QUALITATIVE AND QUANTITATIVE ELEMENTAL ANAYSIS by SHITTU T

QUALITATIVE AND QUANTITATIVE ELEMENTAL ANAYSIS by SHITTU T. O. is licensed under a Creative Commons Attribution-Non. Commercial-Share. Alike 4. 0 International License. • QUALITATIVE AND QUANTITATIVE ELEMENTAL ANAYSIS

Learning goals • At the end of the lecture, you should be able to: • a). Describe qualitative tests for specific elements in organic sample • b). Carry out basic calculations in quantitative elemental analysis.

Qualitative elemental analysis • This method deals with identification of various elements present in an organic sample. • Most organic samples contain C, H , O and other elements such as N, P, S, halogens and metals.

Detection of C and H • C & H can be detected in an organic sample by heating the sample with cupric oxide (CUO) in a combustion tube. • Sample+ CUO CO 2(g) + H 2 O(g) CO 2 turns lime water milky. H 2 O turns anhydrous CUSO 4 from white to blue or anhydrous Co. Cl 2 fom blue to pink.

Detection of N, S and halogens • N, S and halogens can be detected in an organic sample by using Lassaigne’s sodium fusion test. This method converts elements in the organic sample into soluble sodium compound by fusion with sodium metal. • [C, H, N, X, P, S] Na (excess) Na. CN + Na 2 S + Na. X If limited sodium is used, sodiumthiosulphate is formed instead of sodiumcyanide [C, H, N, X, P, S] Na (limited) Na. SCN + Na. X • Na • Na. CN + Na 2 S

Sodium fusion test cont’d • Further work-ups and analyses will then lead to identification of S, Br, I, Cl or N.

Quantitative elemental analysis • This deals with estimation of the proportion by mass or % of the element in a given mass of organic sample.
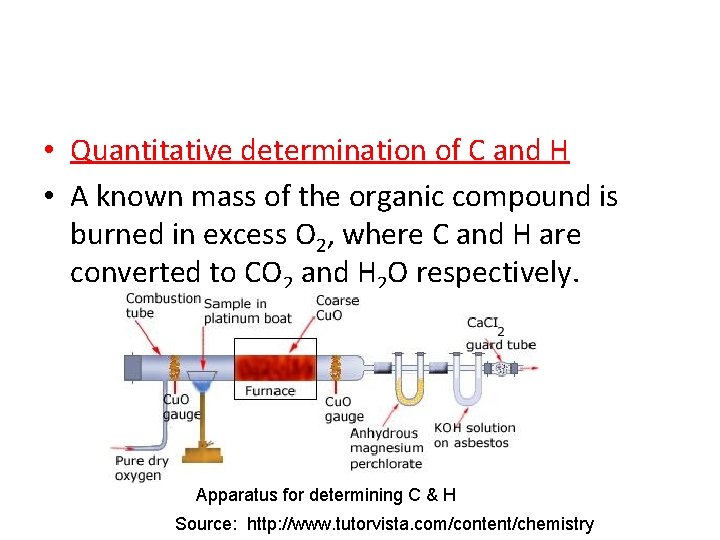
• Quantitative determination of C and H • A known mass of the organic compound is burned in excess O 2, where C and H are converted to CO 2 and H 2 O respectively. Apparatus for determining C & H Source: http: //www. tutorvista. com/content/chemistry

• The CO 2 is absorbed in soda lime (KOH) and weighed. • H 2 O is absorbed in Ca. Cl 2 and also weighed. • The % C and H can then be determined.

Question 1 • 1. 62 g of an unknown organic compound containing only carbon, hydrogen and oxygen, gave 2. 64 g of CO 2 and 0. 9 g of H 2 O on complete combustion. • i. Determine the empirical formula of the compound. • ii. What is the molecular formula of the compound given if its relative molecular mass is 180

Quantitative determination of nitrogen • i. Kjeldahl method • ii. Dumas method • Kjeldahl method • This method is used for quantitative determination of nitrogen in samples such as protein, amines and other nitrogen containing compounds. • The sample is digested by heating it with sulphuric acid in the presence of a catalyst such as Cu or Se. The resulting mixture is made alkaline by addition of Na. OH. • The volume of NH 3(g) liberated is then determined by volumetric analysis by titrating NH 3 formed with a standard H 2 SO 4 or HCl, using a suitable indicator.

Equation of reaction for titration: • NH 3(g) + H 2 SO 4 (aq) (NH 4)2 SO 4(aq)


Question • 1. 4 g sample of a newly isolated organic compound was subjected to kjeldahl analysis. The gas produced was neutralized by 26. 3 cm 3 of 0. 25 M sulphuric acid. • (a). name the gas produced in this analysis • (b). Calculate the percentage of nitrogen in the organic sample. (N= 14. 0, H= 1. 0)

Question 3: • 0. 14 g of an organic sample on suitable treatment gave 27. 44 cm 3 of nitrogen measured at STP. What is the percentage of nitrogen in the sample

Quantitative determination of sulphur and halogens • Sulphur and halogens can be quantitatively determined by gravimetric analysis using Carius method. • Under suitable treatment, the S in the sample is converted to Ba. SO 4 and • halogens to Ag. X (X = Cl, Br, I e. t. c. )

A 0. 8 g sample gave 1. 2 g of Ba. SO 4 in a gravimetric analysis. What is the % of S in the sample.

Practice question • The molecular mass of an organic compound containing C, H and N was found to be 59. on combustion, 0. 13 g of the compound gave 0. 29 g of CO 2 and 0. 18 g of H 2 O. 0. 145 g of the compound on suitable treatment gave 27 cm 3 of N 2 measured at S. T. P. Deduce the structure possible structures of the compound .

• QUALITATIVE AND QUANTITATIVE ELEMENTAL ANAYSIS

Learning goals • At the end of the lecture, you should be able to: • a). Describe qualitative tests for specific elements in organic sample • b). Carry out basic calculations in quantitative elemental analysis.

Qualitative elemental analysis • This method deals with identification of various elements present in an organic sample. • Most organic samples contain C, H , O and other elements such as N, P, S, halogens and metals.

Detection of C and H • C & H can be detected in an organic sample by heating the sample with cupric oxide (CUO) in a combustion tube. • Sample+ CUO CO 2(g) + H 2 O(g) CO 2 turns lime water milky. H 2 O turns anhydrous CUSO 4 from white to blue or anhydrous Co. Cl 2 fom blue to pink.

Detection of N, S and halogens • N, S and halogens can be detected in an organic sample by using Lassaigne’s sodium fusion test. This method converts elements in the oragnic sample into soluble sodium compound by fusion with sodium metal. • [C, H, N, X, P, S] Na (excess) Na. CN + Na 2 S + Na. X If limited sodium is used, sodiumthiosulphate is formed instead of sodiumcyanide [C, H, N, X, P, S] Na (limited) Na. SCN + Na. X • Na • Na. CN + Na 2 S

Procedure in Lassaigne’s sodium fusion test • The sample is heated with excess sodium metal to redness. The heated sample is and plugged into water and then ground in a mortar. The content is allowed to cool and then filtered. • Test for nitrogen: • Lassaignes fitrate + Fe. SO 4(aq) Na 4 Fe(CN)6(aq) + Na 2 SO 4(aq) • 3 Na 4 Fe(CN)6(aq) + 4 Fe. Cl 3 (aq) Fe 4[Fe(CN)6]3(aq)+ 12 Na. Cl prussian blue solution
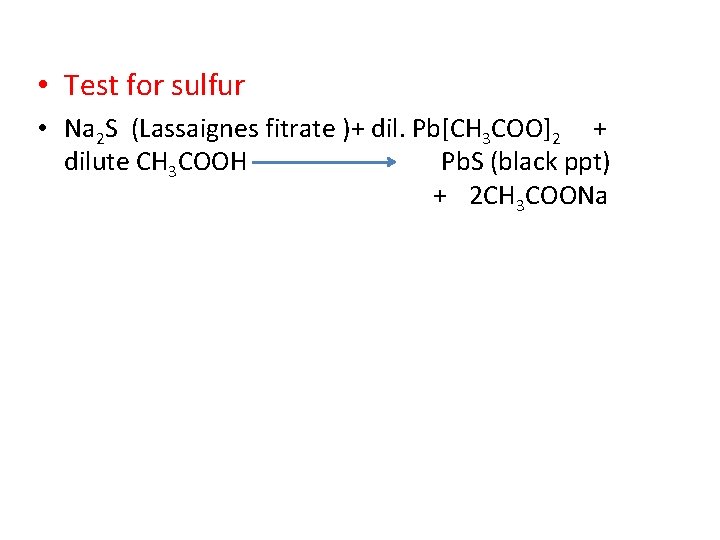
• Test for sulfur • Na 2 S (Lassaignes fitrate )+ dil. Pb[CH 3 COO]2 + dilute CH 3 COOH Pb. S (black ppt) + 2 CH 3 COONa

• Test for halogens To test for halogens, Lassaignes fitrate is first acidified with few drops of dilute H 2 SO 4 and boiled to remove S And CN in the form of HCN(g) and H 2 S(g). These S 2 - and CN- ions can also interfere with the precipitating agents such as Ag. NO 3 • Na. X + Ag. NO 3 dil. HNO 3 Na. NO 3 + Ag. X • White ppt soluble in excess aqueous NH 3, it indicates the presence of Cl- present • Pale yellow ppt sparingly soluble in excess aqueous NH 3, it indicates the presence of Br- present • yellow ppt insoluble in excess aqueous NH 3, it indicates the presence of I- present

Quantitative elemental analysis • This deals with estimation of the proportion by mass or % of the element in a given mass of organic sample. • Quantitative determination of C and H • A known mass of the organic compound is burnt in excess O 2, where C and H are converted to CO 2 and H 2 O respectively. • The CO 2 is absorbed in soda lime (KOH) and weighed. • H 2 O is absorbed in Ca. Cl 2 and also weighed. • The % C and H can then be determined.

Question 1 • 1. 62 g of an unknown organic compound containing only Carbon, hydrogen and oxygen, gave 2. 64 g of CO 2 and 0. 9 g of H 2 O on complete combustion. • i. Determine the empirical formula of the compound. • ii. What is the molecular formula of the compound given if its relative molecular mass is 180

Quantitative determination of nitrogen • i. Kjeldahl method • ii. Dumas method • • • Kjedahl method this method is used for quantitative determination of nitrgen in samples such as protein, amines and other nitrogen containing compounds. The sample is digested by heating it with sulphuric acid in the presence of a catalyst such as Cu or Se. the digestion converts the nitrogen in the sample to ammonia. The ammonia gas formed is not liberated in acid solution due to the presence of SO 42 -. The solution is made alkaline by reacting Na. OH, which converts it to NH 3. The volume of NH 3(g) liberted is then determined by volumetric analysis by titrating NH 3 formed with a standard H 2 SO 4 or HCl, using a suitable indicator. Equation of titration for titration: NH 3(g) + H 2 SO 4 (aq) (NH 4)2 SO 4(aq)


Question 2 • 1. 4 g sample of a newly isolated organic compound was subjected to kjedahl analysis. The gas produced was neutralized by 26. 3 cm 3 of 0. 25 M sulphuric acid. • (a). name the gas produced in this analysis • (b). Calculate the percentage of nitrogen in the organic sample. (N= 14. 0, H= 1. 0)

Question 3: • 0. 14 g of an organic sample on suitable treatment gave 27. 44 cm 3 of nitrogen measured at STP. What is the percentage of nitrogen by mass of nitrogen.

Quantitative determination of sulphur and halogens • Sulphur and halogen can be quantitatively determined by gravimetric analysis using Carius method. The precipitate formed is washed, filtered, dried and weighed. Under suitable treatment, the S in an sample is converted to Ba. SO 4 and halogens to Ag. X

QUALITATIVE AND QUANTITATIVE ELEMENTAL ANAYSIS by SHITTU T. O. is licensed under a Creative Commons Attribution-Non. Commercial-Share. Alike 4. 0 International License. Practice questions • The molecular mass of an organic compound containing C, H and N was found to be 59. on combustion, 0. 13 g of the compound gave 0. 29 g of CO 2 and 0. 18 g of H 2 O. 0. 145 g of the compound on suitable treatment gave 27 cm 3 of N 2 measured at S. T. P. Deduce the structure possible structures of the compound .
- Slides: 34