Pyrimidine Metabolism and Cancer Therapy LEARNING OBJECTIVES 1

Pyrimidine Metabolism and Cancer Therapy

LEARNING OBJECTIVES 1. Nomenclature of pyrimidines* 2. Key features of biosynthetic pathway and origin of atoms in pyrimidine ring 3. Basis of chemotherapy using nucleotide analogs *Key words are highlighted in yellow

Pyrimidine Synthesis • The pyrimidine ring is completely synthesized, then attached to a ribose-5 -phosphate donated by PRPP • Source of carbons and nitrogens less diverse than with purines.
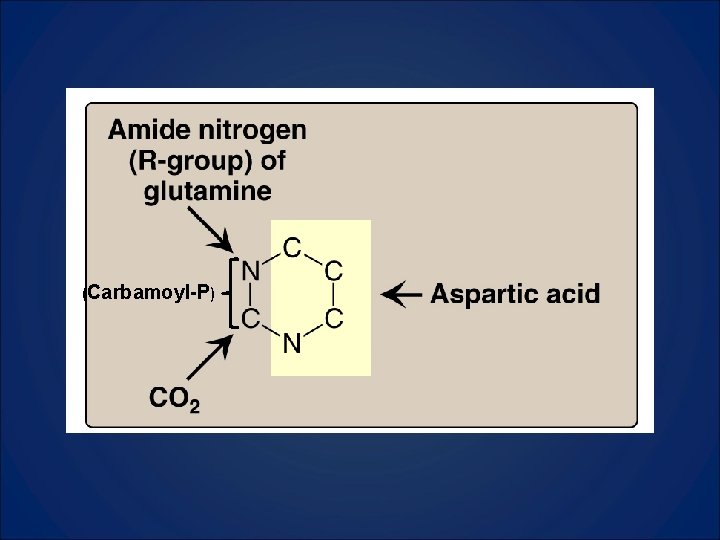
(Carbamoyl-P)
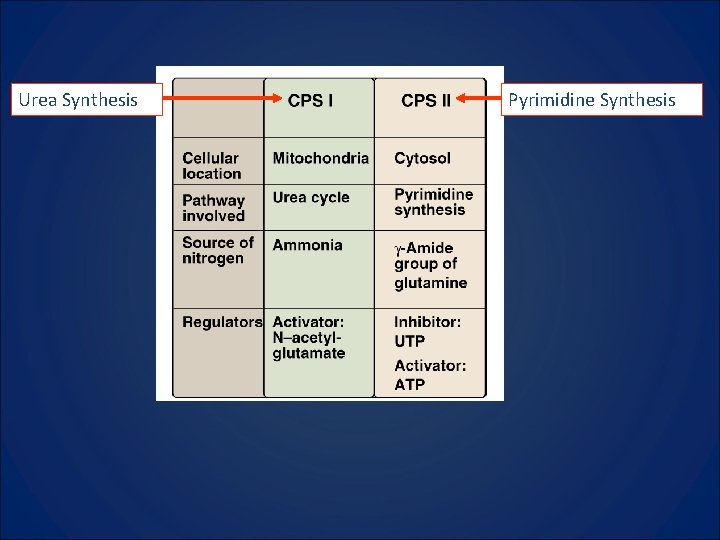
Urea Synthesis Pyrimidine Synthesis
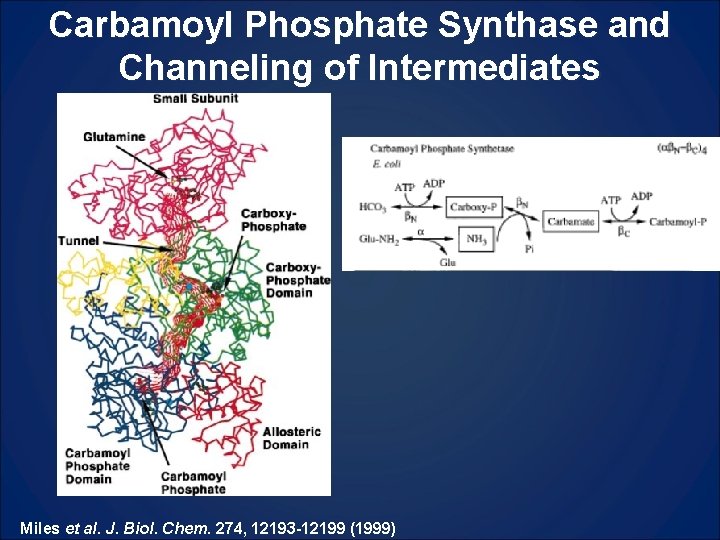
Carbamoyl Phosphate Synthase and Channeling of Intermediates Miles et al. J. Biol. Chem. 274, 12193 -12199 (1999)
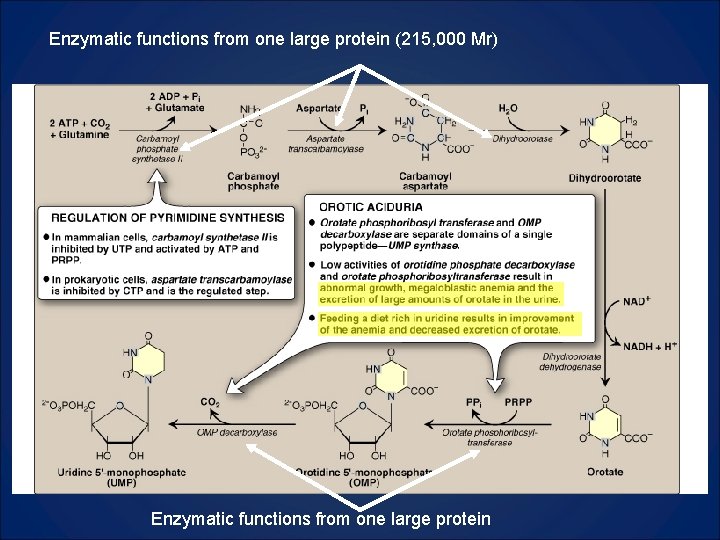
Enzymatic functions from one large protein (215, 000 Mr) Enzymatic functions from one large protein

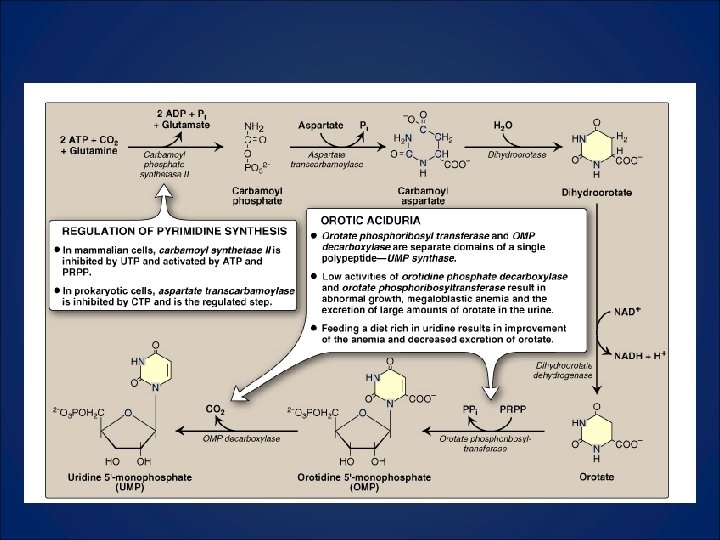
What to Know 1. Compare and contrast CPS I and CPS II 2. CPSII, aspartate transcarbamoylase, and dihydroorotase are three enzymatic functions in one protein. 3. Oratate phosphoribosyltransferase and OMP decarboxylase are two enzymatic functions in one protein. Deficiency leads to Orotic Aciduria. Know symptoms and how to treat. 4. Orotate is made and then attached to a PRPP.
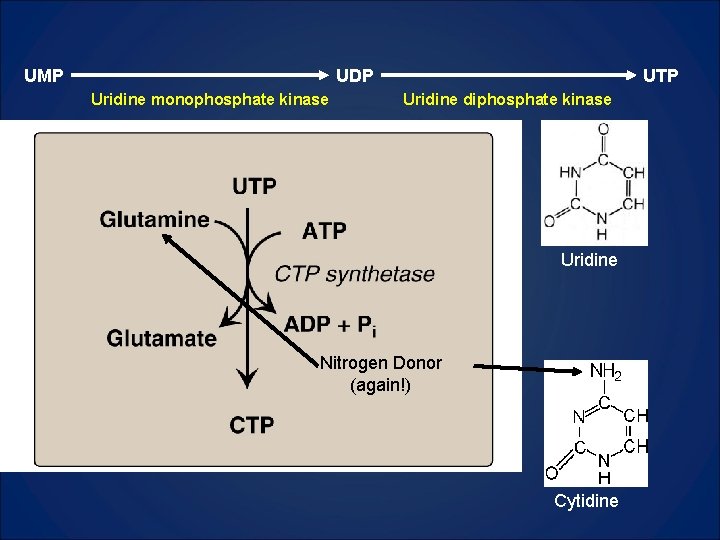
UMP UDP Uridine monophosphate kinase UTP Uridine diphosphate kinase Uridine Nitrogen Donor (again!) Cytidine
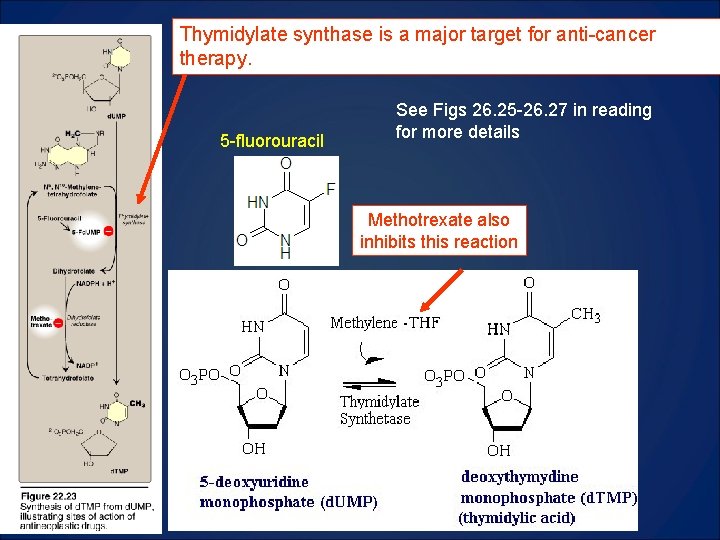
Thymidylate synthase is a major target for anti-cancer therapy. 5 -fluorouracil See Figs 26. 25 -26. 27 in reading for more details Methotrexate also inhibits this reaction
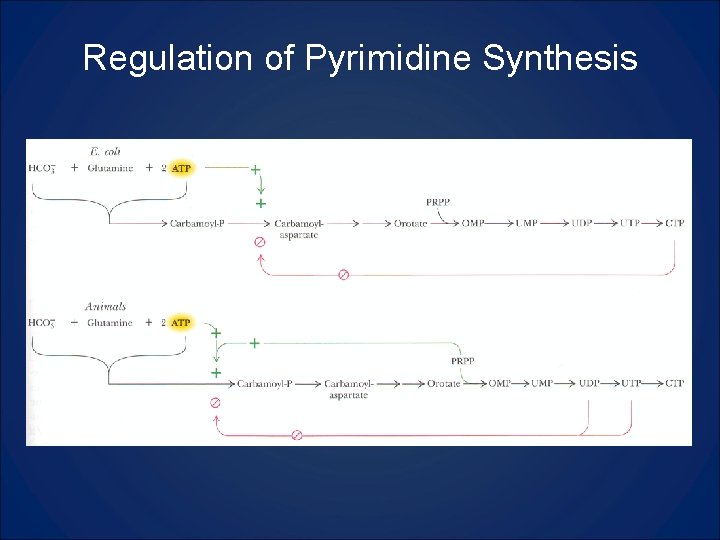
Regulation of Pyrimidine Synthesis
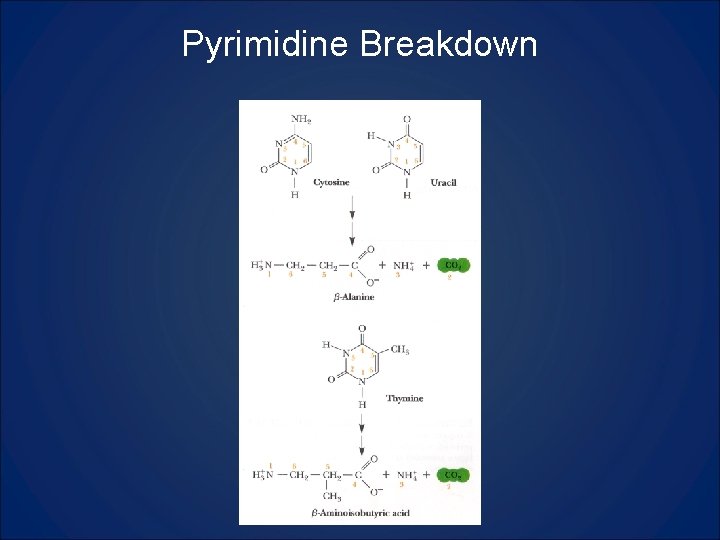
Pyrimidine Breakdown
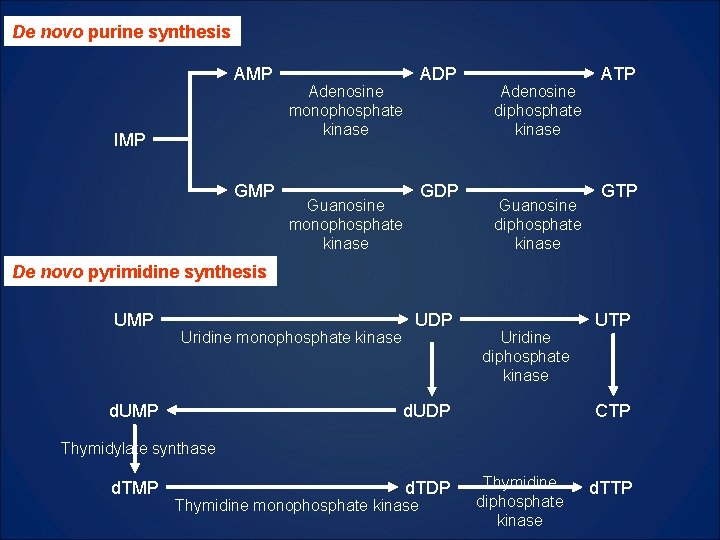
De novo purine synthesis AMP IMP GMP ADP Adenosine monophosphate kinase GDP Guanosine monophosphate kinase Adenosine diphosphate kinase Guanosine diphosphate kinase ATP GTP De novo pyrimidine synthesis UMP Uridine monophosphate kinase d. UMP UDP Uridine diphosphate kinase d. UDP UTP CTP Thymidylate synthase d. TMP d. TDP Thymidine monophosphate kinase Thymidine diphosphate kinase d. TTP
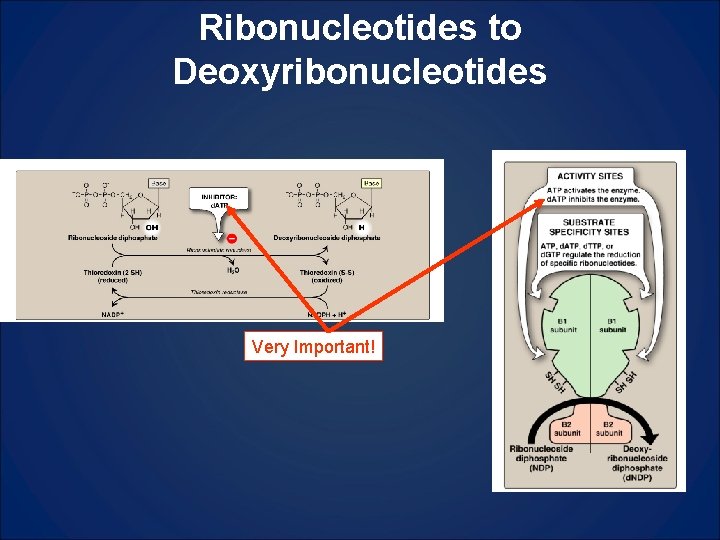
Ribonucleotides to Deoxyribonucleotides Very Important!
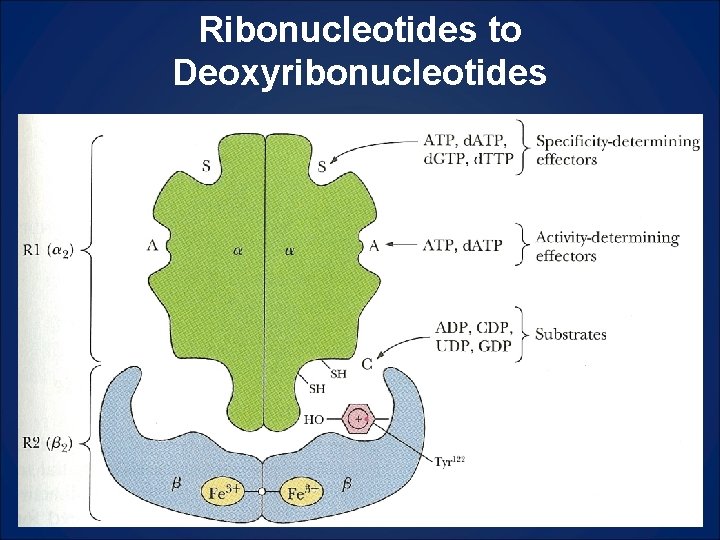
Ribonucleotides to Deoxyribonucleotides
![Logic For Deoxynucleotide Synthesis (Fig 26. 24) in reading • High [ATP], plenty of Logic For Deoxynucleotide Synthesis (Fig 26. 24) in reading • High [ATP], plenty of](http://slidetodoc.com/presentation_image_h2/d47819492e91ee6eb416d106d2e5abae/image-17.jpg)
Logic For Deoxynucleotide Synthesis (Fig 26. 24) in reading • High [ATP], plenty of energy, ok to make DNA • High [ATP] means the ribonucleotide reductase is active (ON) • ATP in specificity site S favors CDP or UDP in catalytic site C [d. CDP] and [d. UDP] ↑ • d. CDP and d. UDP become metabolized to d. TTP (thus justifying the synthesis of d. UMP even though it does not get incorporated into DNA) • As [d. TTP]↑, it will begin to occupy specificity site favoring GDP in catalytic site, thus leading to [d. GP]↑ [d. GTP]↑ • As [d. GTP]↑ it begins to occupy specificity site and thus favor ADP in catalytic site, leading to [d. ADP]↑ which will replace ATP in activity site and turn enzyme off
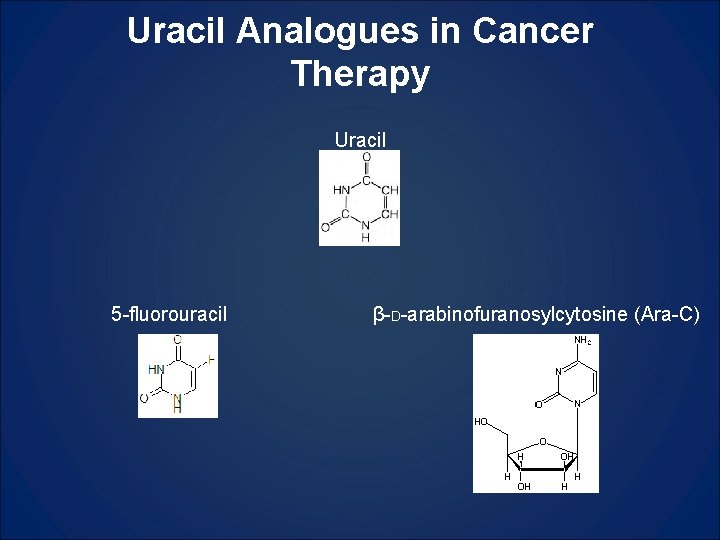
Uracil Analogues in Cancer Therapy Uracil 5 -fluorouracil β-D-arabinofuranosylcytosine (Ara-C)
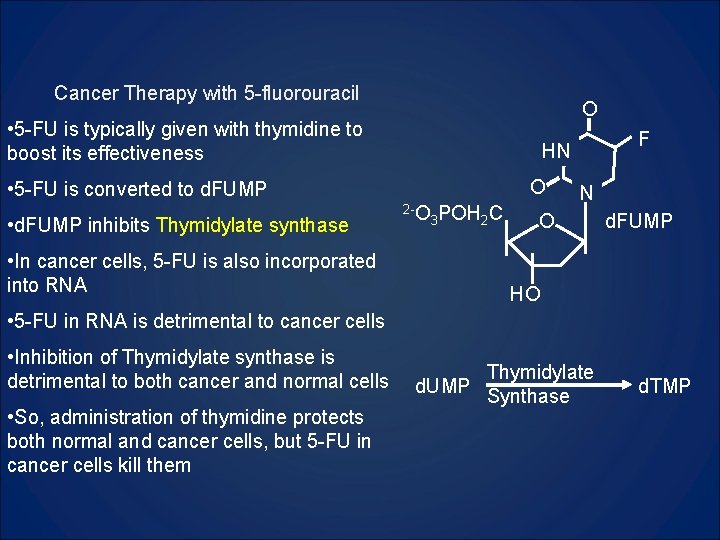
Cancer Therapy with 5 -fluorouracil O • 5 -FU is typically given with thymidine to boost its effectiveness O • 5 -FU is converted to d. FUMP • d. FUMP inhibits Thymidylate synthase • In cancer cells, 5 -FU is also incorporated into RNA F HN 2 -O 3 POH 2 C N O d. FUMP HO • 5 -FU in RNA is detrimental to cancer cells • Inhibition of Thymidylate synthase is detrimental to both cancer and normal cells • So, administration of thymidine protects both normal and cancer cells, but 5 -FU in cancer cells kill them Thymidylate d. UMP Synthase d. TMP
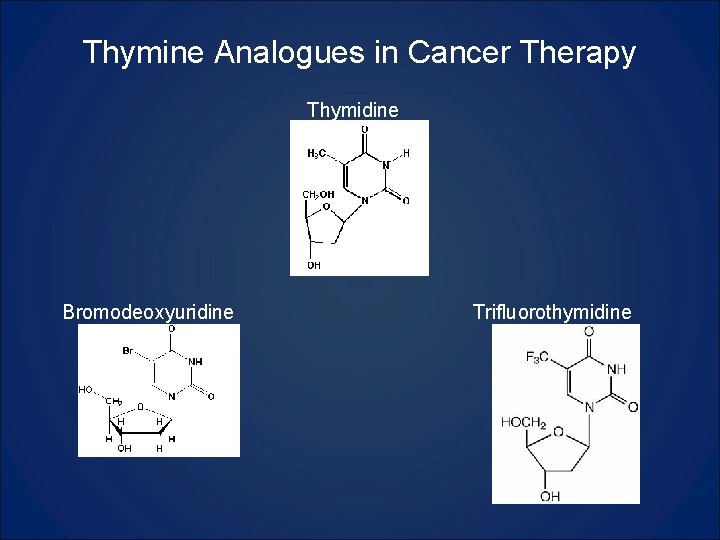
Thymine Analogues in Cancer Therapy Thymidine Bromodeoxyuridine Trifluorothymidine
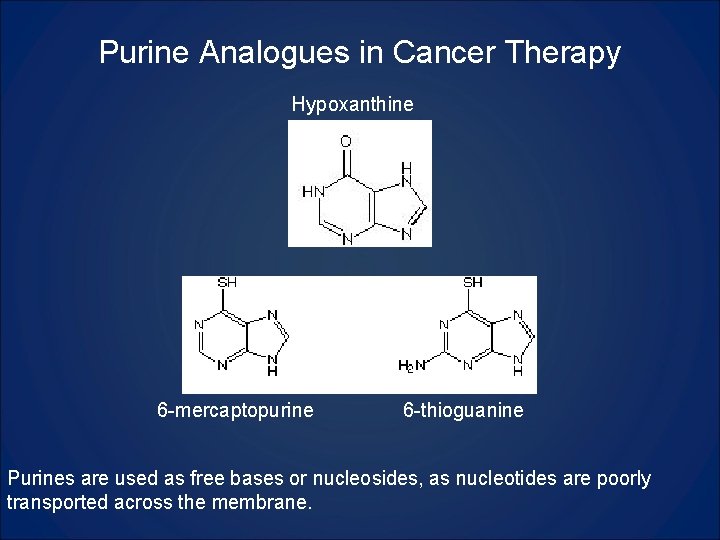
Purine Analogues in Cancer Therapy Hypoxanthine 6 -mercaptopurine 6 -thioguanine Purines are used as free bases or nucleosides, as nucleotides are poorly transported across the membrane.
- Slides: 21