PV Diagrams and processes Contents Basic Concept Example

P-V Diagrams and processes Contents: • Basic Concept • Example • Whiteboards
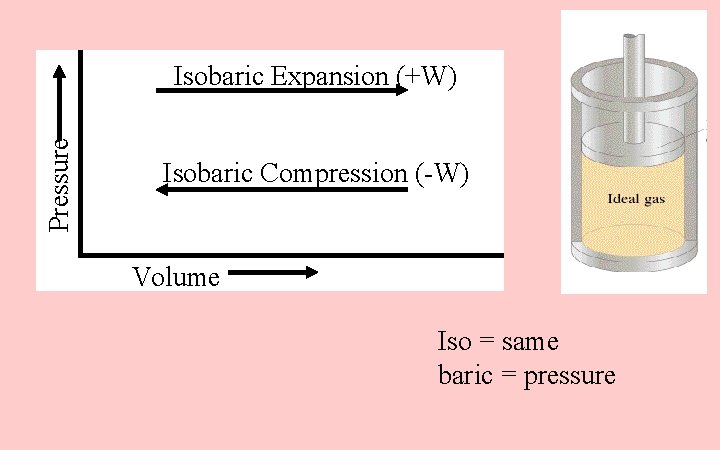
Pressure Isobaric Expansion (+W) Isobaric Compression (-W) Volume Iso = same baric = pressure
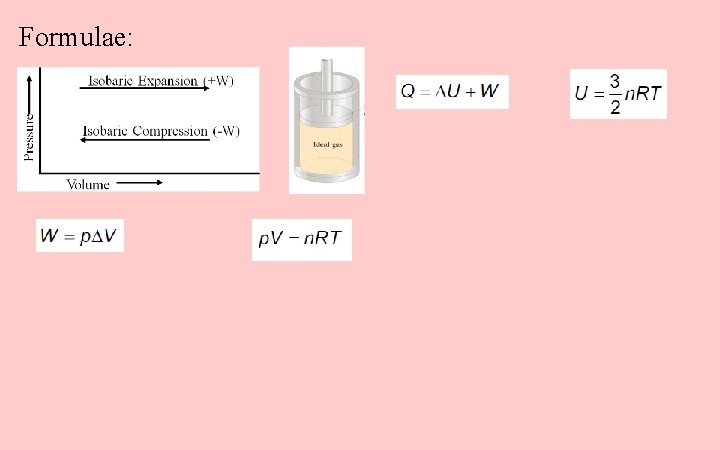
Formulae:
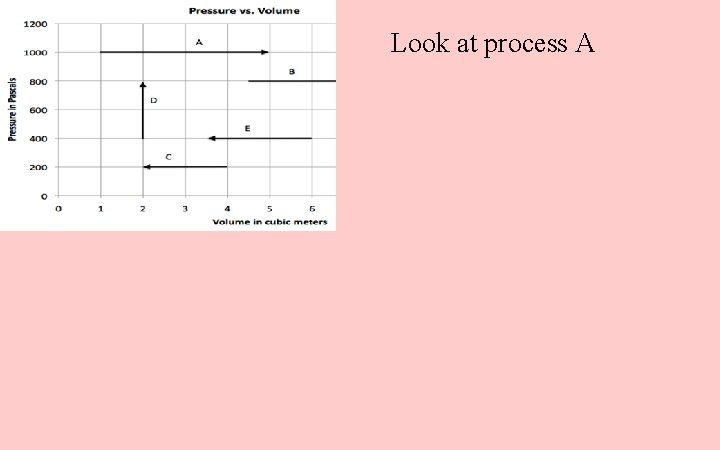
Look at process A
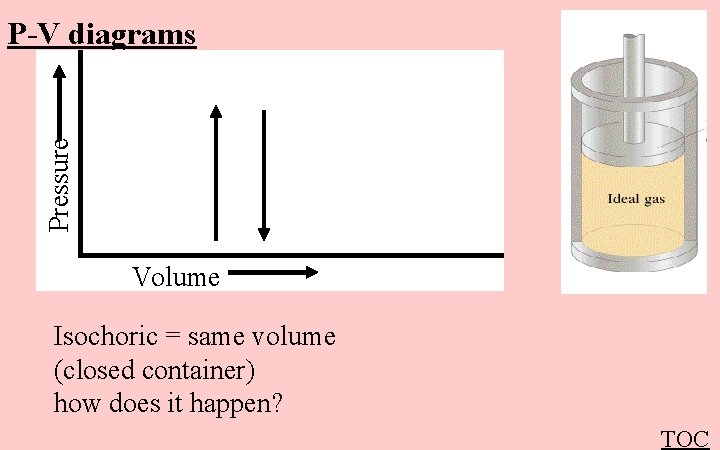
Pressure P-V diagrams Volume Isochoric = same volume (closed container) how does it happen? TOC
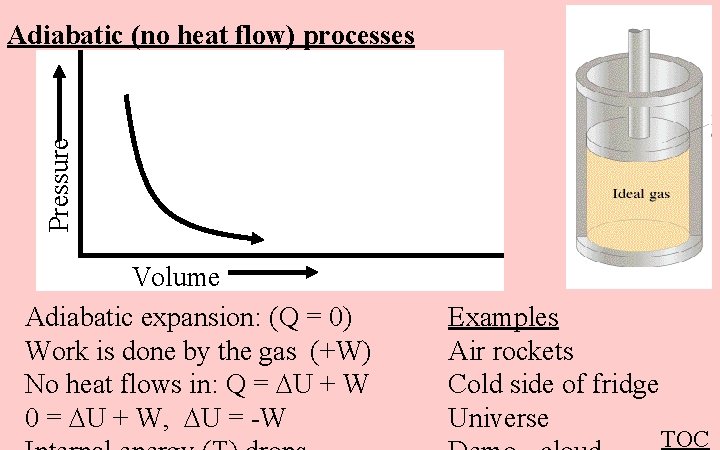
Pressure Adiabatic (no heat flow) processes Volume Adiabatic expansion: (Q = 0) Work is done by the gas (+W) No heat flows in: Q = U + W 0 = U + W, U = -W Examples Air rockets Cold side of fridge Universe TOC
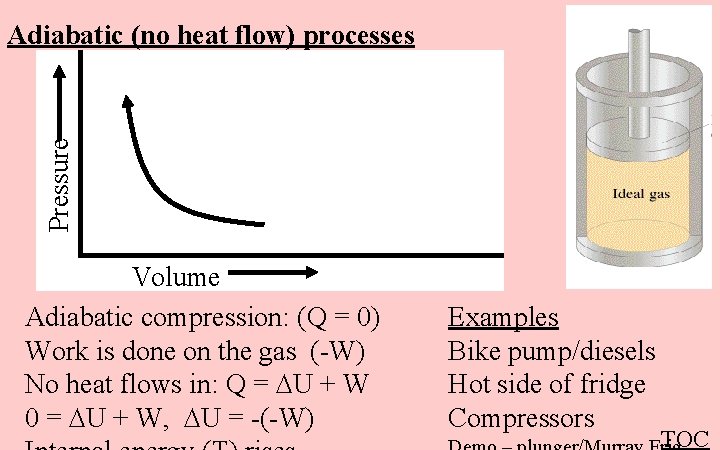
Pressure Adiabatic (no heat flow) processes Volume Adiabatic compression: (Q = 0) Work is done on the gas (-W) No heat flows in: Q = U + W 0 = U + W, U = -(-W) Examples Bike pump/diesels Hot side of fridge Compressors TOC
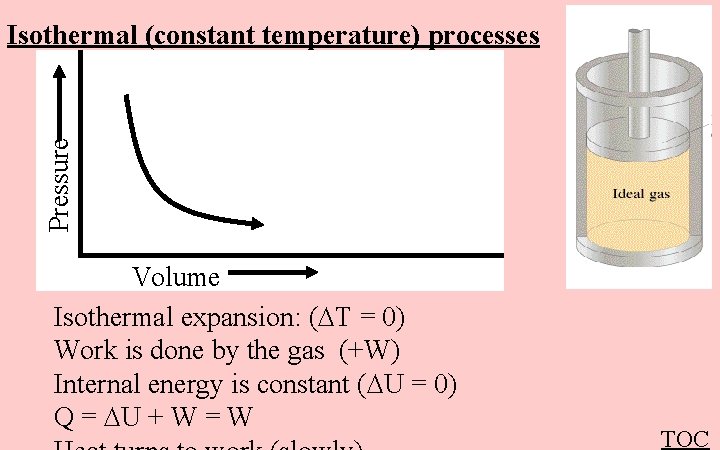
Pressure Isothermal (constant temperature) processes Volume Isothermal expansion: ( T = 0) Work is done by the gas (+W) Internal energy is constant ( U = 0) Q = U + W = W TOC

Pressure Isothermal (constant temperature) processes Volume Isothermal compression: ( T = 0) Work is done on the gas (-W) Internal energy is constant ( U = 0) Q = U + W = W TOC
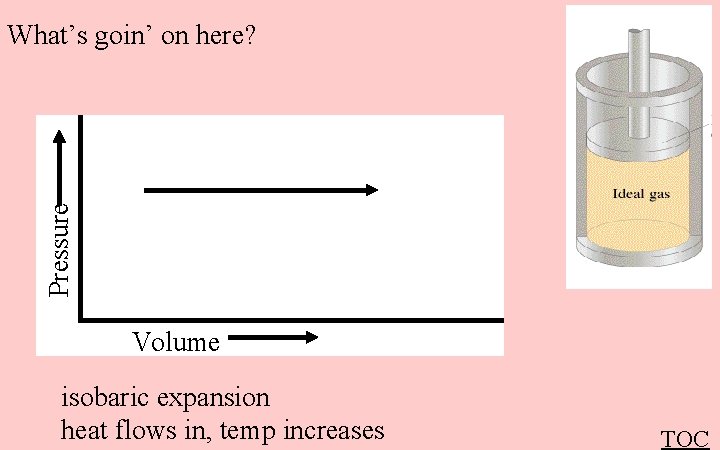
Pressure What’s goin’ on here? Volume isobaric expansion heat flows in, temp increases TOC
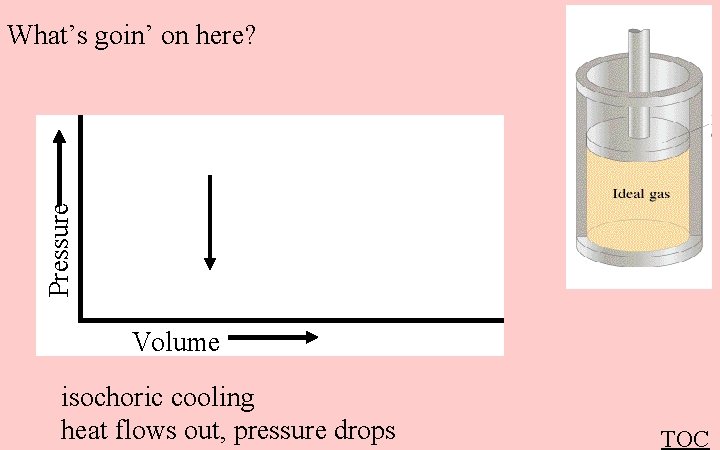
Pressure What’s goin’ on here? Volume isochoric cooling heat flows out, pressure drops TOC
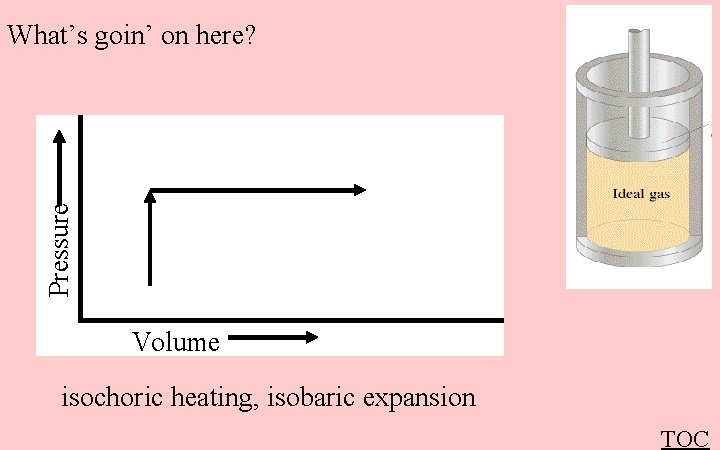
Pressure What’s goin’ on here? Volume isochoric heating, isobaric expansion TOC
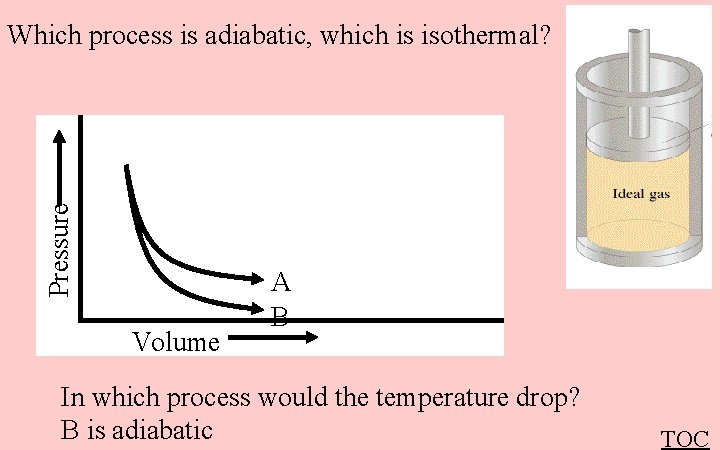
Pressure Which process is adiabatic, which is isothermal? Volume A B In which process would the temperature drop? B is adiabatic TOC
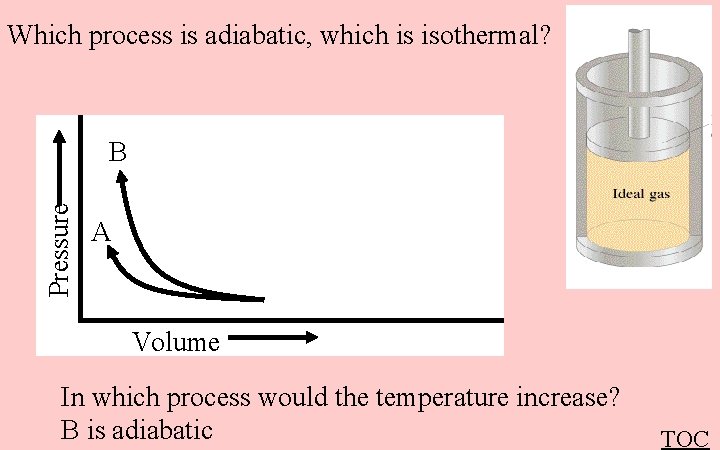
Which process is adiabatic, which is isothermal? Pressure B A Volume In which process would the temperature increase? B is adiabatic TOC
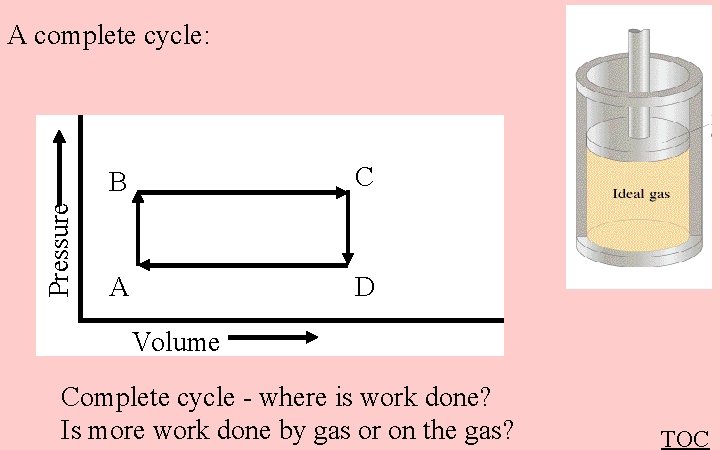
Pressure A complete cycle: B C A D Volume Complete cycle - where is work done? Is more work done by gas or on the gas? TOC
- Slides: 15