Qualification of Hot Air Oven PART I Department


























- Slides: 33

“Qualification of Hot Air Oven” PART - I Department of Pharmaceutical Sciences & Technology Birla Institute of Technology, Mesra Ranchi – 835 215 (Jharkhand) 1

CONTENTS v. Introduction • Validation • Calibration v. Qualification v. Sterilization v. Dry Heat Sterilization v. Hot Air Oven • Validation Test Equipment • Basic Validation Approach • Qualification Approach v. Tablet Compression Machine • Introduction • Qualification Approach v. Routine Monitor after validation v. Conclusion v. References 2

INTRODUCTION 3

VALIDATION • As per USFDA: It is establishing documented evidence which provides a high degree of assurance that a specific process will consistently produce a product meeting its pre-determined specifications and quality attributes. • Purpose of Validation. ? 4

When should be validated ? • Any Change in established method New method developed for a particular problem Established method used in different laboratories, different equipment or different staff Out-of-control situations within internal quality assurance Demonstration of equivalence between two methods (e. g. a rapid new test against a standard method) 5

Who should validate? 6

• The vendor or the user has the ultimate responsibility for the accuracy of the analysis results and also for equipment qualification. • DQ should always be done by the user. • While IQ for a small and low cost instrument is usually done by the user, IQ for large, complex and high cost instruments should be done by the vendor. • OQ can be done by either the user or the vendor. • PQ should always be done by the user because it is very application specific and the vendor may not be familiar with these. 7

CALIBRATION • The process quantitatively defining system responses to known controlled signal output. • To determine, check, or rectify the graduation of any instrument giving quantitative measurements. 8

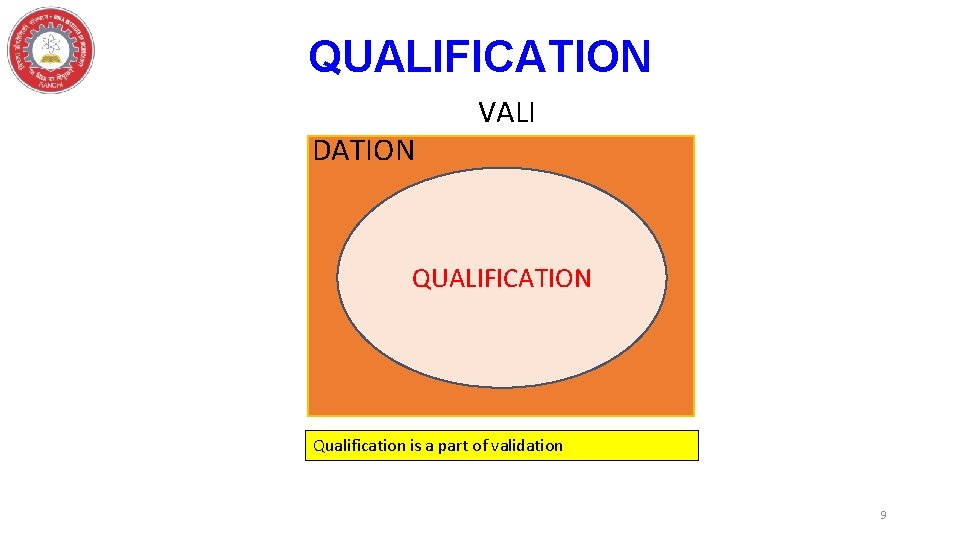
QUALIFICATION DATION VALI QUALIFICATION Qualification is a part of validation 9

QUALIFICATION • The action of proving that any equipment Works correctly and leads to the expected results. • When a validation approach is related to a machine or equipment which is appropriately designed, located, installed, operated and maintained to suit their itended purpose. 10

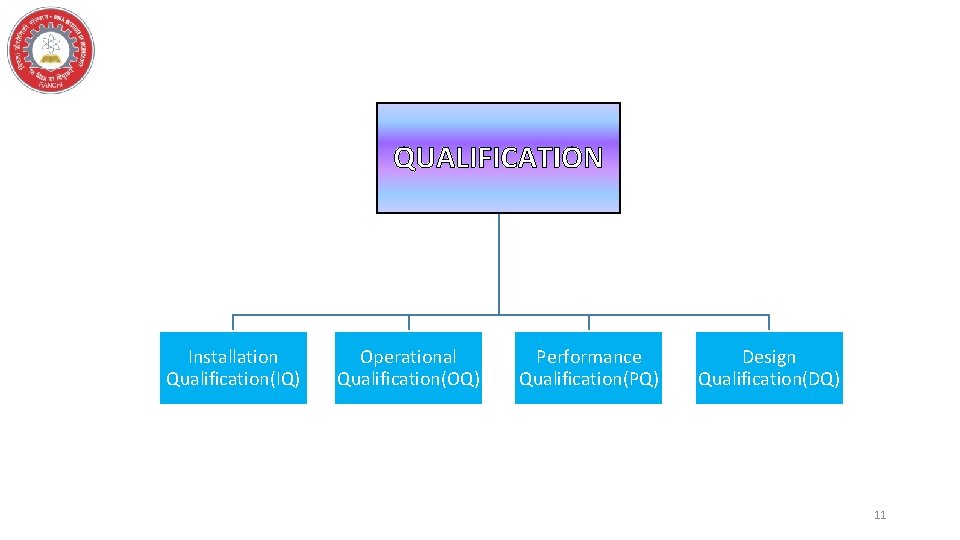
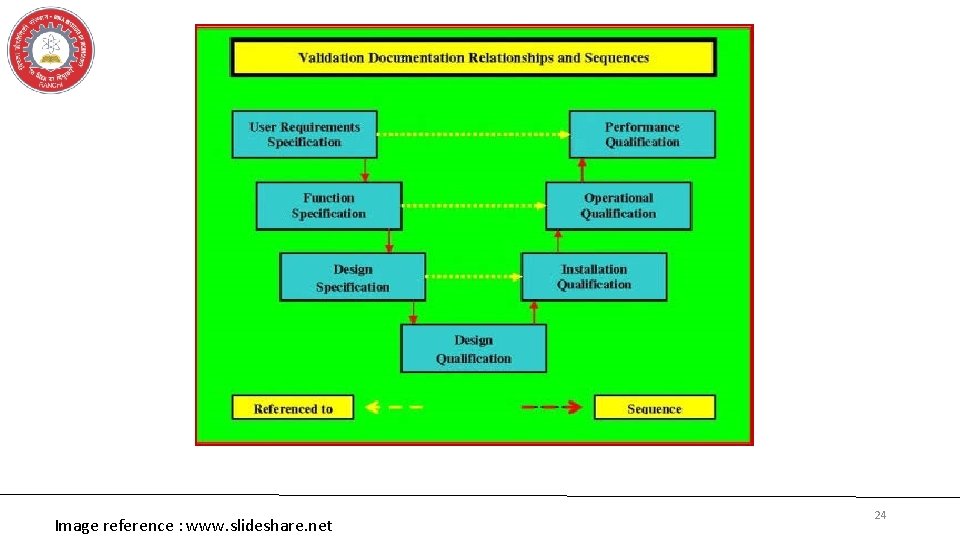
QUALIFICATION Installation Qualification(IQ) Operational Qualification(OQ) Performance Qualification(PQ) Design Qualification(DQ) 11

12

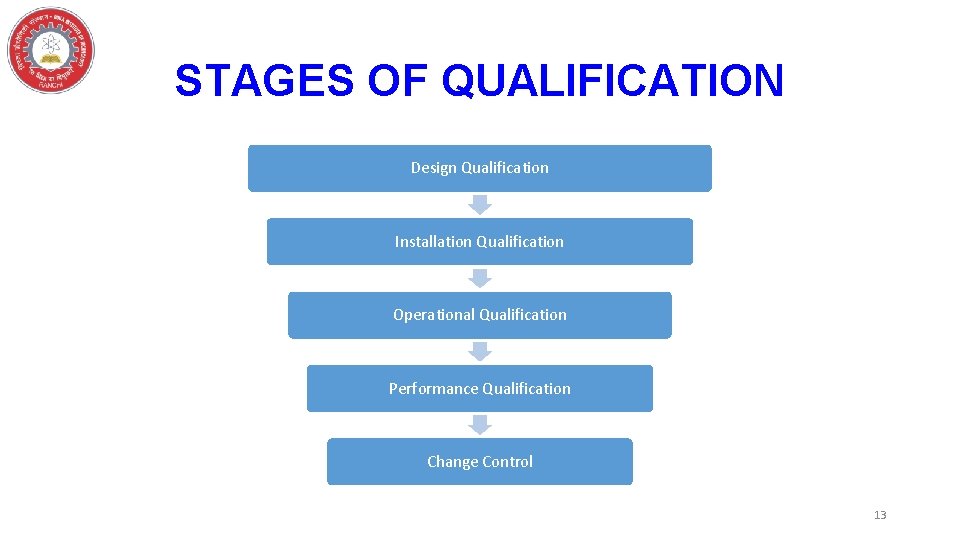
STAGES OF QUALIFICATION Design Qualification Installation Qualification Operational Qualification Performance Qualification Change Control 13

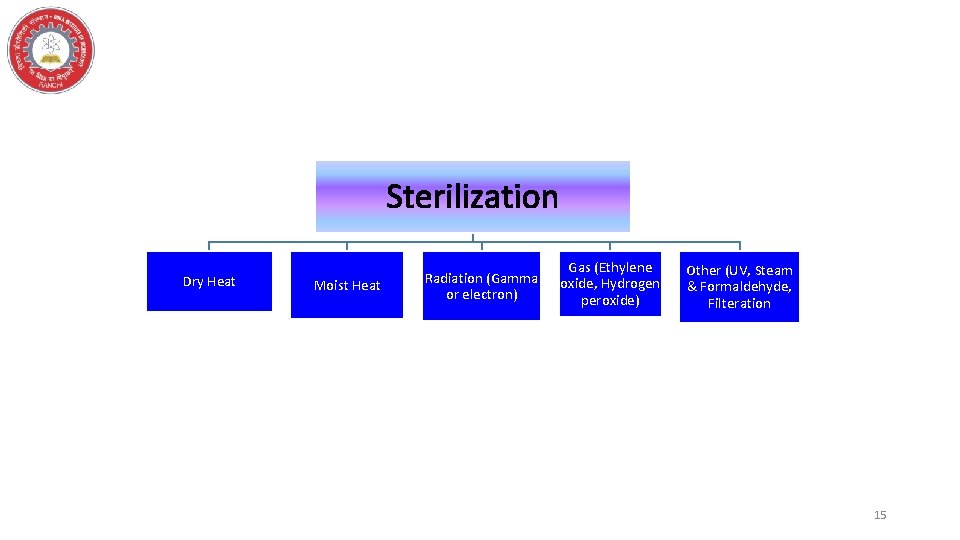
Sterilization • “The act or process, physical or chemical, that destroys or eliminates all viable microbes including resistant bacterial spores from a fluid or a solid. ” 14

Sterilization Dry Heat Moist Heat Radiation (Gamma or electron) Gas (Ethylene oxide, Hydrogen peroxide) Other (UV, Steam & Formaldehyde, Filteration 15

Dry Heat Sterilization • Dry heat is the most commonly used methods to sterilize and/or depyrogenate pharmaceutical components (glasswares) and products. Most often, it is used for depyrogenation of parenteral containers is performed utilizing a dry heat oven. • Dry heat, as the name indicates, utilizes hot air that is either free from water vapor, and where this moisture plays minimal or no role in the process of sterilization. • The process of sterilization within a chamber or hot air tunnel is a critical process and there is regulatory requirement for validation of the process. 16

Types of dry heat sterilizers q. Commonly employed dry heat sterilizers in pharmaceutical industry are • Forced Convention batch sterilizers. • Infrared Tunnel sterilizers. • Forced convention tunnel sterilizers. • Continuous flame sterilizers microwave • Laser/Plasma sterilizers. 17

Types of Dry Heat Sterilizers Commonly utilized dry heat sterilizers in pharmaceutical industry are- Hot Air Oven The Tunnel System. 18

HOT AIR OVEN Higher temperatures and longer exposure times required • Typical cycles: • 160°C for 120 minutes • 170°C for 60 minutes • 180°C for 30 minutes • Used for: • glassware and product containers used in aseptic manufacture, nonaqueous thermo stable powders and liquids (oils) • also used for de pyrogenation of glassware. Image reference : www. slideshare. net 19

Validation Test Equipment used for validation testing of oven are listed below: v. Resistance temperature detectors v. Thermocouples v. Data logger v. Constant temperature bath v. Stopwatch v. Voltmeter v. Optical tachometer 20

Qualification Approach 21

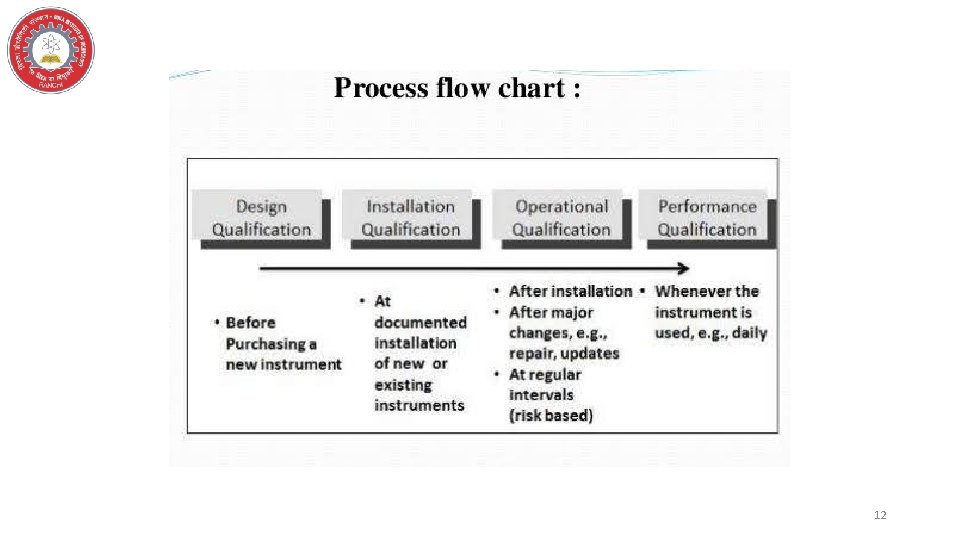
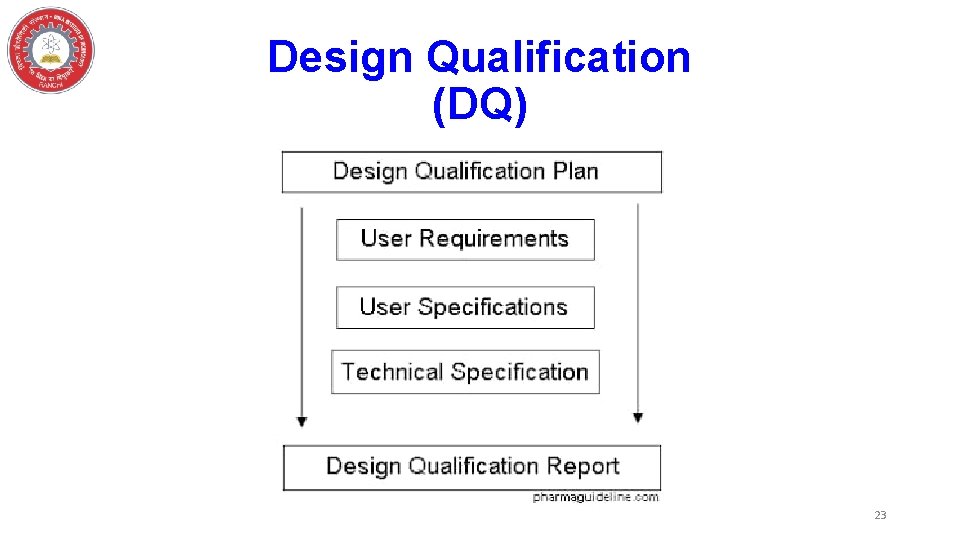
Basic Validation Approach q. Design qualification (DQ): • The DQ outline the key features of the system designed to address the user requirement, regulatory compliance and selection rationale of a particular supplier. q. The following are the key considerations for DQ: • Physical dimensions of the equipment and accessories. • Suitable operating environment of the instrument • Health and safety requirement 22

Design Qualification (DQ) 23

Image reference : www. slideshare. net 24

Installation Qualification (IQ) • It is carried out after or concurrently with the installation of the equipment at the user’s premises. • The purpose is to provide documentary evidence that the correct equipment has been received and installed as per plan and protocol. • IQ documents should be reviewed and approved by designated responsible individuals. • It includes details of- • Structural- Check dimensions, presence of seal • Filters- Proper identification, type, size, air capacity, flow rate Contd. 25

• Electricals- Proper identification, safety cutoff • HVAC- System provides the temperature and pressure differential required. • Air supply- Identify source, duct size. • Air or natural gas- Check that the source and type of supply are consistent with the manufacturer’s recommendations. • Heaters- Record the manufacturer's model no. , the no. of heating elements. • Blowers- Check for use of correct fan belt & that is in good condition. 26

Operational Qualification (OQ) • It is documented verification that the system or subsystem performs as intended throughout all specified operating range • The OQ document should be reviewed and signed by the required department representatives. • The components of system must satisfy the operating ranges as determined by the purchase order specifications. 27

Each of the following process components must be identified & the operating performance & range determined. • Cycle timer - The accuracy of timer must be determined, so that assurance is provided for cycle time. • Door interlocks - If a unit is equipped with double doors, the interlocks must operate such that the door leading to the aseptic area cannot be opened if the door to the non-aseptic area is opened. • Heaters - All of the heating elements must be functional. • Blowers - The air velocity consistent and motor speed of blowers should be noted in the OQ records. 28

• Cooling Coils- If coils are present, the type and size of the coils and temperature of the cooling medium at the inlet and outlet of the coils should be recorded. • Chamber leaks- The perimeter of the doors for batch sterilizers should be checked for air leakage while operating. • Particulates counts- Particulate count should be checked within the containers before and after sterilization to quantitate the particle load contributed to the product by sterilization process. 29

Performance Qualification (PQ) • Verifies that the equipment performs according to design specifications and user defined requirements in a reliable & reproducible manner under normal production conditions • Physical • Heat penetration studies on empty chamber • Heat distribution study on loaded chamber • Heat penetration study on loaded chamber • • Microbiological • Bio-challenge/ Pyro -challenge studies 30

Physical • Heat Penetration Studies On Empty Chamber üTo identify heat distribution patterns including slowest heating points ü Multiple temperature sensing devices should be used(Thermocouples) üTemperature profile locate hot/cold areas in the sterilizer by mapping temperature at various location. • Heat Distribution Study on Loaded Chamber ü. Multiple thermocouples throughout chamber to determine effect of load configuration on temperature distribution. üTemperature distribution for all loads using all container sizes used in production should be tested. üRepeat runs should be performed to check variability. üTemperature profile for each chamber load configuration should be documented 31

Cont. . • Bio-Challenge/ Pyro-Challenge • Heat Penetration Study on Studies Loaded Chamber ü The challenge should ü Designed to determine the demonstrate the lethality delivered location of the slowest heating by the cycle with either point within a oven at various microorganisms or endotoxins locations. ü Each size & type of material üResistant bacterial spores are should be tested by penetration available primarily in the form of spore strips and. Spore suspension. studies. üHot areas in the load are more important for heat labile items. 32

References 1. Laurie BC, Gayle DH, Dry Heat Sterilization and Depyrogenation Validation and Monitoring, In: , James AF, Carleton J, Editors. Validation Of Pharmaceutical Process, informa healthcare; P. 223 -239. 2. Potdar AM. Pharmaceutical Quality Assurance, 1 st ed. Published by Nirali Prakashan; 2006. P. 8. 13 -8. 20. 3. Syed Imtiaz Haider. Pharmaceutical Master Validation Plan. The Ultimate Guide to FDA, GMP and GLP Compliance. informa healthcare; 2010. P. 138 -139. 4. https: //biologicalindicators. mesalabs. com/2015/12/14/validation-of-dry-heatsterilizationprocesses 5. https: //www. pharmatutor. org/articles/validation-dry-heat-sterilization-methods. 6. http: //www. mackpharmatech. com/doc. htm 7. http: //www. ehow. com/list_6809815_equipment 33