QOTD 51414 Please have out to be checked

QOTD 5/14/14 Please have out to be checked off: • Soap reading/prelab Qs • p. 59 Qs QOTD: Rank the following solutions in order of most acidic to least acidic and explain why. • 1 M vinegar (weak acid) • 1 M HCl (strong acid) • 0. 5 M HCl (strong acid) • 1 M Na. OH (strong base) • 1 M NH 3 (weak base) • Pure water

QOTD Answer QOTD: Rank the following solutions in order of most acidic to least acidic and explain why. 1. 1 M HCl (strong acid) 100% ions (excess H+) 2. 0. 5 M HCl (strong acid) 100% ions (excess H+) 3. 1 M vinegar (weak acid) less than 100% ions (~1%) 4. Pure water neutral 5. 1 M NH 3 (weak base) less basic than a strong base 6. 1 M Na. OH (strong base) strong base = most basic = least acidic

Answers Homework Qs! (p. 59)

Check-in Questions: 1. You have a 1. 0 M solution of a strong acid (HCl) and 1. 0 M of a weak acid (acetic acid). Predict which will have a lower p. H, and explain why: The 1. 0 M HCl because it dissociates completely…more H+ means lower p. H

Check-in Questions: 2. How is the Bronsted-Lowry definition of acid/base theory different from Arrhenius? The Bronsted-Lowry theory says that a base can accept a proton (H+) (in addition to donating a OH-).

Homework Questions: 1. Label the following substances as acids or bases. In each case, list the ions you would expect to form in solution: +, IAcid, H a. Hydroiodic Acid, HI b. Rubidium Hydroxide, Rb. OH Base, Rb+, OHc. Selenous acid, H 2 Se. O 4 Acid, 2 H+, Se. O 24 + d. Phosphine, PH 3 Base, PH 4 , OH e. Calcium hydroxide, Ca(OH)2 Base, Ca 2+, 2 OHf. Perchloric acid, HCl. O 4 Acid, H+, Cl. O 4 -

2. Consider a solution of hydrobromic acid, HBr. If you drew a particle view of HBr in water that contained 10 H+ ions, how many Br- ions would you need? Explain your thinking: 10 Br- ions (to cancel out the 10 H+ ions) 3. Consider a solution of mangnesium hydroxide, Mg(OH)2. If you drew a particle view of this substance with 10 Mg 2+ ions, how many hydroxide (OH- ) ions would you need to draw? 20 OH- ions 4. The CO 32 - ion is a weak base. Explain, using Bronsted-Lowry theory, what makes this substance a base: It can accept a proton (H+)

Challenge: Some solutions conduct electricity better than other solutions. Use your knowledge about the different kinds of acids and bases to explain why a 1. 0 M hydrochloric acid solution is a better conductor of electricity than a 1. 0 M acetic acid solution. The strong acid dissociates completely, making it easier for electricity to flow through the solution. (more ions = more moving charges)

Acids and Bases Notes PLEASE SEND A GROUP MEMBER TO GRAB NOTES SHEETS!

Arrhenius Theory • Acid: A substance that adds hydrogen ions, H+, to an aqueous solution • Base: A substance that adds hydroxide ions, OH-, to an aqueous solution • Not the best definition…
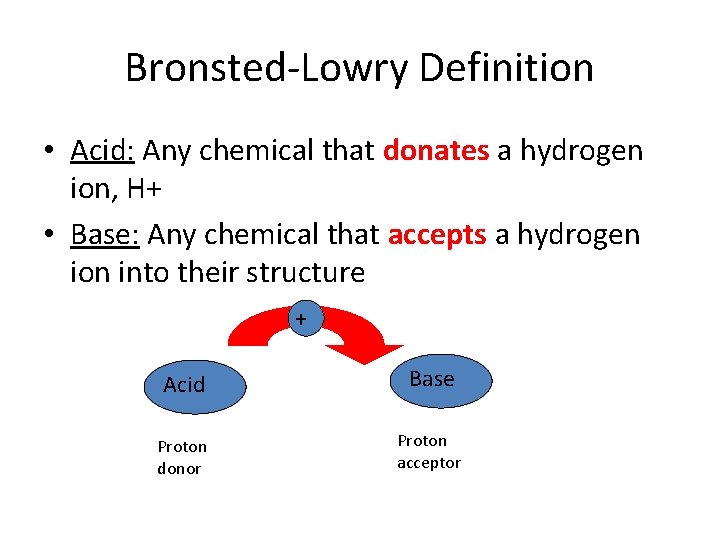
Bronsted-Lowry Definition • Acid: Any chemical that donates a hydrogen ion, H+ • Base: Any chemical that accepts a hydrogen ion into their structure + Acid Base Proton donor Proton acceptor

BAAD • Bases Accept, Acids Donate Example: Acid Base HCl + H 2 O Cl- + H 3 O+ H+ Base Acid NH 3 + H 2 O NH 4+ + OHH+
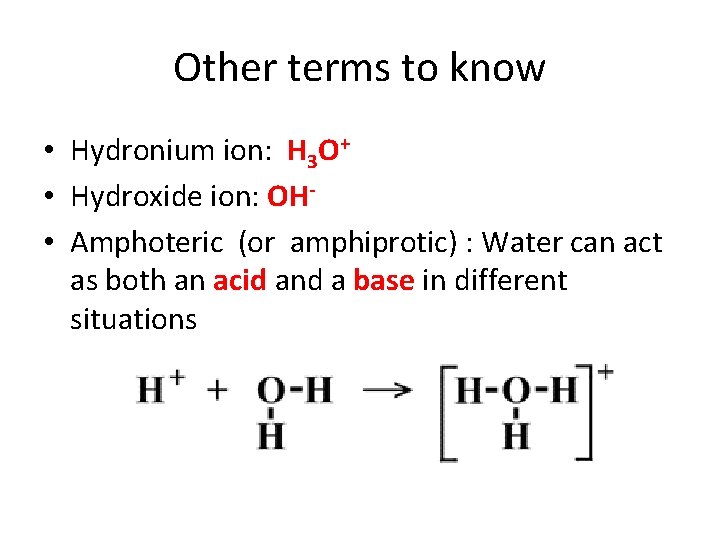
Other terms to know • Hydronium ion: H 3 O+ • Hydroxide ion: OH • Amphoteric (or amphiprotic) : Water can act as both an acid and a base in different situations

Your Turn (partner talk) H 2 O + HBr Br- + H 3 O+ Which compound is acting as an acid? Which is acting like a base? H 2 O HBr

Your Turn (Partner talk) H 2 O + SO 42 - OH- + HSO 4 Which of the compounds is acting as a base? SO 42 -
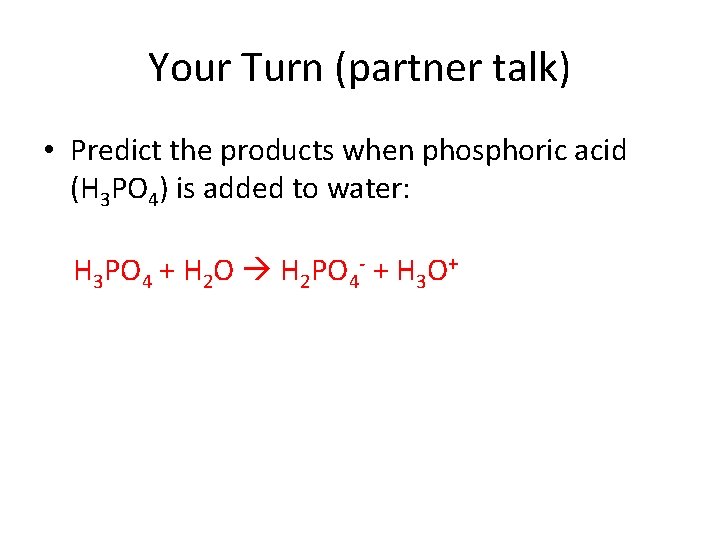
Your Turn (partner talk) • Predict the products when phosphoric acid (H 3 PO 4) is added to water: H 3 PO 4 + H 2 O H 2 PO 4 - + H 3 O+

Properties of Acids • Corrosive • Sour taste • Contains hydrogen ions (H+) when dissolved in water • p. H < 7 • Electrolytes (conduct electricity) • Neutralize bases http: //qldscienceteachers. tripod. com/junior/chem/acid. html

Examples of Acids • • Hydrochloric acid (HCl) in gastric juice Sulphuric acid (H 2 SO 4) Nitric acid (HNO 3) Carbonic acid in soft drinks (H 2 CO 3) Uric acid in urine Ascorbic acid (Vitamin C) in fruit Citric acid in oranges and lemons Acetic acid in vinegar http: //qldscienceteachers. tripod. com/junior/chem/acid. html

Properties of Bases • Taste bitter • Slippery • Electrolytes (conduct electricity) • Base + fat = soap • Neutralize acids • Corrosive http: //qldscienceteachers. tripod. com/junior/chem/acid. html

Examples of Bases • Sodium hydroxide (Na. OH) • Calcium hydroxide ( Ca(OH)2 ) or limewater • Ammonium hydroxide (NH 4 OH) or ammonia water • Magnesium hydroxide ( Mg(OH)2 ) or milk of magnesia • Many bleaches, soaps, toothpastes and cleaning agents http: //qldscienceteachers. tripod. com/junior/chem/acid. html

Indicators • Substances that change color when exposed to an acid or base • Can be used to identify acids and bases, even their strength • Usually a weak acid or base

Examples of indicators • Litmus paper: Red for acid, blue for base • p. H paper: universal indicator, compare colors to known p. H • Cabbage juice: color changes in response to concentration of H+ ions • Phenolphthalein: color is clear/cloudy in an acid and pink in a base

Solutions • Acidic solution: the hydronium ion concentration is higher than the hydroxide ion concentration [H+] > [OH-] • Basic solution: hydroxide ion concentration is higher than the hydronium ion concentration [H+] < [OH-]
![• Neutral solution: concentration of hydronium and hydroxide ions is the same [H+] • Neutral solution: concentration of hydronium and hydroxide ions is the same [H+]](http://slidetodoc.com/presentation_image_h/be511b3425e32968d3f492d2c5d8c7fc/image-24.jpg)
• Neutral solution: concentration of hydronium and hydroxide ions is the same [H+] = [OH-] – Ex. Pure water – Created when equal quantities of an acid and base are combined
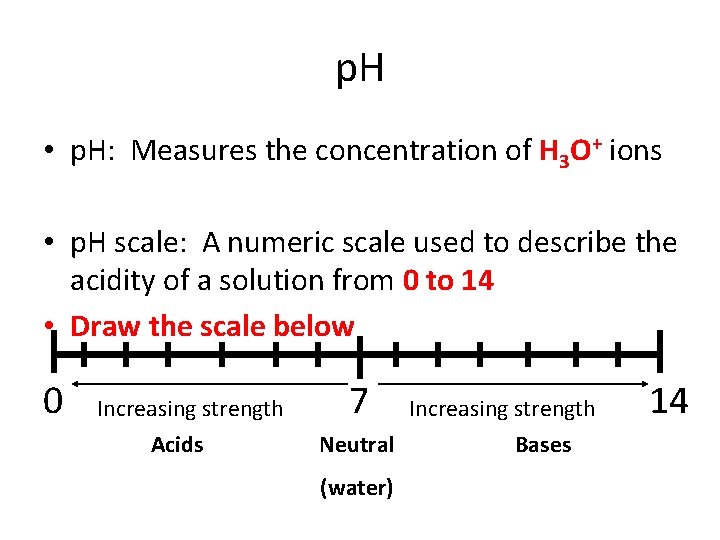
p. H • p. H: Measures the concentration of H 3 O+ ions • p. H scale: A numeric scale used to describe the acidity of a solution from 0 to 14 • Draw the scale below 0 Increasing strength Acids 7 Increasing strength Neutral Bases (water) 14
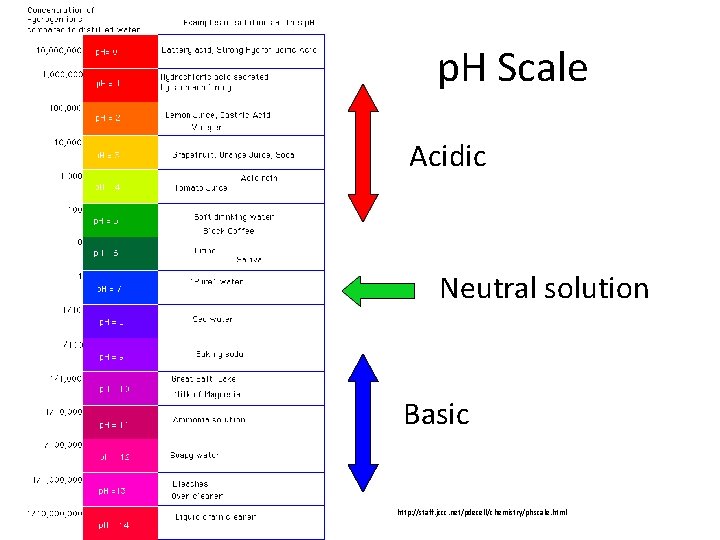
p. H Scale Acidic Neutral solution Basic http: //staff. jccc. net/pdecell/chemistry/phscale. html

Strong vs. Weak Acids and Bases • Strong Acid or Base: all of the molecules dissociate (split apart) into ions • Weak Acid or Base: Only some of the molecules actually dissociate – Not as many H+ or OH- ions in solution
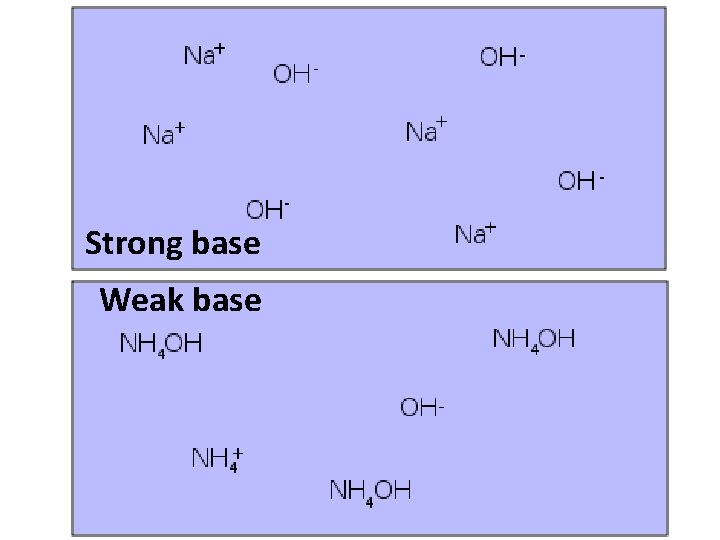
Strong base Weak base

Soap! • Let’s review the soap procedure… • SAFETY! What will you use to protect yourself? GOGGLES FOR EVERYONE! Gloves for the person who pours the Na. OH and mixes it. • How hot will you make the Na. OH and Oils? 50 degrees! They both need to be around 50 (+ or – a couple degrees)
- Slides: 29