QUALITATIVE ANALYSIS Solubility Colour Tests for Cations Tests
QUALITATIVE ANALYSIS Solubility & Colour Tests for Cations Tests for Anions Tests for Gases Tests for Oxidising / Reducing Agents

What is QA ? • The process of determining the identity of an unknown substance, based on a series of chemical tests. • Salts consist of cations (+ ions) and anions (- ions).

Solubility & Colour of salts • The solubility of a salt can help us narrow down the possible identity of the salt. • Soluble : All nitrates, hydrogen carbonates, & sodium, potassium & ammonium salts. Most sulphates, chlorides, bromides & iodides. A few carbonates & hydroxides.

• Insoluble : Most carbonates & hydroxides (except Na+, K+ & NH 4 + ); Pb. SO 4 , Ba. SO 4 , Ca. SO 4 , Ag. Cl , Pb. Cl 2 , Ag. Br, Pb. Br 2, Ag. I, Pb. I 2.

• The colour of a salt also can help narrow down the possible identity of the salt. • E. g. : – – – Blue : Green : Pale Green : Reddish brown : Grey : Black : Cu 2 + / Co 2+ salts Cu 2+ / Ni 2+ salts Fe 3+ salts / Cu metal Metals Metal oxides / sulphides i. e. Cu. O , Cu. S

Action of Heat • On heating a solid, things to observe are : – Change in colour of solid – Change in state – Effervescence ( Test for gas evolved ) • The colour changes involved & the gases liberated will give us a guide to the identity of the cations / anions present.

• Change in colour : – White solid Yellow White – Zinc ions likely to be present. – White solid Orange Yellow – Lead (II) ions likely to be present.
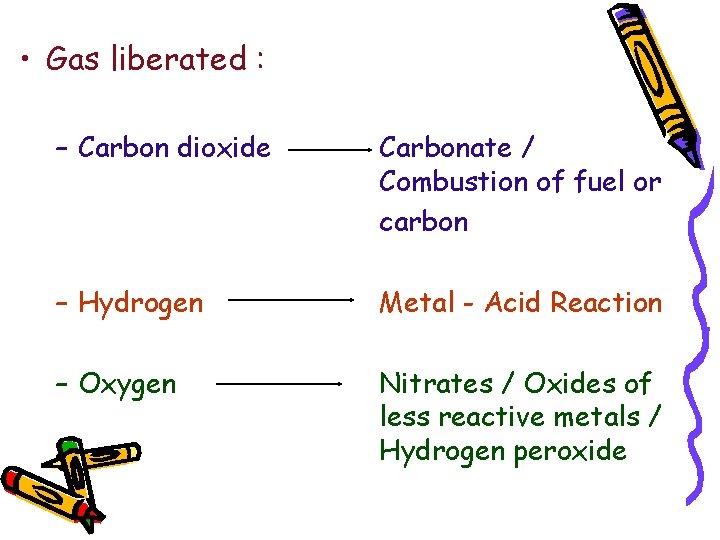
• Gas liberated : – Carbon dioxide Carbonate / Combustion of fuel or carbon – Hydrogen Metal - Acid Reaction – Oxygen Nitrates / Oxides of less reactive metals / Hydrogen peroxide
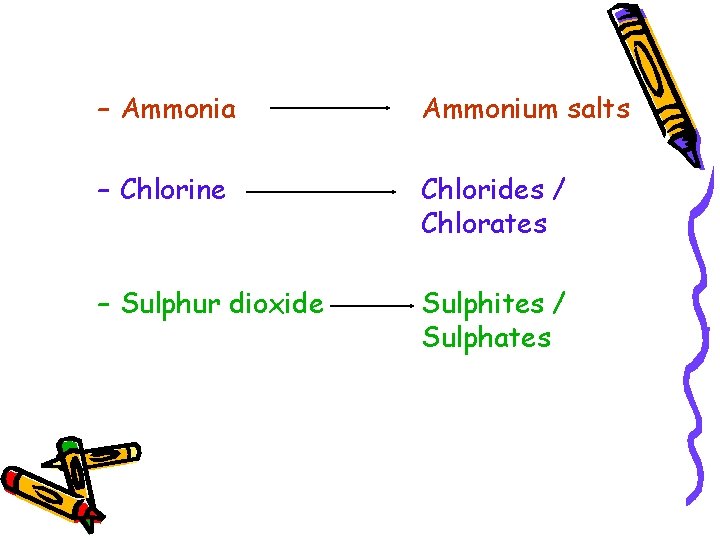
– Ammonia Ammonium salts – Chlorine Chlorides / Chlorates – Sulphur dioxide Sulphites / Sulphates

Testing for Cations • Add sodium hydroxide solution until excess OR • Add aqueous ammonia until excess • Observe for the presence of a precipitate & note its colour; then the solubility of the ppt in excess alkali.
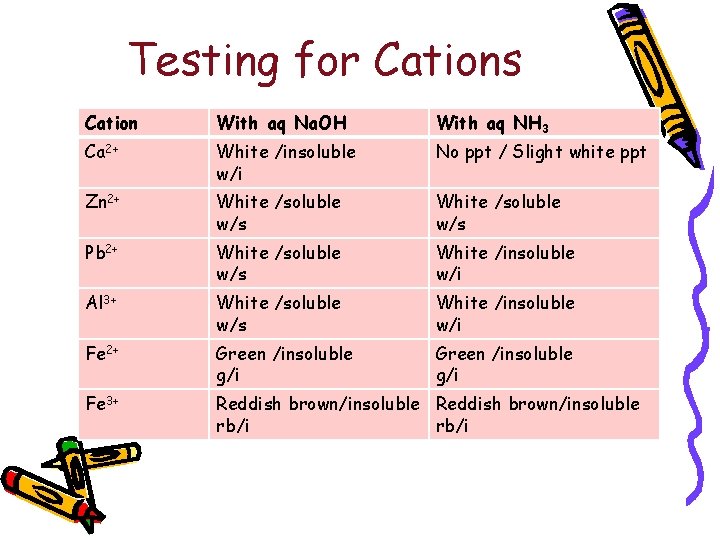
Testing for Cations Cation With aq Na. OH With aq NH 3 Ca 2+ White /insoluble w/i No ppt / Slight white ppt Zn 2+ White /soluble w/s Pb 2+ White /soluble w/s White /insoluble w/i Al 3+ White /soluble w/s White /insoluble w/i Fe 2+ Green /insoluble g/i Fe 3+ Reddish brown/insoluble rb/i

Testing for Cations Cation With aq Na. OH With aq NH 3 Cu 2+ Blue /insoluble b/i Blue /soluble in excess to form a dark blue solution b/s NH 4+ No ppt /on warming, gas Not applicable (WHY? ? ? ) produced turns moist red litmus blue No ppt / ammonia gas

Testing for Anions • Carbonates : – All carbonates react with dilute acids (HCl & H 2 SO 4) to form salt, water & CO 2 gas. – Add dilute acid to a portion of the unknown solid & look for effervescence. Bubble the gas formed into limewater. If the gas is CO 2, a white ppt is formed with limewater (aq calcium hydroxide).

• For Chlorides : – Chloride ions react with aqueous silver nitrate to form a white ppt which is insoluble in dilute nitric acid, but soluble in aqueous ammonia. – To a portion of the solution of the unknown, add dilute nitric acid followed by aqueous silver nitrate. Formation of a white ppt indicates the presence of chloride ions. Ag+(aq) + Cl-(aq) Ag. Cl(s)

• For Iodides : – Iodide ions react with aqueous silver nitrate / aqueous lead (II) nitrate to form a yellow ppt of silver iodide or lead (II) iodide respectively. The ppt is insoluble in both dilute nitric acid & aqueous ammonia.

– To a portion of the solution of the unknown, add dilute nitric acid followed by aqueous silver nitrate / aqueous lead (II) nitrate. Formation of a yellow ppt indicates the presence of iodide ions. Ag+(aq) + I- (aq) Ag. I(s) Pb 2+(aq) + 2 I-(aq) Pb. I 2(s)

• For Sulphates: - To a portion of the solution of the unknown, add dilute nitric acid followed by aqueous barium nitrate OR dilute hydrochloric acid followed by aqueous barium chloride. – Formation of a white ppt indicates the presence of sulphate ions. Ba 2+(aq) + SO 4 2 -(aq) Ba. SO 4(s)

• For Nitrates : – To a portion of the solution of the unknown, add aqueous sodium hydroxide, followed by aluminium foil / Devarda’s alloy. Warm the mixture gently and test for evolution of ammonia gas. – Evolution of a colourless pungent gas that turns moist red litmus blue indicates the presence of nitrate ions.

Test for Reducing Agents • Using Acidified Potassium Manganate (VII) solution – To a sample of the solution of the unknown substance, add an equal portion of aqueous acidified potassium manganate (VII). – If the acidified potassium manganate (VII) solution is decolourised (No purple colouration remaining), the unknown solution contains a reducing agent.

Test for Reducing Agents • Using Acidified Potassium Manganate (VII) solution – Note the colour of the resulting solution. – i. e. Solution X decolourises aq. acidified potassium manganate (VII). Solution X turns from pale green to yellow. (Resulting solution is yellow)

Test for Reducing Agents • Using Acidified Potassium Dichromate (VI) solution – To a sample of the solution of the unknown substance, add an equal portion of aqueous acidified potassium dichromate (VI). – If the acidified potassium dichromate (VI) solution turns from orange to green, the unknown solution contains a reducing agent.

Test for Oxidising Agents • Using Acidified Potassium Iodide solution – To a sample of the solution of the unknown substance, add an equal portion of aqueous acidified potassium iodide. – If the solution turns brown (and formation of a black ppt on standing), the unknown solution contains an oxidising agent.

Test for Oxidising Agents • Using Iron (II) Sulphate solution – To a sample of the solution of the unknown substance, add an equal portion of aqueous iron (II) sulphate. – If the pale green solution turns yellow (oxidation of iron (II) ions to iron (III) ions), the unknown solution contains an oxidising agent.

Tests for Gases • Hydrogen: - Insert a lighted splint into a tube of the gas. If the gas extinguishes the lighted splint with a ‘pop’ sound, the gas is hydrogen gas.

Tests for Gases • Oxygen: - Insert a glowing splint into a tube of the gas. If the gas relights a glowing splint, the gas is oxygen gas.

Tests for Gases • Carbon dioxide: - Bubble the gas into a tube of aqueous calcium hydroxide (limewater). If the gas forms a white precipitate with limewater, the gas is carbon dioxide gas.

Tests for Gases • Chlorine: - Place a piece of moist blue litmus paper at the mouth of a tube of the gas. If the gas turns moist blue litmus red and bleaches it, the gas is chlorine gas.

Tests for Gases • Ammonia: - Place a piece of moist red litmus paper at the mouth of a tube of the gas. If the gas turns moist red litmus blue, the gas is ammonia gas.

Tests for Gases • Sulfur dioxide: - Dip a piece of filter paper into aqueous acidified potassium manganate(VII) and insert the paper into a tube of the gas. If the gas turns purple acidified potassium manganate (VII) colourless (if the gas decolourises purple acidified potassium manganate(VII)), the gas is sulfur dioxide gas.
- Slides: 29