QA and CQI How Merri L Bremer MEd

QA and CQI: How? Merri L. Bremer MEd, RN, RDCS, FASE

Disclosures Relevant Relationship Member, ICAEL Board of Directors Off Label Usage None

Learning Objectives Define QA Discuss ideas for development and implementation of Echo Lab QA

QA: What is it? Many names (QA, QI, CQI) Method of continuously examining processes and making them more effective Focus is on the process, not the individual

QA: What is it not? Punitive Demeaning Demoralizing Divisive Busywork OR…. .

When you watch me, they want me to do it differently…

Benefits Develops and maintains quality in your practice Ensures uniform, consistent standards for interpretation and reporting Excellent continuing education tool

Standards and Guidelines ICAEL Standards ASE Guidelines and Standards SDMS Position Statements ASE Sonographer Minimum Standards

ICAEL QA Components Written policy AUC Instrument maintenance Procedure volumes CME Peer review Correlation Report Timeliness Conferences Record keeping

Writing a QA Policy Identify required elements (ICAEL Standards) Figure out how YOUR TEAM can accomplish them and write them down Sample policies on ICAEL website Try them…. revise and try again if necessary Communicate! Frequently!

Appropriate Use Criteria (AUC) Mandatory requirement for accreditation effective January 1, 2012 Appropriate use must be measured in a minimum of 30 consecutive TTE, 30 consecutive TEE and 30 consecutive Stress patients annually ACCF/ASE/ACEP/ASNC/SCAI/SCCT/SCMR 2007 Appropriateness Criteria for Transthoracic and Transesophageal Echocardiography Percentage of appropriate, inappropriate and uncertain indications for testing must be measured
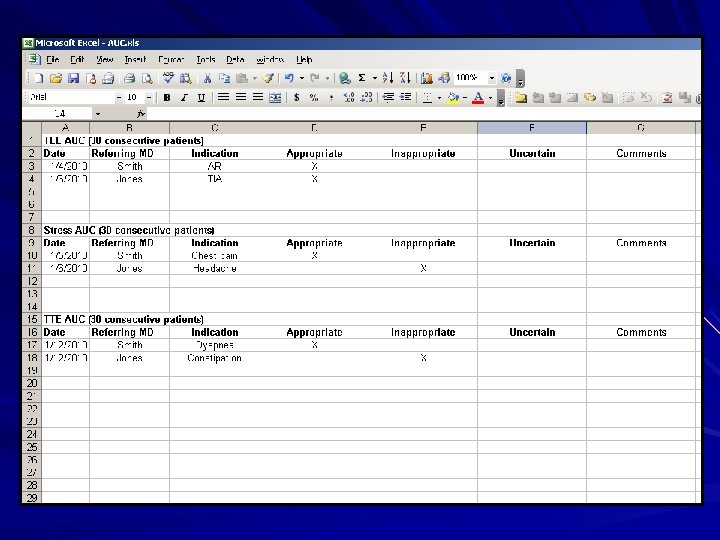

Appropriate Use Criteria A program for education and reporting must be developed and include: – – – Baseline rates of adherence Patterns of adherence Goals for improvement Measurement of improvement Confidential reports on patterns of adherence Ordering physician Ordering practice Interpreting practice

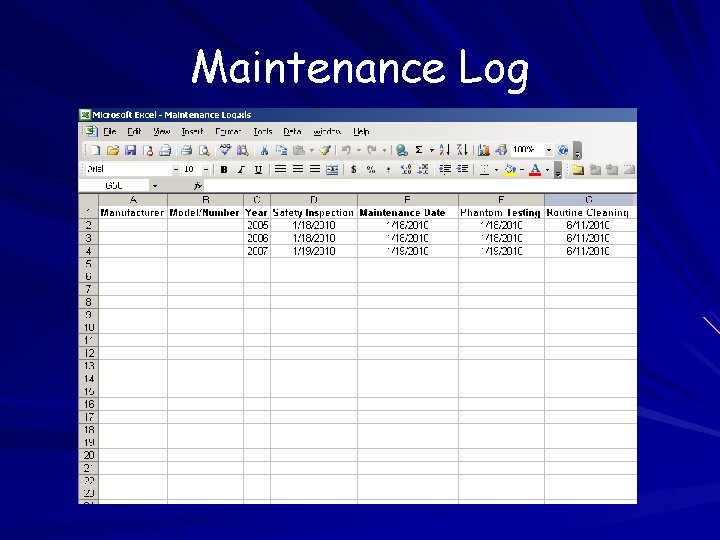
Instrument Maintenance Recording of method and frequency of maintenance Establishment of and adherence to a policy regarding routine safety inspections and testing of all laboratory electrical equipment Establishment of and adherence to an instrument cleaning schedule

Instrument Maintenance Use institutional resources if you have them If you don’t have them, create a policy using the manufacturer’s guidelines and follow it Ask your equipment reps for help!
Maintenance Log

Procedure Volumes Annual individual and laboratory stats Records of individual procedure volumes should include volumes from all laboratories where staff perform/interpret echocardiograms Methods of tracking – Schedule – Procedure list – Billing
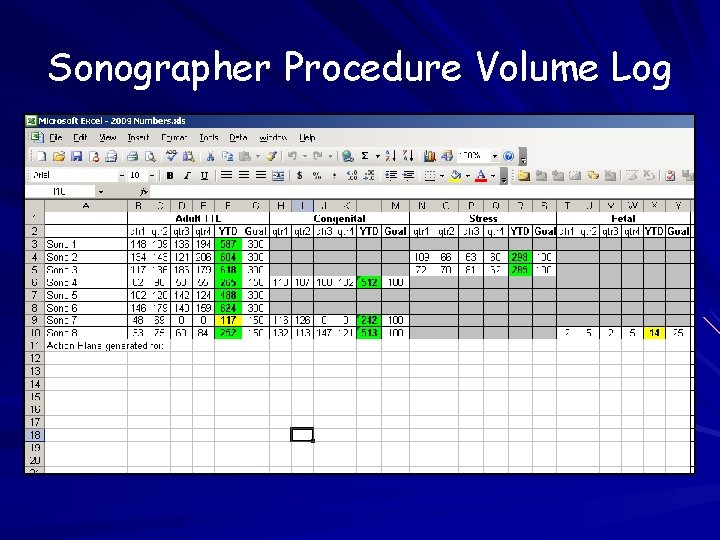
Sonographer Procedure Volume Log
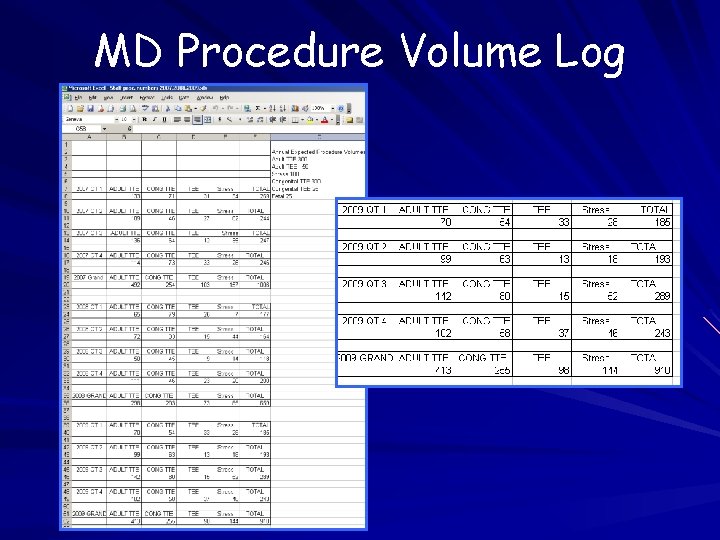
MD Procedure Volume Log

CME Documentation of echocardiographyrelated continuing education for all medical and technical personnel must be maintained Keep in central location; update annually Materials – CD, journal, Internet, videotape materials – Departmental, local, regional and national conferences and courses

CME 15 echo-related CME credits required for all staff (3 year period) Category 1 AMA credit Other approved non-category 1 credit (ASE, SDMS or ARRT) that have content specific to echocardiography
CME Log

Peer Review Feedback is essential for improvement! Intermittent peer review of both performance and interpretation of studies should be performed Optional QA measure, but very useful Both physicians and sonographers should be involved

Peer Review Differences in interpretation styles and performance should be reconciled Individual vs group reviews Confidentiality Document it!

MD Peer Review

Variability EF, wall motion analysis and degree of regurgitation/stenosis must be assessed on a minimum of two cases per modality per quarter to be reviewed in quarterly conferences Represent as many physicians as possible Policy to address discrepancies
Variability Worksheet
Variability Summary

Correlation EF, wall motion analysis and degree of regurgitation/stenosis will be correlated on a minimum of two per modality per quarter with other imaging modalities in quarterly conferences Represent as many physicians as possible Policy to address discrepancies

http: //www. icael. org/icael/pdfs/Correlation_Form. pdf

Report Review Minimum of 10 random reports per quarter Time from performance of study to report sign-off – Inpatient: 24 hours – Outpatient: end of next business day Report completeness (Standards) Represent as many physicians as possible Policy to address discrepancies

QA Conferences Quarterly conferences must be held to review the results of variability, correlation and report timelines, to address discrepancies and to discuss difficult cases Attendance by the medical and technical directors or their designees is required at all meetings

QA Conferences All medical and technical staff are required to attend at least two of the four meetings Minutes of the meetings and attendance must be recorded

Record Keeping If you don’t document it, it didn’t happen Keep data in a central location and back it up Annual summary of information required

Requirements for Success Leadership Commitment BUY-IN Individual Commitment of Resources

Accreditation/QA Resources http: //asecho. org/ http: //www. icael. org/icael/index. htm http: //www. sdms. org/ http: //www. asq. org/learn-aboutquality/index. html

Merri’s Rules for QA Keep it SIMPLE and practical Involve lots of people and ideas Steal shamelessly from others Adapt what you’ve stolen Be methodical Document Share what you’ve found
- Slides: 39