QQ Consider yesterdays lab Summarize your conclusion 1

QQ: Consider yesterday’s lab: Summarize your conclusion. 1) What was your hypothesis? 2) What was your conclusion? 3) Give evidence and supporting details. 4) Evaluation: Why is it important to document your observations and data? 5) What do you think the substances were?

Today’s Objective: I can name cations and anions using scientific rules.

Introducing: Ions

1. 3 Potassium Aluminum Sulfate 2 How close are your 2. Magnesium sulfate sketches? 4 3. Sodium chloride 4. Sucrose

1. Potassium aluminum sulfate 2. Magnesium sulfate 3. Sodium chloride 4. Sucrose
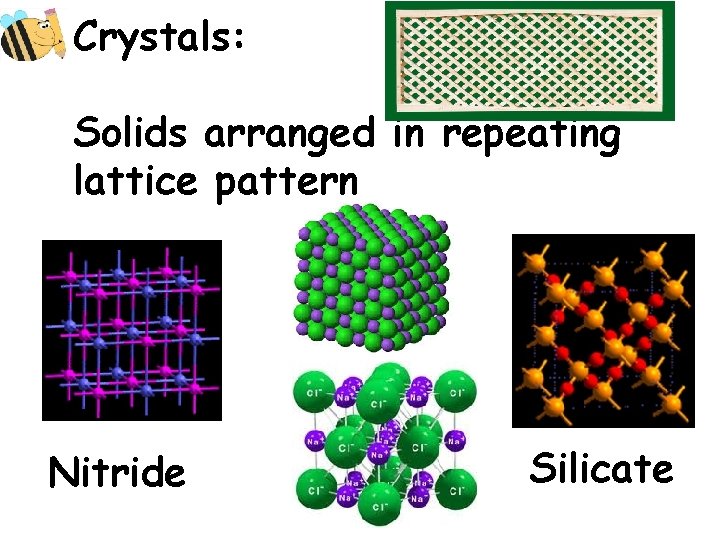
Crystals: Solids arranged in repeating lattice pattern Nitride Silicate
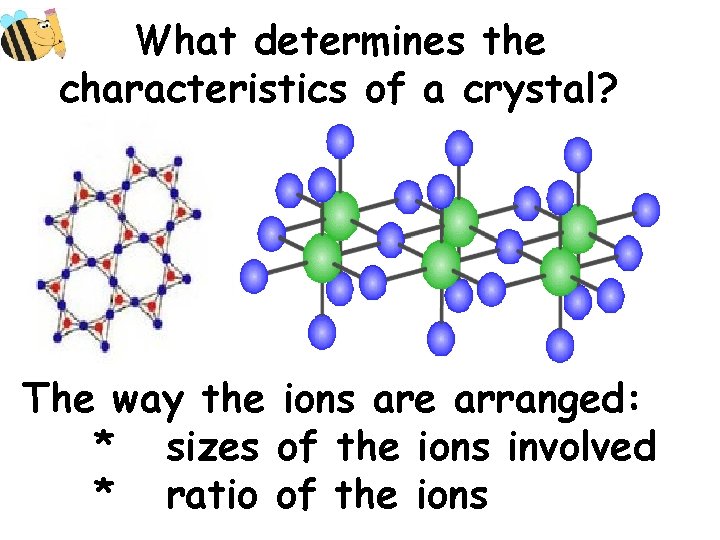
What determines the characteristics of a crystal? The way the ions are arranged: * sizes of the ions involved * ratio of the ions

Properties of Ionic Compounds Crystals Hard & Brittle As solid – poor conductor As liquid – good conductor
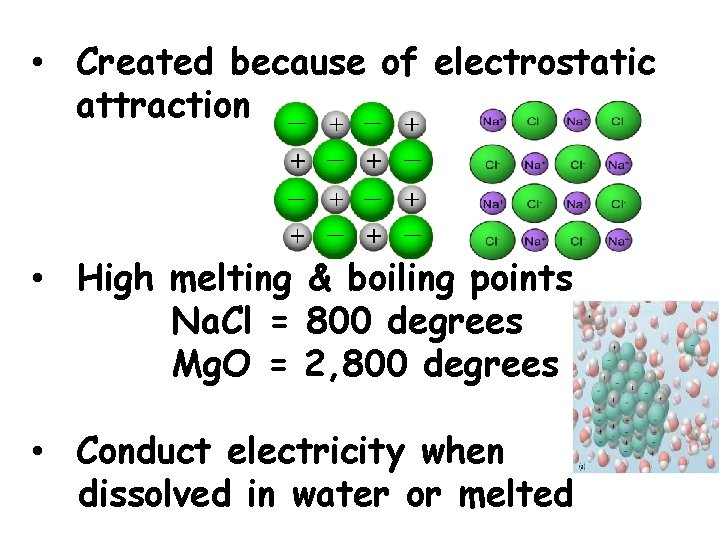
• Created because of electrostatic attraction • High melting & boiling points Na. Cl = 800 degrees Mg. O = 2, 800 degrees • Conduct electricity when dissolved in water or melted
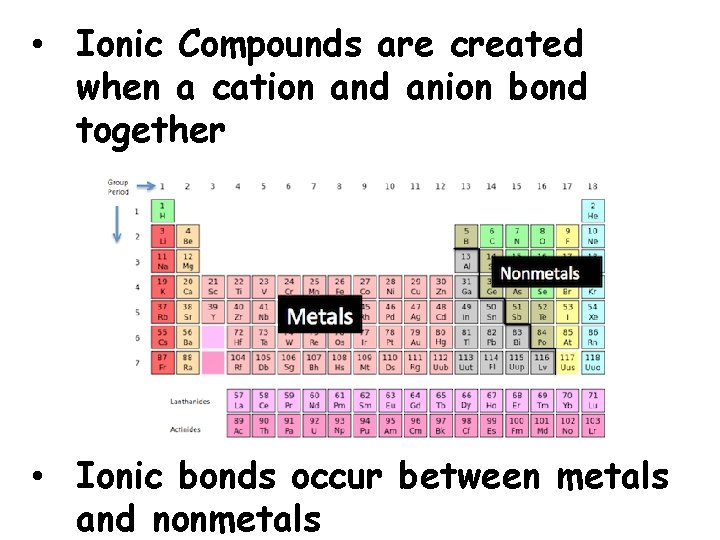
• Ionic Compounds are created when a cation and anion bond together • Ionic bonds occur between metals and nonmetals
Is fluoride the same as fluorine?

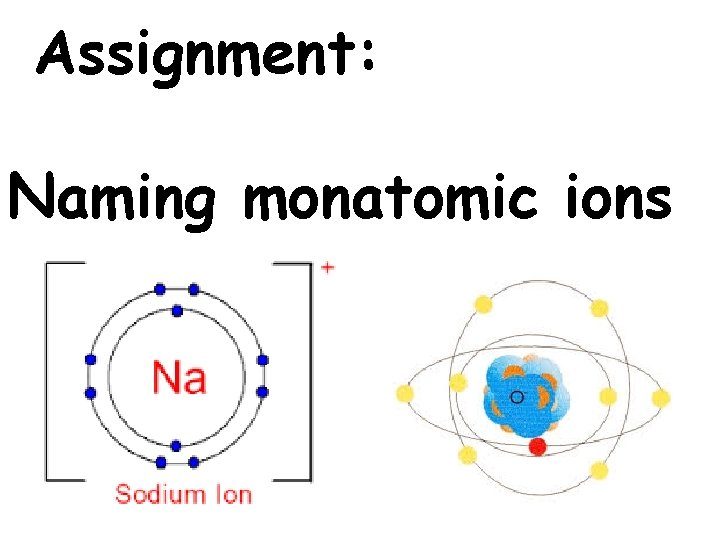
Name an anion: Element Name + -ide -1 Cl Chlor ide -1 S Sulf ide

They’ve lost e

Name a cation: Element Name +1 Na Sodium +2 Mg Magnesium

Formula tells the ratio of the atoms in the compound. Al 2 O 3 Ruby Na. Cl Table Salt Si. O 2 Quartz

Coefficient: 3 x Means x + x Or 3 times x # in front of symbol; tells how many of these 3 Na means 3 sodium atoms
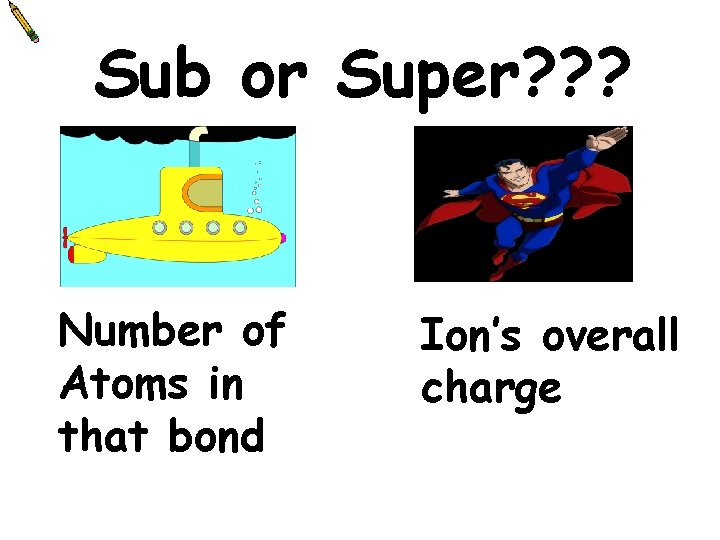
Sub or Super? ? ? Number of Atoms in that bond Ion’s overall charge
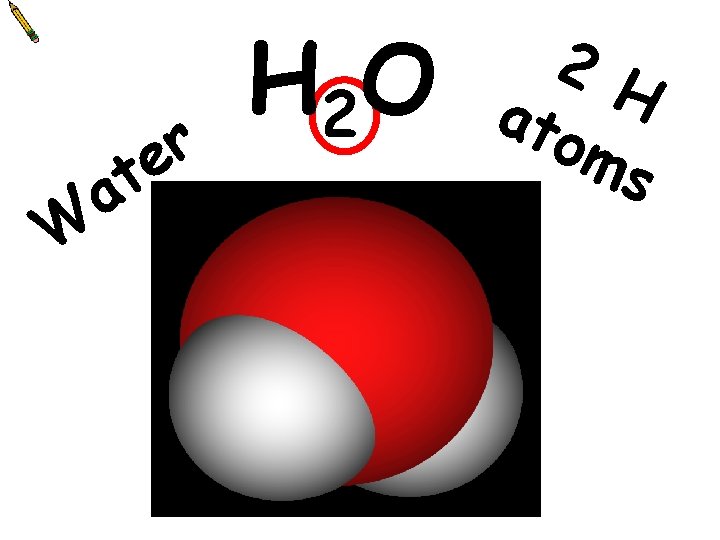
a W r e t H 2 O 2 H ato ms
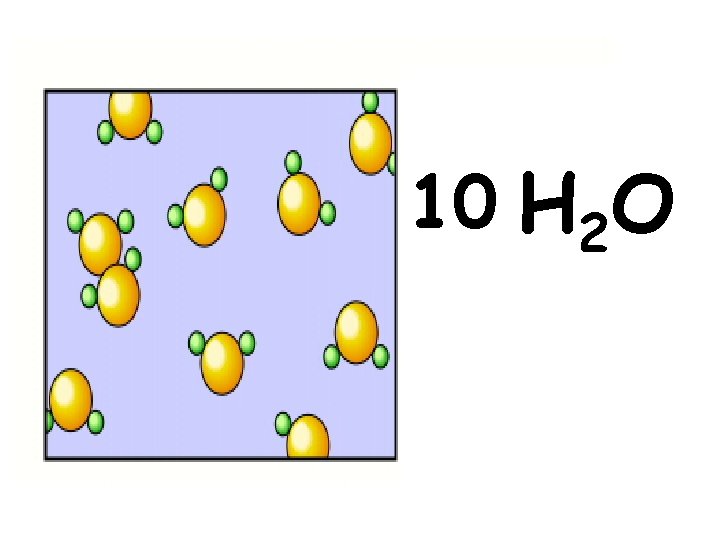
10 H 2 O

Hydroxide -1 OH or OH Overall charge is -1

Assignment: Naming monatomic ions
- Slides: 22