Qualitative analysis the identification of the specific substances

Qualitative analysis – the identification of the specific substances present Quantitative analysis – the measurement of the quantity of a substance present

Qualitative Analysis • Many substances can be identified by their unique physical or chemical properties: 1. Observation of colour of solution 2. For cations that are usually colourless in solution and highly soluble, a flame test may be used (only works when a single cation is present) 3. For gases, perform a gas test to identify if the gas produced is H 2, O 2, or CO 2 4. Other possible qualitative tests 5. Reactions with other substances to produce a new colour and/or precipitate
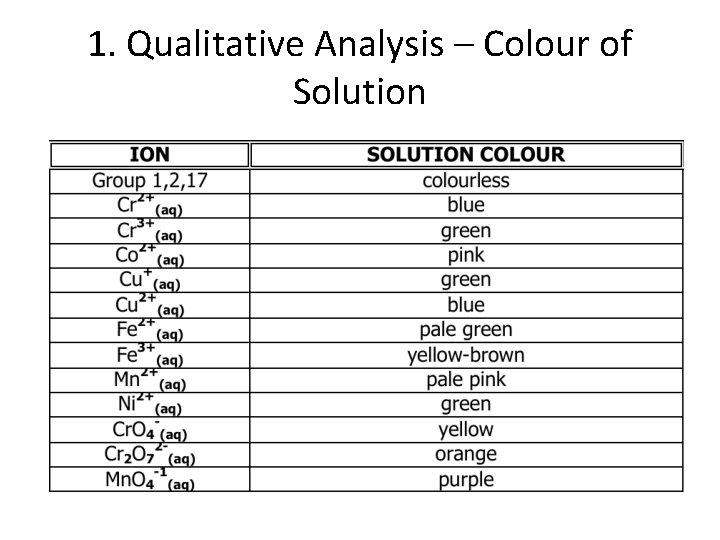
1. Qualitative Analysis – Colour of Solution

2. Qualitative Analysis – Flame Test • The majority of solids are white powders and difficult to distinguish between just by looking. • Flame tests can give a good indication of what metal ion it contains.


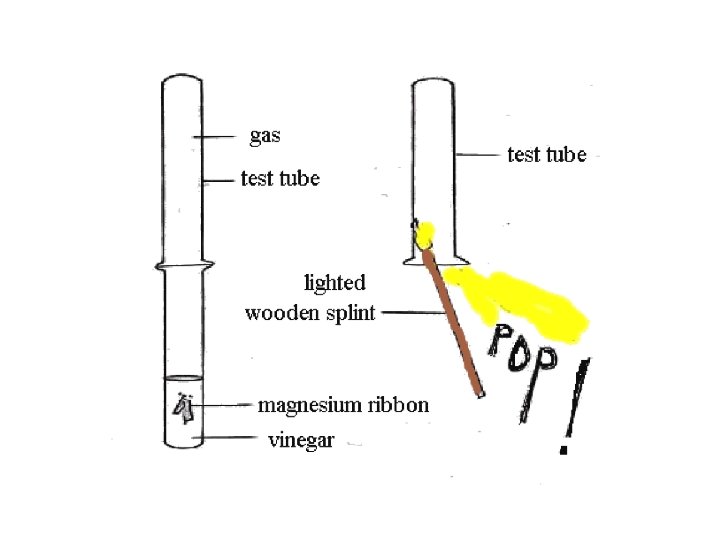
3. Qualitative Analysis – Gas Test • Most gases are clear, colourless and odourless. • So it's not easy to distinguish between them with a simple chemical test.

Test for Hydrogen • Hydrogen is less dense than air and explosive when mixed with Oxygen. • A test-tube filled with hydrogen can be safely held up-turned since no Hydrogen can escape by sinking into denser air.

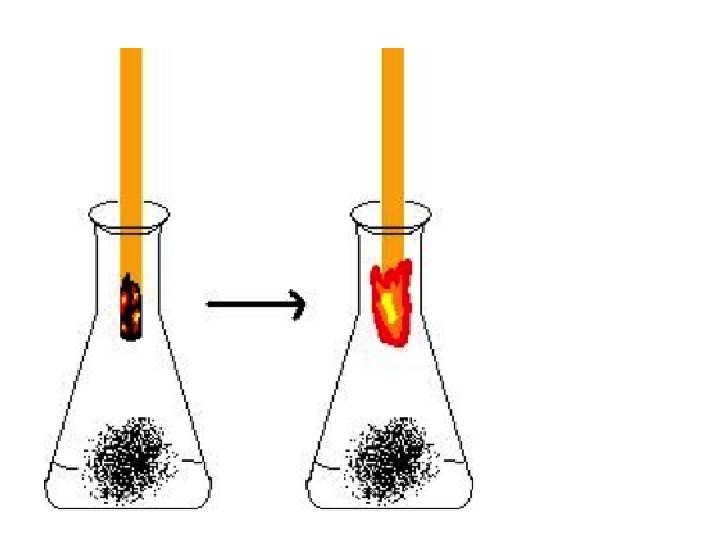
Oxygen Test • Oxygen has a similar density to air, so needs to be contained in a "right-side up" container • So, if we light a splint and blow out the flame so that the end is still glowing (the reason the flame does not reappear is that 79% of the air around it is not Oxygen) • Placing the glowing splint in pure Oxygen means that every gas molecule that collides with the glowing ember is Oxygen. • This increases the rate of reaction, providing enough heat to re-light the flame.

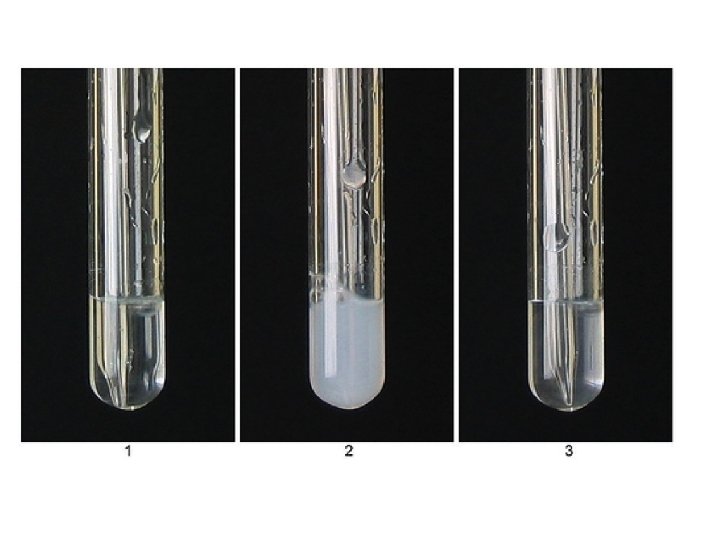
Carbon Dioxide Test • Carbon Dioxide puts out fires. But so do most other gases. • So the test for Carbon Dioxide must be something that only Carbon Dioxide does. • Only Carbon Dioxide turns Lime-water milky white • The white colour is a precipitate of Calcium Carbonate ( like chalk).

4. Other possible tests • Conductivity – to determine if solution is ionic or covalently bonded • p. H – to determine id the solution is acidic or basic – Universal indicator (neutral is green, red is acid, blue is base) – Bromothymol blue (neutral is green, yellow is acid, blue is base) – Phenolphthalein (pink is base)

• Melting point/boiling point • Solubility in water – to determine polar or non polar

5. Qualitative Analysis – Reactions in Solution • This is a type of test where we set up a double displacement reaction using one unknown solution and one known solution • In this test, we would predict that if a precipitate forms then a certain ion must have been present in the unknown solution
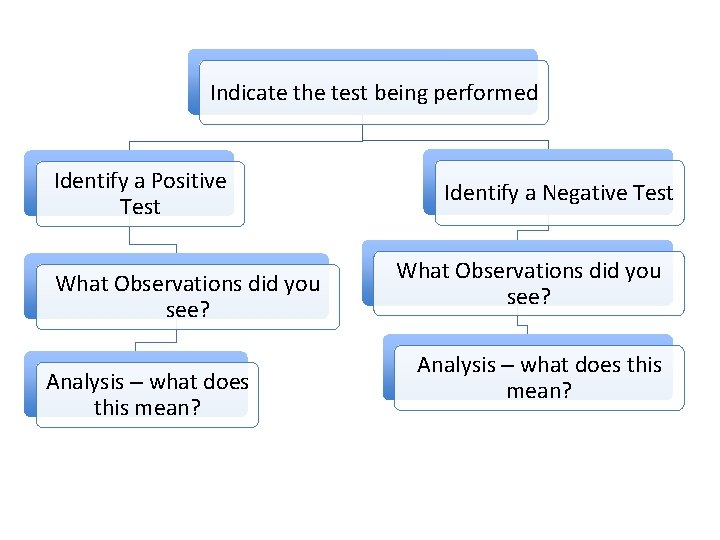
Indicate the test being performed Identify a Positive Test What Observations did you see? Analysis – what does this mean? Identify a Negative Test What Observations did you see? Analysis – what does this mean?

To Complete A Sequential Analysis 1. Locate the possible cations on the solubility table. 1. Determine which anions would precipitate the possible cations. 1. Plan a sequence of precipitation reactions that would use anions to precipitate a single cation at a time. 1. Use filtration between the steps to remove cation precipitates that might interfere with subsequent additions of anions. 1. Draw a flow chart to assist testing & communication.

Example 1 A solution known to contain Pb 2+ Add Na. Cl Positive test is a white precipitate Solution contained Lead(II) ions, precipitated as Pb. Cl 2 Negative test is no precipitate No Lead (II) ions present
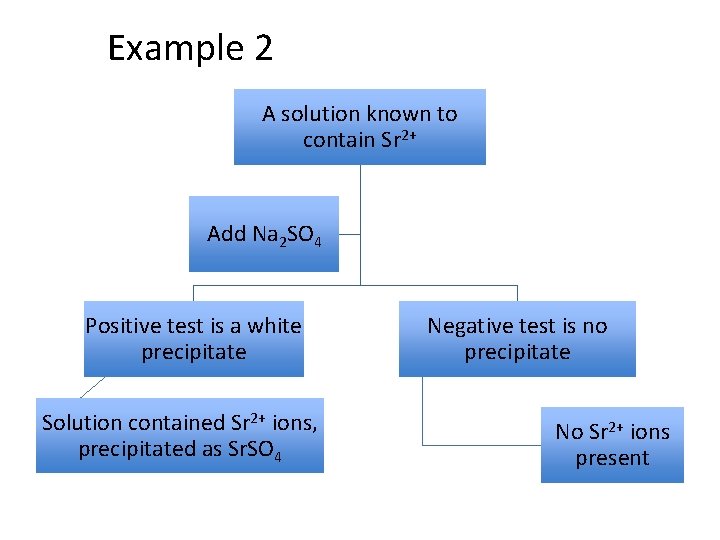
Example 2 A solution known to contain Sr 2+ Add Na 2 SO 4 Positive test is a white precipitate Solution contained Sr 2+ ions, precipitated as Sr. SO 4 Negative test is no precipitate No Sr 2+ ions present
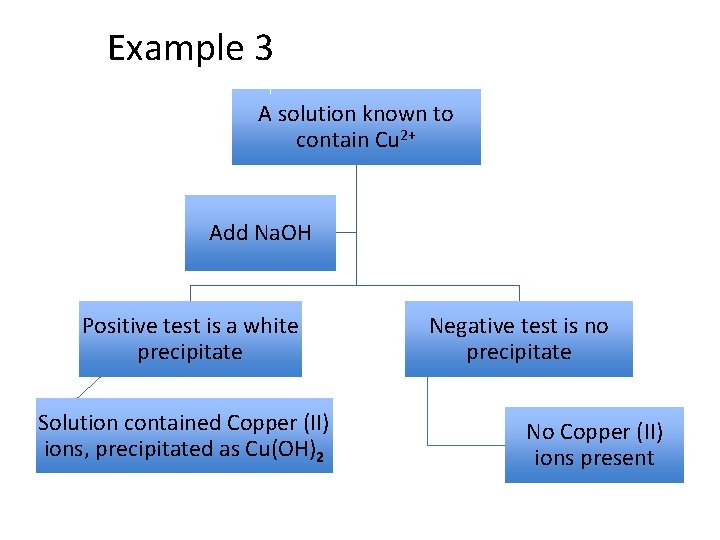
Example 3 A solution known to contain Cu 2+ Add Na. OH Positive test is a white precipitate Solution contained Copper (II) ions, precipitated as Cu(OH)2 Negative test is no precipitate No Copper (II) ions present

To Complete A Sequential Analysis 1. Locate the possible cations on the solubility table. 2. Determine which anions would precipitate the possible cations. 3. Plan a sequence of precipitation reactions that would use anions to precipitate a single cation at a time. 4. Use filtration between the steps to remove cation precipitates that might interfere with subsequent additions of anions. 5. Draw a flow chart to assist testing & communication.

You try! • Design a sequential analysis for each of the ions below, to determine if it is present in the solution. a. Ag+ b. Ca 2+ c. Mg 2+

Example with Two Ions • A solution is thought to contain lead (II) ion or magnesium ion. Design a sequential qualitative analysis to determine whether the solution contains either or both of these ions

Solution known to contain Pb 2+ (aq) or Mg 2+ (aq) Add Na. Cl – the Na+ is always soluble, the Cl- will ppt with Pb 2+, the Cl- is soluble with Mg 2+ Pb 2+ If Solution contains ions, a ppt will form of Pb. Cl 2 Solution contained Pb 2+ ions, precipitated as Pb. Cl 2 If Solution does not contain Pb 2+ ions then no ppt will form No Lead (II) ions present

• If a positive test occurred, filter off the ppt and run a second test for the second cation
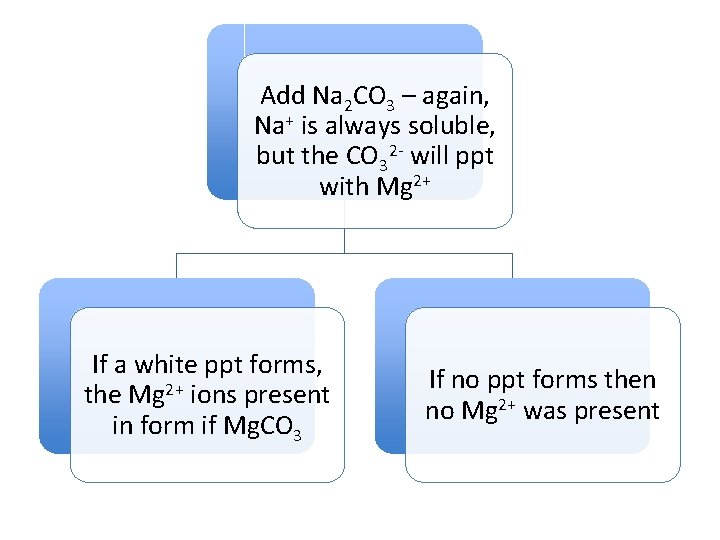
Add Na 2 CO 3 – again, Na+ is always soluble, but the CO 32 - will ppt with Mg 2+ If a white ppt forms, the Mg 2+ ions present in form if Mg. CO 3 If no ppt forms then no Mg 2+ was present

Another Example with Two Ions • A solution is thought to contain calcium ion and mercury (II) ion. Design a sequential qualitative analysis to determine whether the solution contains either or both of these ions

Solution known to contain Ca 2+ (aq) or Hg 2+ (aq) Add Na. Cl – the Na+ is always soluble, the Cl- will ppt with Hg 2+, the Cl- is soluble with Ca 2+ Hg 2+ If Solution contains ions, a ppt will form of Hg. Cl 2 Solution contained Hg 2+ ions, precipitated as Hg. Cl 2 If Solution does not contain Hg 2+ ions then no ppt will form No Hg 2+ ions present

• If a positive test occurred, filter off the ppt and run a second test for the second cation
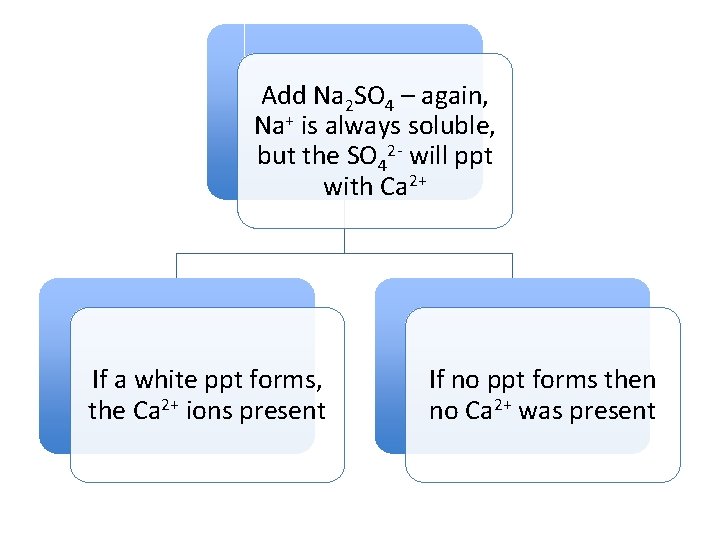
Add Na 2 SO 4 – again, Na+ is always soluble, but the SO 42 - will ppt with Ca 2+ If a white ppt forms, the Ca 2+ ions present If no ppt forms then no Ca 2+ was present

You Try!! • Design a sequential analysis for each of the pairs of ions below, to determine if one or both are present in the solution. a. Ba 2+ and Pb 2+ b. Ag+ and Zn 2+ c. Sr 2+ and Mn 2+ d. Cu+ and Fe 3+

Example containing three ions! Solution known to contain Ag+1 aq or Cu 2+aq or Ba 2+ ion Chose an anion that will produce a ppt for one of the cations Add Na. Cl – the Na+ is always soluble, but the Cl- will ppt with Ag 1+ If Solution contains ions, a ppt will form of Ag. Cl If Solution does not contain Ag 1+ ions then no ppt will form

• If a positive test occurred, filter off the ppt and run a second test for the second cation
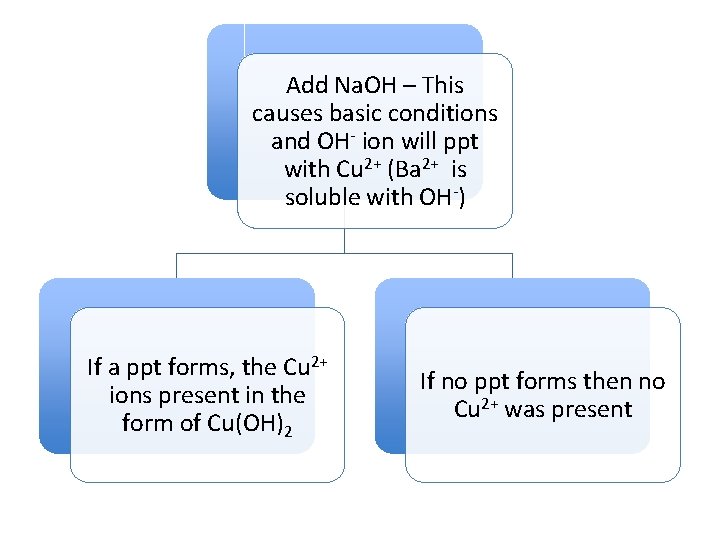
Add Na. OH – This causes basic conditions and OH- ion will ppt with Cu 2+ (Ba 2+ is soluble with OH-) If a ppt forms, the Cu 2+ ions present in the form of Cu(OH)2 If no ppt forms then no Cu 2+ was present

• If a positive test occurred, filter off the ppt and run a third test for the third cation
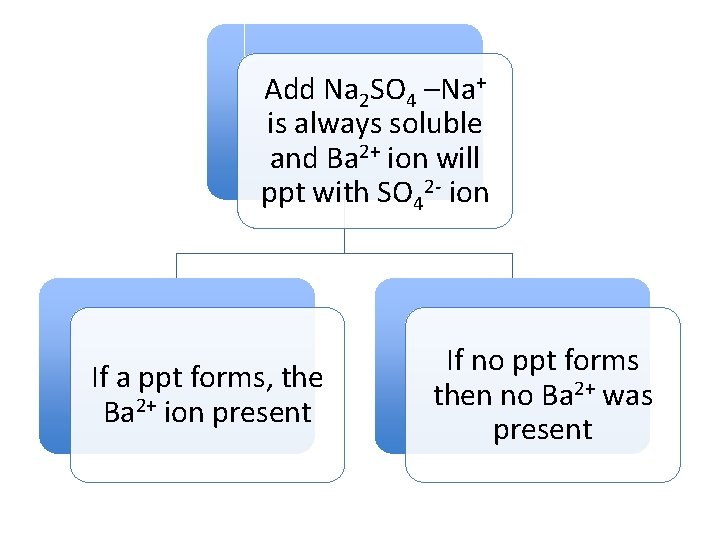
Add Na 2 SO 4 –Na+ is always soluble and Ba 2+ ion will ppt with SO 42 - ion If a ppt forms, the Ba 2+ ion present If no ppt forms then no Ba 2+ was present

• You should perform the colour of solution test and the PPT tests to narrow down which ions you think are present. • You should also determine the colours that each ion would produce in a flame test before performing the flame test • The flame test is not used to confirm or eliminate the presence of a particular ion.
- Slides: 37