residue in 1 m. L of aqua regia and heat cautiously until the mixture is dry. Then add 1 m. L 2 M HCl solution Filtrate may contain : Ni 2+ Identification: 1. Add 5 drop NH 4 Cl solution + NH 4 OH solution (until alkaline) + DMG reagent red residue Filtrate may contain : Co 2+ Identification: 1. Add 0. 5 m. L amyl alcohol, 0. 5 g solid KSCN and shake well amyl alcohol layer coloured blue 2. Add 5 drop 2 M HCl + -nitroso naphtol + chloroform Chloroform layer coloured

Separation and Identification of Groups 4 Filtrate may contain : Zn. Cl 2, Mn. Cl 2 Heat until H 2 S removed, Add 10 drops 6 M Na. OH + H 2 O 2. Boil gently until the evolution of O 2 ceases (2 -3 minutes) Filtrate may contain : Na 2 Zn. O 2 Identification: 1. Acidity with acetic acid and pass H 2 S white residue of Zn. S 2. Acidity with H 2 SO 4, add 0, 5 m. L of 0, 1% Cu. SO 4 solution and 1 m. L (NH 4)2[Hg(CNS)4] reagent (stir) violet residue 3. Acidity with H 2 SO 4, add K 4[Fe(CN)6] white residue Residue may contain : Mn. O 2 Wash the residue with 1 m. L of water Dissolve residue in to 5 drops of HCl concentrated then dilute solution until 1 m. L Filtrate may contain : Mn 2+ Identification: 1. To the filtrate add 5 drops of 6 M HNO 3 solution and a few drops of Na. Bi. O 3/KIO 4/Pb. O 2 warm for a few minutes violet solution

Separation and Identification of Groups 5 Residue Groups V may contain : Ca. CO 3 (white), Ba. CO 3 (white), Sr. CO 3 (white) Wash the residue with aquadest (2 -3 times) Dissolved residue with few hot 2 M acetic acid solution Then add 3 drop NH 4 OAc + K 2 Cr. O 4 Residue may contain : Ba. Cr. O 4 Identification : Dissolve the residue in a little concentrated HCl (flame test) Green (yellowish-green) flame Wash the residue with hot water Dissolve residue in 1 m. L of hot 2 M HCl solution Filtrate may contain : Ba 2+ Identification: 1. add a few drops of 2 M H 2 SO 4 solution white residue of Ba. SO 4 Filtrate may contain : Ca 2+, Sr 2+ Filtrate add 2 m. L saturated (NH 4)SO 4 solution, followed by 0. 2 g Na 2 S 2 O 4 heat in a beaker of boiling water for 5 minute Residue may contain : Sr. SO 4 Filtrate may contain : Ca 2+

Separation and Identification of Groups 5 Residue may contain : Sr. SO 4 Identification : Wash with a little water, then dry the residue. With a few drops concentrated HCl apply the flame test crimson flame (Sr present) Filtrate may contain : Ca 2+ Identification : Add a little (NH 4)2 C 2 O 4 solution , 2 m. L 2 M acetic acid and warm on a water bath White residue of Ca. C 2 O 4

Separation and Identification of Groups 6 Filtrate Groups V I may contain : Mg 2+, Na+, K+ Put filtrate in evaporation crucible It is evaporated to dryness and heated until all the ammonium salts have been volatilized The dry residue is extracted with water Filtrate may contain : Na+ Identification : Add a little Uranyl zink acetate reagent, shake and allow to stand for a few minutes yellow crystalline residu Filtrate may contain : K 2+ Identification : Add a little Sodium hexanitritocobaltate(III) solution and drops dilute acetic acid. Stir and allow to stand for 1 -2 minutes yellow residue of K 2[Co(NO 2)6] Filtrate may contain : Mg 2+ Identification : 1. add 2 drops of magneson reagent, + Na. OH solution until alkaline blue residue 2. Add 2 drops 2 M NH 4 Cl + NH 4 OH +Na 2 HPO 4 solution white residue of Mg. NH 4 PO 4
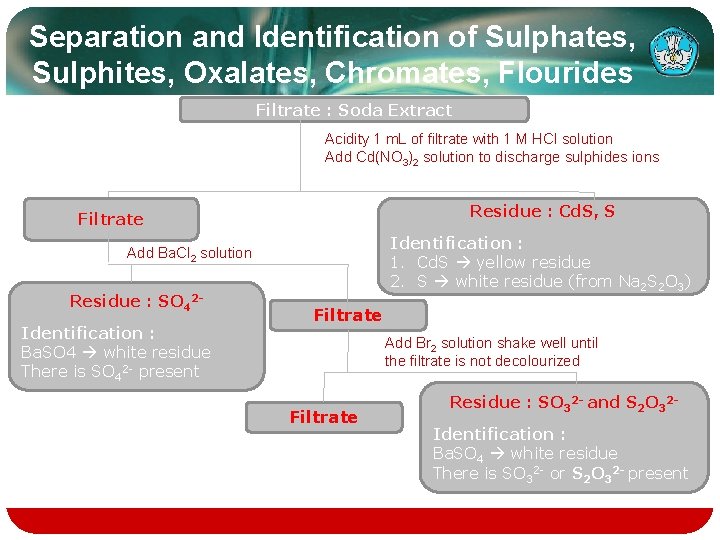
Separation and Identification of Sulphates, Sulphites, Oxalates, Chromates, Flourides Filtrate : Soda Extract Acidity 1 m. L of filtrate with 1 M HCl solution Add Cd(NO 3)2 solution to discharge sulphides ions Residue : Cd. S, S Filtrate Identification : 1. Cd. S yellow residue 2. S white residue (from Na 2 S 2 O 3) Add Ba. Cl 2 solution Residue : SO 42 Identification : Ba. SO 4 white residue There is SO 42 - present Filtrate Add Br 2 solution shake well until the filtrate is not decolourized Filtrate Residue : SO 32 - and S 2 O 32 Identification : Ba. SO 4 white residue There is SO 32 - or S 2 O 32 - present

Separation and Identification of Sulphades, Sulphites, Oxalates, Chromates, Flourides Filtrate Add Na. OAc solution (centrifuge) Residue : H 2 Cr. O 4/H 2 Cr 2 O 7, HF and H 2 C 2 O 4 Identification : 1. Ba. Cr. O 4 yellow residue (there is H 2 Cr. O 4 or H 2 Cr 2 O 7 present) 2. Ba. F 2 white (if there have a lot of HF and H 2 C 2 O 4 ) Filtrate : Identification : 1. Add Ca. Cl 2 white residue of Ca. C 2 O 4 and white residue of Ca. F 2 (centrifuge) 2. Place the residue in 2 M HOAc solution and add 1 drops of 0, 2 M KMn. O 4 shake and warming (If KMn. O 4 is bleached, add KMn. O 4 solution in excess until the colour is constant) 3. If all of the residue is dissolved just H 2 C 2 O 4 present 4. If there is still the residue H 2 C 2 O 4 and HF present 5. If adding 1 drops of 0, 2 M KMn. O 4 is not decolourized Ca. F 2 residue and just HF present

Separation and Identification of Sulphades, Sulphites, Oxalates, Chromates, Flourides Residue may contain : Ag. Cl (white), Ag. SCN (white), Ag. Br (white-yellow), Ag. I (yellow) If there is SCN-, Ag. SCN must be removed first as follows : Add 1 m. L of HNO 3 concentrate and steam solution until almost dry (repeat once more until oxidation Ag. SCN has finished) Wash the residue has been free Ag. SCN with 1 m. L of H 2 O and 5 drops of 6 M HNO 3 until free of Ag+ Add 10 drops of 1 M (NH 4)2 CO 3 (centrifuge) Filtrate : Ag(NH 3)2+, Cl. Identification : 1. At a part of filtrate add 1 drops 1 M KBr yellowish white residu of Ag. Br , Clpresent 2. Acidity with 6 M HNO 3 white residue of Ag. Cl, Clpresent Residue may contain : Ag. Br, Agl

Separation and Identification of Sulphades, Sulphites, Oxalates, Chromates, Flourides Residue may contain : Ag. Br, Agl Add a few of Zn solid and 1 m. L 2 M H 2 SO 4 (by reduce coming about HBr and HI) centrifuge Identification filtrate about Iodide : 1. To the filtrate add 1 drop of 0, 1 M Fe. Cl 3 (1: 1) and place 1 drop of solution upon starch paper blue spot of I 2. If Iodide is present must be remove first as follows : A part of the filtrate adding 0, 1 Fe. Cl 3 (1: 1) and boil gently until all iodide are volatilized (test with starch paper) 3. For the filtrate of free from Iodide add 3 drops of 0, 1 M KMn. O 4 and 3 drops of 3 M H 2 SO 4 then shake well. KMn. O 4 excess must be remove with 10% H 2 O 2 Add CHCl 3 layer coloured brown Bromide present

Preliminary Test For Solid Samples Generally test Appearance : Observe whether it is Ø crystalline or powder Ø magnetic Ø possesses any characteristic odour or colour Flame test Test with H 2 SO 4 (dilute and concentrated) Test with Na. OH Test with KHSO 4 Solubility test
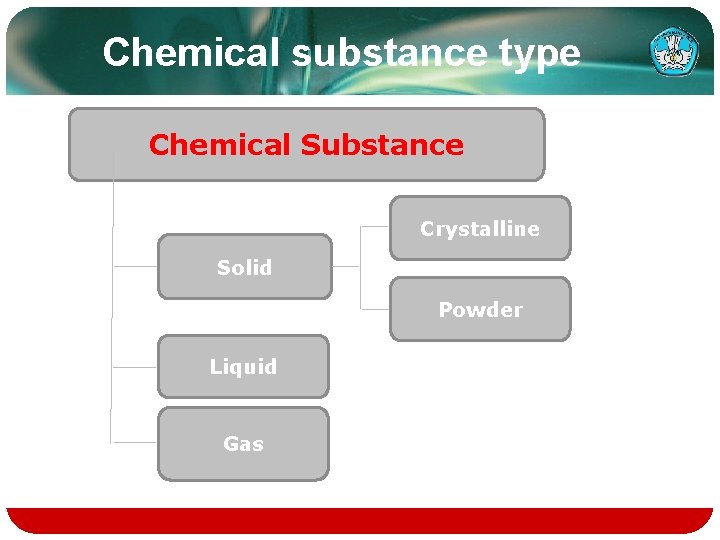
Chemical substance type Chemical Substance Crystalline Solid Powder Liquid Gas
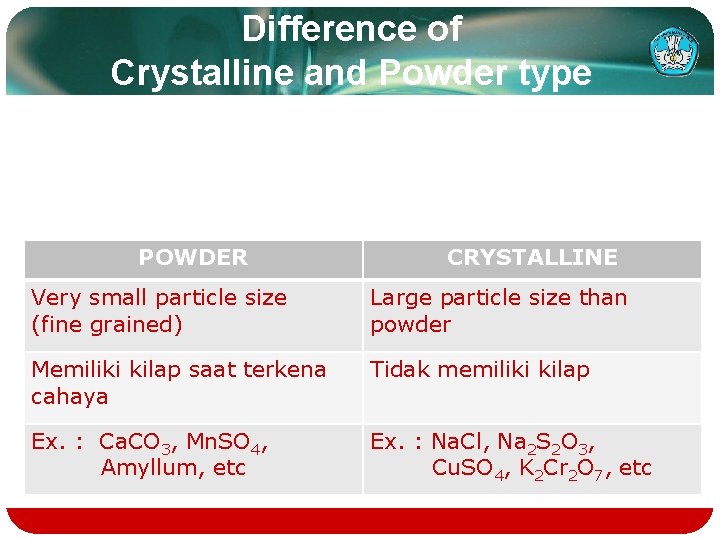
Difference of Crystalline and Powder type POWDER CRYSTALLINE Very small particle size (fine grained) Large particle size than powder Memiliki kilap saat terkena cahaya Tidak memiliki kilap Ex. : Ca. CO 3, Mn. SO 4, Amyllum, etc Ex. : Na. Cl, Na 2 S 2 O 3, Cu. SO 4, K 2 Cr 2 O 7, etc
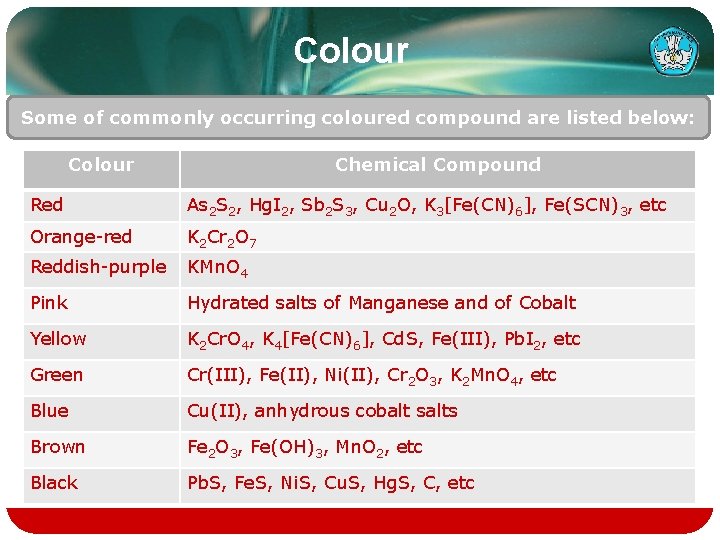
Colour Some of commonly occurring coloured compound are listed below: Colour Chemical Compound Red As 2 S 2, Hg. I 2, Sb 2 S 3, Cu 2 O, K 3[Fe(CN)6], Fe(SCN)3, etc Orange-red K 2 Cr 2 O 7 Reddish-purple KMn. O 4 Pink Hydrated salts of Manganese and of Cobalt Yellow K 2 Cr. O 4, K 4[Fe(CN)6], Cd. S, Fe(III), Pb. I 2, etc Green Cr(III), Fe(II), Ni(II), Cr 2 O 3, K 2 Mn. O 4, etc Blue Cu(II), anhydrous cobalt salts Brown Fe 2 O 3, Fe(OH)3, Mn. O 2, etc Black Pb. S, Fe. S, Ni. S, Cu. S, Hg. S, C, etc

Hygroscopis Properties 1. Some chemical substance can easily catch water from air Chemical substance wet looks 2. Generally, chloride salt have hygroscopis properties. Example : Ca. Cl 2, Mg. Cl 2, Co. Cl 2 , Fe. Cl 3, Mn. Cl 2, etc 3. Other example : Na. OH, KOH, etc
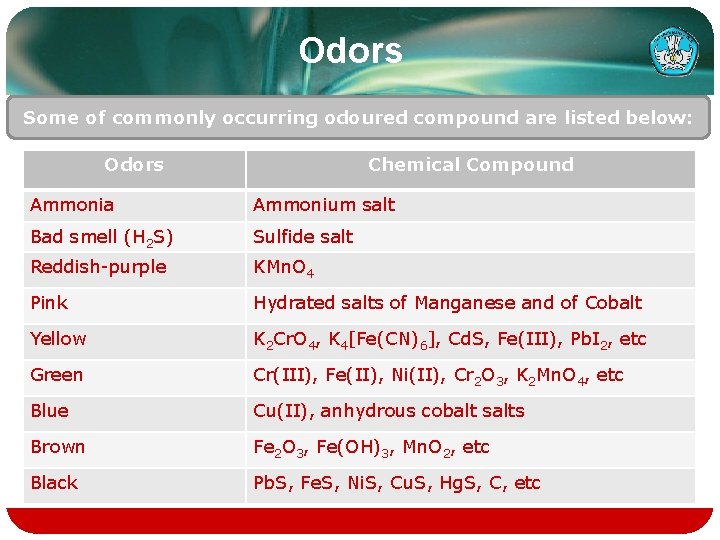
Odors Some of commonly occurring odoured compound are listed below: Odors Chemical Compound Ammonia Ammonium salt Bad smell (H 2 S) Sulfide salt Reddish-purple KMn. O 4 Pink Hydrated salts of Manganese and of Cobalt Yellow K 2 Cr. O 4, K 4[Fe(CN)6], Cd. S, Fe(III), Pb. I 2, etc Green Cr(III), Fe(II), Ni(II), Cr 2 O 3, K 2 Mn. O 4, etc Blue Cu(II), anhydrous cobalt salts Brown Fe 2 O 3, Fe(OH)3, Mn. O 2, etc Black Pb. S, Fe. S, Ni. S, Cu. S, Hg. S, C, etc
- Slides: 26